-
question_answer1)
The function of anhydrous \[AlC{{l}_{3}}\] in the Friedel-Craft?s reaction is to [MNR 1986, 1995; Roorkee 1999; BHU 2001; CPMT 2002; MPPET 2001]
A)
Absorb water done
clear
B)
Absorb HCl done
clear
C)
To produce electrophile done
clear
D)
To produce nucleophile done
clear
View Solution play_arrow
-
question_answer2)
Benzene reacts with \[C{{H}_{3}}COCl\] in the presence of \[AlC{{l}_{3}}\] to give [DPMT 1983; CBSE PMT 1991]
A)
\[{{C}_{6}}{{H}_{5}}Cl\] done
clear
B)
\[{{C}_{6}}{{H}_{5}}COCl\] done
clear
C)
\[{{C}_{6}}{{H}_{5}}C{{H}_{3}}\] done
clear
D)
\[{{C}_{6}}{{H}_{5}}COC{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer3)
Acylation process is preferred than direct alkylation because (by the Friedel-Craft's reaction)
A)
In alkylation, a poisonous gas is evolved done
clear
B)
In alkylation, large amount of heat is evolved done
clear
C)
In alkylation, polyalkylated product is formed done
clear
D)
Alkylation is very costly done
clear
View Solution play_arrow
-
question_answer4)
Benzene cannot undergo
A)
Substitution done
clear
B)
Addition done
clear
C)
Elimination done
clear
D)
Oxidation done
clear
View Solution play_arrow
-
question_answer5)
Coaltar is main source of [DPMT 1984]
A)
Aromatic compounds done
clear
B)
Aliphatic compounds done
clear
C)
Cycloalkanes done
clear
D)
Heterocyclic compounds done
clear
View Solution play_arrow
-
question_answer6)
Which of the following is not formed by the ozonolysis of o-xylene
A)
Glyoxal done
clear
B)
Ethyl glyoxal done
clear
C)
Dimethyl glyoxal done
clear
D)
Methyl glyoxal done
clear
View Solution play_arrow
-
question_answer7)
The number of \[\sigma \] and \[\pi \] bonds in a molecule of benzene is [MP PMT/PET 1988; BHU 1995; CPMT 1997]
A)
\[6\sigma \text{ and }9\pi \] done
clear
B)
\[9\sigma \text{ and }3\pi \] done
clear
C)
\[12\sigma \text{ and }3\pi \] done
clear
D)
\[6\sigma \text{ and }6\pi \] done
clear
View Solution play_arrow
-
question_answer8)
The ratio of \[\sigma \] and \[\pi \] bonds in benzene is [CPMT 1991; BHU 1995]
A)
2 done
clear
B)
4 done
clear
C)
6 done
clear
D)
8 done
clear
View Solution play_arrow
-
question_answer9)
Carbon atoms in benzene molecule is inclined at an angle of [BHU 1985]
A)
\[{{120}^{o}}\] done
clear
B)
\[{{180}^{o}}\] done
clear
C)
\[{{109}^{o}}2{8}?\] done
clear
D)
\[{{60}^{o}}\] done
clear
View Solution play_arrow
-
question_answer10)
When benzene is treated with excess of \[C{{l}_{2}}\] in the presence of \[{{I}_{2}}\], the end product is
A)
Monochlorobenzene done
clear
B)
Trichlorobenzene done
clear
C)
Hexachlorobenzene done
clear
D)
Benzene hexachloride done
clear
View Solution play_arrow
-
question_answer11)
Chemical name of the insecticide gammexene is [CPMT 1981; MP PET 1995; MP PMT 1996; CBSE PMT 1999; MP PET 1999]
A)
DDT done
clear
B)
Benzene hexachloride done
clear
C)
Chloral done
clear
D)
Hexachloroethane done
clear
View Solution play_arrow
-
question_answer12)
Gammexane is obtained from benzene when it reacts with
A)
\[B{{r}_{2}}\] in bright sunlight (in the absence of a catalyst) done
clear
B)
\[C{{l}_{2}}\] in bright sunlight (in the absence of a catalyst) done
clear
C)
\[C{{H}_{3}}Cl\] in the presence of anhydrous \[AlC{{l}_{3}}\] done
clear
D)
\[COC{{l}_{2}}\] in the presence of anhydrous \[AlC{{l}_{3}}\] done
clear
View Solution play_arrow
-
question_answer13)
Point out the wrong statement in relation to the structure of benzene
A)
It forms only one monosubstitution product done
clear
B)
The C - C bond distance in benzene is uniformly 1.397 Å done
clear
C)
It is a resonance hybrid of a number of canonical forms done
clear
D)
It has three delocalised \[\pi \]- molecular orbitals done
clear
View Solution play_arrow
-
question_answer14)
Which equation represents an example of Friedel-Craft?s reaction [MNR 1993; CPMT 1996]
A)
\[{{C}_{6}}{{H}_{6}}+{{C}_{2}}{{H}_{5}}Cl\xrightarrow{AlC{{l}_{3}}}{{C}_{6}}{{H}_{5}}{{C}_{2}}{{H}_{5}}+HCl\] done
clear
B)
\[{{C}_{2}}{{H}_{5}}OH+HCl\xrightarrow{ZnC{{l}_{2}}}{{C}_{2}}{{H}_{5}}Cl+{{H}_{2}}O\] done
clear
C)
\[{{C}_{6}}{{H}_{5}}Cl+C{{H}_{3}}COCl\xrightarrow{AlC{{l}_{3}}}{{C}_{6}}{{H}_{5}}COC{{H}_{3}}+C{{l}_{2}}\] done
clear
D)
\[{{C}_{2}}{{H}_{5}}Br+Mg\xrightarrow{Ether}{{C}_{2}}{{H}_{5}}Mgr\] done
clear
View Solution play_arrow
-
question_answer15)
The most stable carbonium ion among the following is [JIPMER 2002; AFMC 2002]
A)
\[{{C}_{6}}{{H}_{5}}\overset{+}{\mathop{C}}\,H{{C}_{6}}{{H}_{5}}\] done
clear
B)
\[{{C}_{6}}{{H}_{5}}\overset{+}{\mathop{C}}\,{{H}_{2}}\] done
clear
C)
\[C{{H}_{3}}\overset{+}{\mathop{C}}\,{{H}_{2}}\] done
clear
D)
\[{{C}_{6}}{{H}_{5}}C{{H}_{2}}\overset{+}{\mathop{C}}\,{{H}_{2}}\] done
clear
View Solution play_arrow
-
question_answer16)
The reaction of toluene with chlorine in presence of ferric chloride gives predominantly [IIT-JEE 1986; DCE 2000]
A)
Benzoyl chloride done
clear
B)
m-chlorotoluene done
clear
C)
Benzyl chloride done
clear
D)
o- and p-chlorotoluenes done
clear
View Solution play_arrow
-
question_answer17)
The product formed when toluene is heated in light with \[C{{l}_{2}}\] and in absence of halogen carrier is
A)
Benzotrichloride done
clear
B)
Gammexene done
clear
C)
Chlorobenzene done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer18)
Attacking or reactive or electrophilic species in nitration of benzene is or In the nitration of benzene with concentrated \[HN{{O}_{3}}\text{ and }{{H}_{2}}S{{O}_{4}}\] the attack on ring is made by [CBSE PMT 1994; MP PET 1996, 2000; Pb. PMT 1998; BHU 2001; BVP 2004; DCE 2003]
A)
\[NO_{2}^{-}\] done
clear
B)
\[NO_{2}^{+}\] done
clear
C)
\[NO_{3}^{-}\] done
clear
D)
\[N{{O}_{2}}\] done
clear
View Solution play_arrow
-
question_answer19)
Which of the following reactions takes place when a mixture of concentrated \[HN{{O}_{3}}\] and \[{{H}_{2}}S{{O}_{4}}\] reacts on benzene at \[350K\] [CPMT 1985]
A)
Sulphonation done
clear
B)
Nitration done
clear
C)
Hydrogenation done
clear
D)
Dehydration done
clear
View Solution play_arrow
-
question_answer20)
Nitration of benzene by nitric acid and sulphuric acid is [MNR 1989; CPMT 1990; BCECE 2005]
A)
Electrophilic substitution done
clear
B)
Electrophilic addition done
clear
C)
Nucleophilic substitution done
clear
D)
Free radical substitution done
clear
View Solution play_arrow
-
question_answer21)
Necessary conditions for halogenation are [CPMT 1976]
A)
Cold and dark done
clear
B)
Presence of halogen carrier done
clear
C)
Both A and B done
clear
D)
None done
clear
View Solution play_arrow
-
question_answer22)
\[{{C}_{6}}{{H}_{6}}+C{{H}_{3}}Cl\underset{AlC{{l}_{3}}}{\mathop{\xrightarrow{\text{anhydrous}}}}\,{{C}_{6}}{{H}_{5}}C{{H}_{3}}+HCl\] is an example of [NCERT 1979; CPMT 1974, 85, 90; Bihar CEE 1995; BHU 1979, 2001; MP PET 1995; MP PMT 1995; KCET 1993; EAMCET 1998; AIIMS 1998; CBSE PMT 2000; AFMC 2000; JIPMER 2000]
A)
Friedel-Craft's reaction done
clear
B)
Kolbe's synthesis done
clear
C)
Wurtz reaction done
clear
D)
Grignard reaction done
clear
View Solution play_arrow
-
question_answer23)
The reaction of benzene with chlorine in the presence of iron gives [MP PET 1993]
A)
Benzene hexachloride done
clear
B)
Chlorobenzene done
clear
C)
Benzyl chloride done
clear
D)
Benzoyl chloride done
clear
View Solution play_arrow
-
question_answer24)
Benzene was discovered by [NCERT 1981]
A)
Ramsay done
clear
B)
Dalton done
clear
C)
Faraday done
clear
D)
Priestley done
clear
View Solution play_arrow
-
question_answer25)
The correct structure of benzene was proposed by [CPMT 1972]
A)
Faraday done
clear
B)
Davy done
clear
C)
Kekule done
clear
D)
Wohler done
clear
View Solution play_arrow
-
question_answer26)
The centric structure of benzene was proposed by [CPMT 1982, 83, 89]
A)
Dewar done
clear
B)
Ladenberg done
clear
C)
Kekule done
clear
D)
Armstrong and Baeyer done
clear
View Solution play_arrow
-
question_answer27)
The bond order of individual carbon-carbon bonds in benzene is [IIT-JEE 1981; MP PET 2000]
A)
One done
clear
B)
Two done
clear
C)
Between one and two done
clear
D)
One and two, alternately done
clear
View Solution play_arrow
-
question_answer28)
Six carbon atoms of benzene are of
A)
One type done
clear
B)
Two types done
clear
C)
Three types done
clear
D)
Six types done
clear
View Solution play_arrow
-
question_answer29)
On heating a mixture of sodium benzoate and sodalime, the following is obtained [CPMT 1990; AIIMS 1996; MP PET 1999; AFMC 1999]
A)
Toluene done
clear
B)
Phenol done
clear
C)
Benzene done
clear
D)
Benzoic acid done
clear
View Solution play_arrow
-
question_answer30)
Benzene on treatment with a mixture of conc. \[HN{{O}_{3}}\] and conc. \[{{H}_{2}}S{{O}_{4}}\] at \[{{100}^{o}}C\] gives
A)
Nitrobenzene done
clear
B)
m-dinitrobenzene done
clear
C)
p-dinitrobenzene done
clear
D)
o-dinitrobenzene done
clear
View Solution play_arrow
-
question_answer31)
What is the end product which is obtained on the nitration of toluene [MP PMT/PET 1988]
A)
o-nitrotoluene done
clear
B)
p-nitrotoluene done
clear
C)
2, 4-dinitrotoluene done
clear
D)
2, 4, 6-trinitrotoluene done
clear
View Solution play_arrow
-
question_answer32)
Which of the following processes is reversible
A)
Halogenation done
clear
B)
Sulphonation done
clear
C)
Nitration done
clear
D)
None done
clear
View Solution play_arrow
-
question_answer33)
The attacking (electrophilic) species in sulphonation of benzene is [RPMT 1997; CPMT 1999, 2002]
A)
\[S{{O}_{2}}\] done
clear
B)
\[S{{O}_{3}}\] done
clear
C)
\[SO_{4}^{2-}\] done
clear
D)
\[HSO_{3}^{-}\] done
clear
View Solution play_arrow
-
question_answer34)
Which xylene is most easily sulphonated
A)
Ortho done
clear
B)
Para done
clear
C)
Meta done
clear
D)
All at the same rate done
clear
View Solution play_arrow
-
question_answer35)
Toluene on oxidation with dilute \[HN{{O}_{3}}\] and alkaline \[KMn{{O}_{4}}\] gives [DPMT 1981]
A)
Benzaldehyde done
clear
B)
Phenol done
clear
C)
Nitrotoluene done
clear
D)
Benzoic acid done
clear
View Solution play_arrow
-
question_answer36)
Benzene vapour mixed with air when passed over \[{{V}_{2}}{{O}_{5}}\] catalyst at \[775K\] gives [AFMC 1991; CPMT 2001; MP PMT 2003]
A)
Glyoxal done
clear
B)
Oxalic acid done
clear
C)
Maleic anhydride done
clear
D)
Fumaric acid done
clear
View Solution play_arrow
-
question_answer37)
Most common reactions of benzene (aromatic hydrocarbon) and its derivatives are [DPMT 1984; MP PMT 1989; AFMC 1997; BHU 1996, 98]
A)
Electrophilic addition reactions done
clear
B)
Electrophilic substitution reactions done
clear
C)
Nucleophilic addition reactions done
clear
D)
Nucleophilic substitution reactions done
clear
View Solution play_arrow
-
question_answer38)
Which is most readily nitrated [Roorkee 1992]
A)
Benzene done
clear
B)
Phenol done
clear
C)
Aniline done
clear
D)
Nitrobenzene done
clear
View Solution play_arrow
-
question_answer39)
o, p-directing groups are mostly
A)
Activating groups done
clear
B)
Deactivating groups done
clear
C)
Neutral groups done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer40)
Which among the following is the strongest o, p-directing group [CBSE PMT 1989]
A)
OH done
clear
B)
Cl done
clear
C)
\[{{C}_{6}}{{H}_{5}}\] done
clear
D)
Br done
clear
View Solution play_arrow
-
question_answer41)
The compound that is most reactive towards electrophilic nitration is [IIT-JEE 1985; AIIMS 1998; MP PET/PMT 1998]
A)
Toluene done
clear
B)
Benzene done
clear
C)
Benzoic acid done
clear
D)
Nitrobenzene done
clear
View Solution play_arrow
-
question_answer42)
Amongst the following, the compound that can be most readily sulphonated is [IIT-JEE 1982; MADT Bihar 1995; KCET 2005]
A)
Benzene done
clear
B)
Nitrobenzene done
clear
C)
Toluene done
clear
D)
Chlorobenzene done
clear
View Solution play_arrow
-
question_answer43)
Which of the following would be least reactive towards bromine [NCERT 1981]
A)
Nitrobenzene done
clear
B)
Phenol done
clear
C)
Anisole done
clear
D)
Chlorobenzene done
clear
View Solution play_arrow
-
question_answer44)
Amongst the following, the compound that is nitrated with difficulty is
A)
Benzene done
clear
B)
Nitrobenzene done
clear
C)
Toluene done
clear
D)
Phenol done
clear
View Solution play_arrow
-
question_answer45)
Select the true statement about benzene from amongst the following [CBSE PMT 1992]
A)
Because of unsaturation benzene easily undergoes addition reactions done
clear
B)
There are two types of C - C bonds in benzene molecule done
clear
C)
There is a cyclic delocalisation of \[\pi \] electrons in benzene done
clear
D)
Monosubstitution of benzene group gives three isomeric substances done
clear
View Solution play_arrow
-
question_answer46)
Anhydrous \[AlC{{l}_{3}}\] is used in the Friedel-Craft?s reaction because it is [CBSE PMT 1991]
A)
Electron rich done
clear
B)
Soluble in ether done
clear
C)
Insoluble to chloride and aluminium ions done
clear
D)
Electron deficient done
clear
View Solution play_arrow
-
question_answer47)
(i) Chlorobenzene and (ii) benzene hexachloride are obtained from benzene by the reaction of chlorine, in the presence of
A)
(i) Direct sunlight and (ii) anhydrous \[AlC{{l}_{3}}\] done
clear
B)
(i) Sodium hydroxide and (ii) sulphuric acid done
clear
C)
(i) Ultraviolet light and (ii) anhydrous \[FeC{{l}_{3}}\] done
clear
D)
(i) Anhydrous \[AlC{{l}_{3}}\] and (ii) direct sunlight done
clear
View Solution play_arrow
-
question_answer48)
In Friedel Craft?s alkylation, besides \[AlC{{l}_{3}}\] the other reactants are [AFMC 1997; CBSE PMT 1999]
A)
\[{{C}_{6}}{{H}_{6}}+C{{H}_{3}}Cl\] done
clear
B)
\[{{C}_{6}}{{H}_{6}}+C{{H}_{4}}\] done
clear
C)
\[{{C}_{6}}{{H}_{6}}+N{{H}_{3}}\] done
clear
D)
\[{{C}_{6}}{{H}_{6}}+C{{H}_{3}}COCl\] done
clear
View Solution play_arrow
-
question_answer49)
Nitration of benzene is a [RPMT 1999]
A)
Electrophilic displacement done
clear
B)
Electrophilic addition done
clear
C)
Nucleophilic addition done
clear
D)
Nucleophilic displacement done
clear
View Solution play_arrow
-
question_answer50)
Benzene shows [RPMT 1999]
A)
Substitution done
clear
B)
Addition done
clear
C)
Oxidation done
clear
D)
All of these done
clear
View Solution play_arrow
-
question_answer51)
Benzene can be obtained in the reaction [RPET 2000; Bihar MEE 1997]
A)
Ethene + 1, 3-butadiene done
clear
B)
Trimerisation of ethyne done
clear
C)
Reduction of PhCHO done
clear
D)
All of these done
clear
View Solution play_arrow
-
question_answer52)
Thiophene and benzene are separated by [RPET 2000]
A)
Sulphonation of thiophene done
clear
B)
Sulphonation of benzene done
clear
C)
Nitration of thiophene done
clear
D)
Nitration of benzene done
clear
View Solution play_arrow
-
question_answer53)
Which of the following is a hydrocarbon [AFMC 1992]
A)
Urea done
clear
B)
Benzene done
clear
C)
Ammonium cyanate done
clear
D)
Phenol done
clear
View Solution play_arrow
-
question_answer54)
Aromatic compounds burn with sooty flame because [BIT 1991]
A)
They have a ring structure of carbon atoms done
clear
B)
They have a relatively high percentage of hydrogen done
clear
C)
They have a relatively high percentage of carbon done
clear
D)
They resist reaction with oxygen of air done
clear
View Solution play_arrow
-
question_answer55)
Among the following compound which one is planar in shape [MP PMT 2000]
A)
Methane done
clear
B)
Acetylene done
clear
C)
Benzene done
clear
D)
Isobutane done
clear
View Solution play_arrow
-
question_answer56)
Among the following statements on the nitration of aromatic compounds, the false one is [IIT-JEE 1997]
A)
The rate of nitration of benzene is almost the same as that of hexadeuterobenzene done
clear
B)
The rate of nitration of toluene is greater than that of benzene done
clear
C)
The rate of nitration of benzene is greater than that of hexadeuterobenzene done
clear
D)
Nitration is an electrophilic substitution reaction done
clear
View Solution play_arrow
-
question_answer57)
Methyl group attached to benzene can be oxidised to carboxyl group by reacting with [KCET 1993]
A)
\[F{{e}_{2}}{{O}_{3}}\] done
clear
B)
\[AgN{{O}_{3}}\] done
clear
C)
\[KMn{{O}_{4}}\] done
clear
D)
\[C{{r}_{2}}{{O}_{3}}\] done
clear
View Solution play_arrow
-
question_answer58)
How is
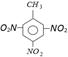
is widely used [MP PET 2002]
A)
Insecticide done
clear
B)
Drug done
clear
C)
Explosive done
clear
D)
Dye done
clear
View Solution play_arrow
-
question_answer59)
The compound ?A? when treated with \[HN{{O}_{3}}\](in presence of \[{{H}_{2}}S{{O}_{4}}\]) gives compound ?B? which is then reduced with Sn and HCl to aniline. The compound ?A? is [MP PET 2002]
A)
Toulene done
clear
B)
Benzene done
clear
C)
Ethane done
clear
D)
Acetamide done
clear
View Solution play_arrow
-
question_answer60)
Which is formed when benzene is heated with chlorine in the presence of sunlight [CPMT 2000; KCET (Med.) 2000; MP PMT 1993; MP PET 2002 AIIMS 1999]
A)
\[{{C}_{6}}{{H}_{5}}CC{{l}_{3}}\] done
clear
B)
\[{{C}_{6}}{{H}_{5}}CHC{{l}_{2}}\] done
clear
C)
\[{{C}_{6}}{{H}_{5}}C{{H}_{2}}C{{l}_{2}}\] done
clear
D)
\[{{C}_{6}}{{H}_{6}}C{{l}_{6}}\] done
clear
View Solution play_arrow
-
question_answer61)
The compound used as an explosive is [Kerala (Engg.) 2002; MP PET 2002; MP PMT 1993]
A)
2,4, 6-tribromoaniline done
clear
B)
1,3, 5-trinitrobenzene done
clear
C)
2,4, 6-trichlorotoluene done
clear
D)
1,3, 5-trichlorobenzene done
clear
E)
2,4, 6-trinitrotoluene done
clear
View Solution play_arrow
-
question_answer62)
Adding of \[C{{l}_{2}}\] to benzene in the presence of \[AlC{{l}_{3}}\] is an example of [Bihar MEE 1996]
A)
Addition done
clear
B)
Halogenation done
clear
C)
Substitution done
clear
D)
Elimination done
clear
E)
None of these done
clear
View Solution play_arrow
-
question_answer63)
What happens when naphthalene balls are put inside kerosene [Kerala (Med.) 2002]
A)
Precipitates done
clear
B)
Dissolves upon heating done
clear
C)
Dissolves easily done
clear
D)
Does not dissolve done
clear
E)
None of these done
clear
View Solution play_arrow
-
question_answer64)
Three fused benzene rings are found in [Kerala (Engg.) 2002]
A)
Naphthalene done
clear
B)
Anthracene done
clear
C)
Phenanthroline done
clear
D)
Triphenyl methane done
clear
View Solution play_arrow
-
question_answer65)
Product obtained after nitration of nitrobenzene is [RPMT 1997]
A)
TNT done
clear
B)
1, 3-dinitrobenzene done
clear
C)
Picric acid done
clear
D)
1, 4-dinitrobenzene done
clear
View Solution play_arrow
-
question_answer66)
After ozonolysis of benzene (not hydrolysis), the product is [RPMT 1997; CPMT 1997]
A)
Benzene triozonide done
clear
B)
Glyoxal done
clear
C)
Ethanediol done
clear
D)
All of them done
clear
View Solution play_arrow
-
question_answer67)
Which acid will not form hydrocarbon [CPMT 1997]
A)
Cinnamic acid done
clear
B)
Isothallic acid done
clear
C)
Salicylic acid done
clear
D)
Picric acid done
clear
View Solution play_arrow
-
question_answer68)
Catalytic dehydrogenation of n-haptane in presence of \[C{{r}_{2}}{{O}_{3}}/A{{l}_{2}}{{O}_{3}}\] at 750 K gives [Roorkee 1999]
A)
iso-heptane done
clear
B)
1-heptene done
clear
C)
toluene done
clear
D)
2, 3-dimethylpentene-1 done
clear
View Solution play_arrow
-
question_answer69)
\[{{C}_{6}}{{H}_{6}}\underset{{{H}_{2}}S{{O}_{4}}}{\mathop{\xrightarrow{HN{{O}_{3}}}}}\,X\underset{FeC{{l}_{3}}}{\mathop{\xrightarrow{C{{l}_{2}}}}}\,Y\]. In the above sequence Y is [AIIMS 1999]
A)
1-nitrochloro benzene done
clear
B)
3-nitrochlorobenzene done
clear
C)
4-nitrochlorobenzene done
clear
D)
1,2-nitrochlorobenzene done
clear
View Solution play_arrow
-
question_answer70)
Which of the following has lowest knocking property
A)
Olefins done
clear
B)
Straight chain paraffins done
clear
C)
Aromatic hydrocarbons done
clear
D)
Branched chain paraffins done
clear
View Solution play_arrow
-
question_answer71)
In which of the following, the bond length between carbon and carbon atom is equal [CPMT 1997]
A)
2-butene done
clear
B)
Benzene done
clear
C)
1-butene done
clear
D)
1-propyne done
clear
View Solution play_arrow
-
question_answer72)
Benzene is prepared in laboratory from which one of the following compounds [MP PMT 1996]
A)
\[{{C}_{6}}{{N}_{5}}{{N}_{2}}Cl\] done
clear
B)
\[{{C}_{6}}{{H}_{5}}OH\] done
clear
C)
\[{{C}_{6}}{{H}_{5}}COONa\] done
clear
D)
\[{{C}_{6}}{{H}_{5}}S{{O}_{3}}H\] done
clear
View Solution play_arrow
-
question_answer73)
Which of the following is not used in Friedel-crafts reaction [KCET 2000]
A)
Phenyl acetanilide done
clear
B)
Bromobenzene done
clear
C)
Benzene done
clear
D)
Chlorobenzene done
clear
View Solution play_arrow
-
question_answer74)
In chlorination of benzene, the reactive species is [MP PET 2000]
A)
\[C{{l}^{+}}\] done
clear
B)
\[C{{l}^{-}}\] done
clear
C)
\[C{{l}_{2}}\] done
clear
D)
\[C{{l}_{2}}^{-}\] done
clear
View Solution play_arrow
-
question_answer75)
Which of following having delocalised electron [BCECE 2005]
A)
Benzene done
clear
B)
Cyclohexane done
clear
C)
CH4 done
clear
D)
C2H6 done
clear
View Solution play_arrow
-
question_answer76)
Benzene molecule is [MP PET 2001; Pb. PMT 2004]
A)
Tetrahedral done
clear
B)
Planar done
clear
C)
Pyramidal done
clear
D)
Trigonal done
clear
View Solution play_arrow
-
question_answer77)
Pyridine is less basic than triethylamine because [AIIMS 2005]
A)
Pyridine has aromatic character done
clear
B)
Nitrogen in pyridine is \[s{{p}^{2}}\] hybridized done
clear
C)
Pyridine is a cyclic system done
clear
D)
In pyridine, lone pair of nitrogen is delocalized done
clear
View Solution play_arrow
-
question_answer78)
Electrophile in the case of chlorination of benzene in the presence of \[FeC{{l}_{3}}\] is [CBSE PMT 1996]
A)
\[C{{l}^{+}}\] done
clear
B)
\[C{{l}^{-}}\] done
clear
C)
Cl done
clear
D)
\[FeC{{l}_{3}}\] done
clear
View Solution play_arrow
-
question_answer79)
Which one of the following will undergo meta substitution on monochlorination [AIIMS 1991]
A)
Ethoxy ethane done
clear
B)
Chlorobenzene done
clear
C)
Ethyl benzoate done
clear
D)
Phenol done
clear
View Solution play_arrow
-
question_answer80)
Nitration of toluene takes place at [NCERT 1990]
A)
o-position done
clear
B)
m-position done
clear
C)
p-position done
clear
D)
Both o- and p-positions done
clear
View Solution play_arrow
-
question_answer81)
Which of the following is not o, p?directing group
A)
\[-N{{H}_{2}}\] done
clear
B)
\[-OH\] done
clear
C)
\[-X\] (halogens) done
clear
D)
\[-CHO\] done
clear
View Solution play_arrow
-
question_answer82)
Benzene can react with [UPSEAT 2003]
A)
\[B{{r}_{2}}\] water done
clear
B)
\[HN{{O}_{3}}\] done
clear
C)
\[{{H}_{2}}O\] done
clear
D)
\[C{{H}_{3}}OH\] done
clear
View Solution play_arrow
-
question_answer83)
The compound ?A? having formula \[{{C}_{8}}{{H}_{10}}\] (aromatic) which gives 1 mononitro substitute and 3 nitrosubstitute compound is [DPMT 2002]
A)
m-Xylene done
clear
B)
p-Xylene done
clear
C)
o-Xylene done
clear
D)
Ethyl benzene done
clear
View Solution play_arrow
-
question_answer84)
Catalytic hydrogenation of benzene gives [AIIMS 1996]
A)
Xylene done
clear
B)
Cyclohexane done
clear
C)
Benzoic acid done
clear
D)
Toluene done
clear
View Solution play_arrow
-
question_answer85)
Benzene is obtained from [CPMT 1996]
A)
Coaltar done
clear
B)
Plant done
clear
C)
Animal done
clear
D)
Charcoal done
clear
View Solution play_arrow
-
question_answer86)
The 'middle oil' fraction of coaltar distillation contains [MP PET 2001]
A)
Benzene done
clear
B)
Anthracene done
clear
C)
Naphthalene done
clear
D)
Xylene done
clear
View Solution play_arrow
-
question_answer87)
Lindane can be obtained by reaction of benzene with [DCE 2000]
A)
\[C{{H}_{3}}Cl\]/anhy.\[AlC{{l}_{3}}\] done
clear
B)
\[C{{l}_{2}}\]/sunlight done
clear
C)
\[{{C}_{2}}{{H}_{5}}I\]/anhy.\[AlC{{l}_{3}}\] done
clear
D)
\[C{{H}_{3}}COCl/AlC{{l}_{3}}\] done
clear
View Solution play_arrow
-
question_answer88)
Which of the following oil is obtained from benzene after fractional distillation of coal tar [BHU 2004]
A)
Light oil done
clear
B)
Heavy oil done
clear
C)
Middle oil done
clear
D)
Anthracene oil done
clear
View Solution play_arrow
-
question_answer89)
Hydrocarbon \[{{C}_{6}}{{H}_{6}}\]decolourise \[B{{r}_{2}}\] water and gives ppt. with ammonical \[AgN{{O}_{3}}\] Hydrocarbon can be [MP PET 2004]
A)
1, 3, 5 Cyclohexatriene done
clear
B)
1, 5 Hexadiyne done
clear
C)
2, 4 Hexadiyne done
clear
D)
None done
clear
View Solution play_arrow
-
question_answer90)
Decreasing order of C-C bond length is [JEE Orissa 2004] \[{{C}_{2}}{{H}_{4}}\] \[{{C}_{2}}{{H}_{2}}\] \[{{C}_{6}}{{H}_{6}}\] \[{{C}_{2}}{{H}_{6}}\]
A)
IV > III > I > II done
clear
B)
I > II > IV > III done
clear
C)
II > I > IV > III done
clear
D)
IV > I > III > II done
clear
View Solution play_arrow
-
question_answer91)
Benzene can be obtained by heating either benzoic acid with X or phenol with Y. X and Y are respectively [KCET 2004]
A)
Zinc dust and soda lime done
clear
B)
Soda lime and zinc dust done
clear
C)
Zinc dust and sodium hydroxide done
clear
D)
Soda lime and copper done
clear
View Solution play_arrow
-
question_answer92)
Order of reactivity of \[{{C}_{2}}{{H}_{6}},{{C}_{2}}{{H}_{4}}\] and \[{{C}_{2}}{{H}_{2}}\] is [MH CET 2004]
A)
\[{{C}_{2}}{{H}_{6}}>{{C}_{2}}{{H}_{4}}>{{C}_{2}}{{H}_{2}}\] done
clear
B)
\[{{C}_{2}}{{H}_{2}}>{{C}_{2}}{{H}_{6}}>{{C}_{2}}{{H}_{4}}\] done
clear
C)
\[{{C}_{2}}{{H}_{2}}>{{C}_{2}}{{H}_{4}}>{{C}_{2}}{{H}_{6}}\] done
clear
D)
All are equally reactive done
clear
View Solution play_arrow
-
question_answer93)
Which of the following yield both alkane and alkene [AFMC 2004]
A)
Kolbe?s reaction done
clear
B)
Williamson?s synthesis done
clear
C)
Wurtz reaction done
clear
D)
Sandmeyer reaction done
clear
View Solution play_arrow
-
question_answer94)
Aromatisation of n-heptane by passing over \[(A{{l}_{2}}{{O}_{3}}+C{{r}_{2}}{{O}_{3}})\] catalyst at 773 K gives [DCE 2004]
A)
Benzene done
clear
B)
Toluene done
clear
C)
Mixture of both done
clear
D)
Heptylene done
clear
View Solution play_arrow
-
question_answer95)
Amongst the following the most basic compound is [AIEEE 2005]
A)
Benzylamine done
clear
B)
Aniline done
clear
C)
Acetanilide done
clear
D)
p-nitroaniline done
clear
View Solution play_arrow
-
question_answer96)
When toluene is treated with \[KMn{{O}_{4}}\], what is produced [AFMC 2005]
A)
Benzene done
clear
B)
Chlorobenzene done
clear
C)
Benzaldehyde done
clear
D)
Benzoic acid done
clear
View Solution play_arrow
-
question_answer97)
In presence of light & heat toluene chlorinated & react with aqueous \[NaOH\] to give [Kerala CET 2005]
A)
o-Cresol done
clear
B)
p-Cresol done
clear
C)
Mixture of o- Cresol & p-Cresol done
clear
D)
Benzoic acid done
clear
E)
1, 3, 5 trihydroxy toluene done
clear
View Solution play_arrow
 is widely used [MP PET 2002]
is widely used [MP PET 2002] 