-
question_answer1)
| Directions: (Q. 1 to 5) |
| Case I: Read the passage given below and answer the following questions from 1 to 5. |
| The solubility of gases increases with increase of pressure. William Henry made a systematic investigation of the solubility of a gas in a liquid. According to Henry's law "the mass of a gas dissolved per unit volume of the solvent at constant temperature is directly proportional to the pressure of the gas in equilibrium with the solution". |
| Dalton during the same period also concluded independently that the solubility of a gas in a liquid solution depends upon the partial pressure of the gas. If we use the mole fraction of gas in the solution as a measure of its solubility, then Henry's law can be modified as "the partial pressure of the gas in the vapour phase is directly proportional to the mole fraction of the gas in the solution". |
Henry's law constant for the solubility of methane in benzene at 298 K is \[4.27\times {{10}^{5}}\] mm Hg. The solubility of methane in benzene at 298 K under 760 mm Hg is
A)
\[4.27\times {{10}^{-5}}\] done
clear
B)
\[1.78\times {{10}^{-3}}\] done
clear
C)
\[4.27\times {{10}^{-3}}\] done
clear
D)
\[1.78\text{ }\times {{10}^{-5}}\] done
clear
View Solution play_arrow
-
question_answer2)
The partial pressure of ethane over a saturated solution containing \[6.56\times {{10}^{-2}}\text{ }g\] of ethane is 1 bar. If the solution contains \[5.00\times {{10}^{-2}}\text{ }g\]of ethane then what will be the partial pressure (in bar) of the gas?
A)
0.762 done
clear
B)
1.312 done
clear
C)
3.81 done
clear
D)
5.0 done
clear
View Solution play_arrow
-
question_answer3)
\[{{K}_{H}}\](kbar) values for \[A{{r}_{(g)}}\], \[C{{O}_{2(g)}}\], \[HCH{{O}_{(g)}}\]and \[C{{H}_{4(g)}}\] are 40.39, 1.67, \[1.83\times {{10}^{-5}}\]and 0.413 respectively. Arrange these gases in the order of their increasing solubility.
A)
\[HCHO<C{{H}_{4}}<C{{O}_{2}}<Ar\] done
clear
B)
\[HCHO<C{{O}_{2}}<C{{H}_{4}}<Ar\] done
clear
C)
\[Ar<C{{O}_{2}}<C{{H}_{4}}<HCHO\] done
clear
D)
\[Ar<C{{H}_{4}}<C{{O}_{2}}<HCHO\] done
clear
View Solution play_arrow
-
question_answer4)
When a gas is bubbled through water at 298 K, a very dilute solution of the gas is obtained. Henry's law constant for the gas at 298 K is 150 kbar. If the gas exerts a partial pressure of 2 bar, the number of millimoles of the gas dissolved in 1 L of water is
A)
0.55 done
clear
B)
0.87 done
clear
C)
0.37 done
clear
D)
0.66 done
clear
View Solution play_arrow
-
question_answer5)
Which of the following statements is correct?
A)
\[{{K}_{H}}\] increases with increase of temperature. done
clear
B)
\[{{K}_{H}}\] decreases with increase of temperature. done
clear
C)
\[{{K}_{H}}\] remains constant with increase of temperature. done
clear
D)
\[{{K}_{H}}\] first increases then decreases, with incresase of temperature. done
clear
View Solution play_arrow
-
question_answer6)
| Directions: (Q. 6 to 10) |
| Case II: Read the passage given below and answer the following questions. |
| According to Raoult's law, the partial pressure of two components of the solution may be given as: |
| \[{{P}_{A}}=P_{A}^{{}^\circ }{{x}_{A}}\] and \[{{P}_{B}}=P_{B}^{{}^\circ }{{x}_{B}}\] |
| For an ideal solution (obeys Raoult's law always) |
| \[\Delta {{H}_{mix}}=0,\,\Delta {{V}_{mix}}=0\] |
| All solutions do not obey Raoult's law over entire range of concentration. These are known as non- ideal solutions. |
| For non-ideal solutions, \[{{P}_{A}}\ne P_{A}^{{}^\circ }{{x}_{A}}\] or \[{{P}_{B}}\ne P_{B}^{{}^\circ }{{x}_{B}}\] |
| Positive deviation\[\Rightarrow {{P}_{A}}>P_{A}^{{}^\circ }{{x}_{A}}\] and \[{{P}_{B}}>P_{A}^{{}^\circ }{{x}_{B}}\] |
| Negative deviation \[\Rightarrow {{P}_{A}}<P_{A}^{{}^\circ }{{x}_{A}}\] and \[{{P}_{B}}<P_{B}^{{}^\circ }{{x}_{B}}\] |
| In the following questions (Q. No. 6-10), a statement of assertion followed by a statement of reason is given. Choose the correct answer out of the following choices on the basis of the above passage. |
| |
| Assertion: An ideal solution obeys Raoult's law. |
| Reason: In an ideal solution, solute-solute as well as solvent-solvent interactions are similar to solute-solvent interactions. |
A)
Assertion and reason both are correct statements and reason is correct explanation for assertion. done
clear
B)
Assertion and reason both are correct statements but reason is not correct explanation for assertion. done
clear
C)
Assertion is correct statement but reason is wrong statement. done
clear
D)
Assertion is wrong statement but reason is correct statement. done
clear
View Solution play_arrow
-
question_answer7)
| Assertion: Acetone and aniline show negative deviations. |
| Reason: H-bonding between acetone and aniline is stronger than that between acetone-acetone and aniline-aniline. |
A)
Assertion and reason both are correct statements and reason is correct explanation for assertion. done
clear
B)
Assertion and reason both are correct statements but reason is not correct explanation for assertion. done
clear
C)
Assertion is correct statement but reason is wrong statement. done
clear
D)
Assertion is wrong statement but reason is correct statement. done
clear
View Solution play_arrow
-
question_answer8)
| Assertion: Azeotropic mixtures are formed only by non-ideal solutions and they may have boiling points either greater than both the components or lesser than both the components. |
| Reason: The composition of the vapour phase is same as that of the liquid phase of an azeotropic mixture. |
A)
Assertion and reason both are correct statements and reason is correct explanation for assertion. done
clear
B)
Assertion and reason both are correct statements but reason is not correct explanation for assertion. done
clear
C)
Assertion is correct statement but reason is wrong statement. done
clear
D)
Assertion is wrong statement but reason is correct statement. done
clear
View Solution play_arrow
-
question_answer9)
| Assertion: The solutions which show negative deviations from Raoult's law are called maximum boiling azeotropes. |
| Reason: 68% nitric acid and 32% water by mass form maximum boiling azeotrope. |
A)
Assertion and reason both are correct statements and reason is correct explanation for assertion. done
clear
B)
Assertion and reason both are correct statements but reason is not correct explanation for assertion. done
clear
C)
Assertion is correct statement but reason is wrong statement. done
clear
D)
Assertion is wrong statement but reason is correct statement. done
clear
View Solution play_arrow
-
question_answer10)
| Assertion: \[\Delta {{H}_{mix}}\] and \[\Delta {{V}_{mix}}\]are positive for an ideal solution. |
| Reason: The interactions between the particle of the components of an ideal solution are almost identical as between particles in the liquids. |
A)
Assertion and reason both are correct statements and reason is correct explanation for assertion. done
clear
B)
Assertion and reason both are correct statements but reason is not correct explanation for assertion. done
clear
C)
Assertion is correct statement but reason is wrong statement. done
clear
D)
Assertion is wrong statement but reason is correct statement. done
clear
View Solution play_arrow
-
question_answer11)
| Directions: (Q. 11 to 15) |
| Case III: Read the passage given below and answer the following questions from 11 to 15. |
| Few colligative properties are : |
| Relative lowering of vapour pressure: Depends only on molar concentration of solute (mole fraction) and independent of its nature. |
| Depression in freezing point: It is proportional to the molal concentration of solution. |
| Elevation of boiling point: It is proportional to the molal concentration of solute. |
| Osmotic pressure: It is proportional to the molar concentration of solute. |
| A solution of glucose is prepared with 0.052 g at glucose in 80.2 g of water. (\[{{K}_{f}}=1.86\text{ }K\text{ }kg\,mo{{l}^{-1}}\] and \[{{K}_{b}}=5.2K\,kg\,mo{{l}^{-1}}\]) |
Molality of the given solution is
A)
0.0052m done
clear
B)
0.0036m done
clear
C)
0.0006 m done
clear
D)
1.29 m done
clear
View Solution play_arrow
-
question_answer12)
Boiling point for the solution will be
A)
373.05 K done
clear
B)
373.15 K done
clear
C)
373.02 K done
clear
D)
372.98 K done
clear
View Solution play_arrow
-
question_answer13)
The depression in freezing point of solution will be
A)
0.0187 K done
clear
B)
0.035 K done
clear
C)
0.082 K done
clear
D)
0.067 K done
clear
View Solution play_arrow
-
question_answer14)
Mole fraction of glucose in the given solution is
A)
\[6.28\times {{10}^{-5}}\] done
clear
B)
\[1.23\times {{10}^{-\,4}}\] done
clear
C)
0.00625 done
clear
D)
0.00028 done
clear
View Solution play_arrow
-
question_answer15)
If same amount of sucrose \[\left( {{C}_{12}}{{H}_{22}}{{O}_{11}} \right)\] is taken instead of glucose then
A)
elevation in boiling point will be higher done
clear
B)
depression in freezing point will be higher done
clear
C)
depression in freezing point will be lower done
clear
D)
both [a] and [b]. done
clear
View Solution play_arrow
-
question_answer16)
| Directions: (Q. 16 to 20) |
| Case IV: Read the passage given below and answer the following questions from 16 to 20. |
| An ideal solution may be defined as the solution which obeys Raoult's law exactly over the entire range of concentration. The solutions for which vapour pressure is either higher or lower than that predicted by Raoult's law are called non- ideal solutions. |
| Non-ideal solutions can show either positive or negative deviations from Raoult's law depending on whether the A-B interactions in solution are stronger or weaker than A-A and B-B interactions. |
| Which of the following solutions is/are ideal solution(s)? |
| (I) Bromoethane and iodoethane |
| (II) Acetone and chloroform |
| (III) Benzene and acetone |
| (IV) n-Heptane and n-Hexane |
A)
Only I done
clear
B)
I and II done
clear
C)
II and III done
clear
D)
I and IV done
clear
View Solution play_arrow
-
question_answer17)
For which of the following solutions \[\Delta {{H}_{mix}}\]and \[\Delta {{V}_{mix}}\]is negative?
A)
Acetone and aniline done
clear
B)
Ethyl alcohol and cyclohexane done
clear
C)
Acetone and \[C{{S}_{2}}\] done
clear
D)
Benzene and toluene done
clear
View Solution play_arrow
-
question_answer18)
Which of the following is not true for positive deviations?
A)
The AB interactions in solution are weaker than the \[A-A\]and \[B-B\]interactions. done
clear
B)
\[{{P}_{A}}<P_{A}^{{}^\circ }{{x}_{A}}\] and \[{{P}_{B}}<P_{B}^{{}^\circ }{{x}_{B}}\] done
clear
C)
Carbon tetrachloride and chloroform mixture is an example of positive deviations. done
clear
D)
All of these. done
clear
View Solution play_arrow
-
question_answer19)
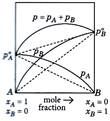
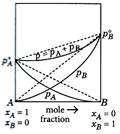
For water and nitric acid mixture which of the given graph is correct?
A)
B)
C)
Both of these done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer20)
| Water-HCl mixture |
| I. shows positive deviations |
| II. forms minimum boiling azeotrope |
| III. shows negative deviations |
| IV. forms maximum boiling azeotrope. |
A)
I and II done
clear
B)
II and III done
clear
C)
I and IV done
clear
D)
III and IV done
clear
View Solution play_arrow
-
question_answer21)
| Directions: (Q. 21 to 25) |
| Case V: Read the passage given below and answer the following questions. |
| The phenomenon of the flow of solvent through a semipermeable membrane from pure solvent to the solution is called osmosis. |
| Sometimes a pressure is applied to stop the process of osmosis, this is known as osmotic pressure. It is denoted by \[\pi \]. Osmotic pressure is expressed as : \[\pi \] = CRT. |
| Since, osmotic pressure depends upon the molar concentration of solution, therefore it is a colligative property. |
| In the following questions (Q. No. 21-25), a statement of assertion followed by a statement of reason is given. Choose the correct answer out of the following choices on the basis of the above passage. |
| |
| Assertion: If red blood cells were removed from the body and placed in pure water, pressure inside the cells increases. |
| Reason: The concentration of salt content in the cells increases. |
A)
Assertion and reason both are correct statements and reason is correct explanation for assertion. done
clear
B)
Assertion and reason both are correct statements but reason is not correct explanation for assertion. done
clear
C)
Assertion is correct statement but reason is wrong statement. done
clear
D)
Assertion is wrong statement but reason is correct statement. done
clear
View Solution play_arrow
-
question_answer22)
| Assertion: The osmotic pressure of a solution obtained by mixing 100 mL of 3.4% solution of urea and 100 mL of 1.6% solution of cane sugar at 293 K is 7.46 bar. |
| Reason: The total osmotic pressure will be equal to the sum of partial osmotic pressures. |
A)
Assertion and reason both are correct statements and reason is correct explanation for assertion. done
clear
B)
Assertion and reason both are correct statements but reason is not correct explanation for assertion. done
clear
C)
Assertion is correct statement but reason is wrong statement. done
clear
D)
Assertion is wrong statement but reason is correct statement. done
clear
View Solution play_arrow
-
question_answer23)
| Assertion: When a solution is separated from the pure solvent by a semipermeable membrane, the solvent molecules pass through it from pure solvent side to the solution side. |
| Reason: Diffusion of solvent occurs from a region of high concentration to a region of low concentration solution. |
A)
Assertion and reason both are correct statements and reason is correct explanation for assertion. done
clear
B)
Assertion and reason both are correct statements but reason is not correct explanation for assertion. done
clear
C)
Assertion is correct statement but reason is wrong statement. done
clear
D)
Assertion is wrong statement but reason is correct statement. done
clear
View Solution play_arrow
-
question_answer24)
| Assertion: Two solutions having same osmotic pressure at a given temperature are called isotonic solutions. |
| Reason: Osmotic pressure is not a colligative property. |
A)
Assertion and reason both are correct statements and reason is correct explanation for assertion. done
clear
B)
Assertion and reason both are correct statements but reason is not correct explanation for assertion. done
clear
C)
Assertion is correct statement but reason is wrong statement. done
clear
D)
Assertion is wrong statement but reason is correct statement. done
clear
View Solution play_arrow
-
question_answer25)
| Assertion: The preservation of meat by salting and fruits by adding sugar protects against bacterial action. |
| Reason: A bacterium on salted meat or candid fruit loses water due to osmosis shrivels and ultimately dies. |
A)
Assertion and reason both are correct statements and reason is correct explanation for assertion. done
clear
B)
Assertion and reason both are correct statements but reason is not correct explanation for assertion. done
clear
C)
Assertion is correct statement but reason is wrong statement. done
clear
D)
Assertion is wrong statement but reason is correct statement. done
clear
View Solution play_arrow
 done
clear
done
clear
 done
clear
done
clear
