question_answer 1) Indium impurity in germanium makes it into a:
A)
\[n\]-type of semiconductor
done
clear
B)
p-type semiconductor
done
clear
C)
insulator
done
clear
D)
intrinsic semiconductor
done
clear
View Answer play_arrow
question_answer 2) Kinetic energy of an electron in the first Bohrs orbit of the hydrogen atom is :
A)
\[-6.5eV\]
done
clear
B)
\[+13.6eV\]
done
clear
C)
\[-29.6\text{ }eV\]
done
clear
D)
\[-13.6\text{ }eV\]
done
clear
View Answer play_arrow
question_answer 3) If the refractive angles of the prisms made of crown glass are\[10{}^\circ \]and\[20{}^\circ \]respectively. Then the ratio of their colour deviation power will be:
A)
4 :1
done
clear
B)
3 :1
done
clear
C)
2 :1
done
clear
D)
1:1
done
clear
View Answer play_arrow
question_answer 4) If a current of 10 amp flows in 1 sec through a coil and the induced emf is 10 volt. Then the self-inductance of the coil will be:
A)
\[-1H\]
done
clear
B)
\[-\frac{5}{4}H\]
done
clear
C)
\[-\frac{4}{5}\]
done
clear
D)
\[-\frac{2}{5}H\]
done
clear
View Answer play_arrow
question_answer 5) If a long hollow copper pipe carries a current then the produced magnetic field will be:
A)
both inside and outside the pipe
done
clear
B)
neither inside and nor outside pipe
done
clear
C)
inside the pipe only
done
clear
D)
outside the pipe only
done
clear
View Answer play_arrow
question_answer 6) The bodies moving toward each other collide and move away in opposite direction. This is same as rise in temperature of bodies is due to apart of kinetic energy converted into:
A)
electrical energy
done
clear
B)
nuclear energy
done
clear
C)
mechanical energy
done
clear
D)
heat energy
done
clear
View Answer play_arrow
question_answer 7) A particle executes S.H.M. of amplitude Then the distance from the initial position, if its kinetic energy is equal to its potential energy:
A)
0.71 A
done
clear
B)
1.42 A
done
clear
C)
0.375 A
done
clear
D)
0.91 A
done
clear
View Answer play_arrow
question_answer 8) The two electric bulbs have 40 watt and 60w rating at 220V. Then the ratio of their resistance will be:
A)
1 : 4
done
clear
B)
3 : 2
done
clear
C)
4 : 3
done
clear
D)
2 : 3
done
clear
View Answer play_arrow
question_answer 9) If the earth losses its gravity then for body:
A)
weight becomes zero but not mass
done
clear
B)
mass becomes zero but not the weight
done
clear
C)
both mass and weight become zero
done
clear
D)
neither mass nor weight become zero
done
clear
View Answer play_arrow
question_answer 10) A box is lying on an inclined plane. If the box starts sliding when the angle of inclination is \[45{}^\circ ,\]then its coefficient of friction will be:
A)
1
done
clear
B)
2
done
clear
C)
\[\sqrt{3}\]
done
clear
D)
\[\frac{\sqrt{3}}{2}\]
done
clear
View Answer play_arrow
question_answer 11) In a semiconductor separation between conduction and valence band is of the order of :
A)
0 eV
done
clear
B)
1 eV
done
clear
C)
10 eV
done
clear
D)
50 eV
done
clear
View Answer play_arrow
question_answer 12) If an electron jumps from 1st orbit to IIIrd orbit, then it will:
A)
not gain energy
done
clear
B)
absorb energy
done
clear
C)
release energy
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 13) A person whose nearest distance of distinct vision is 60 cm uses a reading lens of 15 cm focal length. Then the magnification will be:
A)
5
done
clear
B)
3
done
clear
C)
2
done
clear
D)
1
done
clear
View Answer play_arrow
question_answer 14) If the magnetic material which moves from strong to weaker parts of magnetic field. Then it is called :
A)
anti-ferromagnetic
done
clear
B)
ferromagnetic
done
clear
C)
paramagnetic
done
clear
D)
diamagnetic
done
clear
View Answer play_arrow
question_answer 15) A resonance air column of length 17.4 cm resonates with a tuning fork of frequency 512 Hz. The speed of sound in air is :
A)
356 m/s
done
clear
B)
330 m/s
done
clear
C)
343 m/s
done
clear
D)
372 m/s
done
clear
View Answer play_arrow
question_answer 16) When a copper sphere is heated then the percentage increase is maximum in :
A)
diameter
done
clear
B)
volume
done
clear
C)
length
done
clear
D)
area
done
clear
View Answer play_arrow
question_answer 17) If the capacity of a spherical conductor is 1 picofarad then its diameter is :
A)
\[18\times {{10}^{-3}}m\]
done
clear
B)
\[9\times {{10}^{-3}}m\]
done
clear
C)
\[4.5\times {{10}^{-3}}m\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 18) A body is thrown vertically up from the ground. It reaches maximum height of 20 m in 5 sec then it will reach the ground from its maximum height position after:
A)
10 sec
done
clear
B)
2 - 5 sec
done
clear
C)
7.5 sec
done
clear
D)
5 sec
done
clear
View Answer play_arrow
question_answer 19) A man is standing on a spring platform. Reading of spring balance is 60 kg wt. If man jumps outside from the platform then the reading of spring balance will:
A)
become zero
done
clear
B)
increase
done
clear
C)
remains same
done
clear
D)
first [b] then [a]
done
clear
View Answer play_arrow
question_answer 20) If two balls, each of mass 0.06 kg moving in opposite direction with speed 4 m/s, collide and rebound with the same speed then impulse imarted to each ball become of other will be :
A)
0.48 kg m/s
done
clear
B)
0.21 kg m/s
done
clear
C)
9.12 kg m/s
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 21) The intrinsic semiconductor become an insulator at:
A)
0 K
done
clear
B)
300 K
done
clear
C)
\[0{}^\circ C\]
done
clear
D)
\[-100{}^\circ C\]
done
clear
View Answer play_arrow
question_answer 22) The spectrum obtained from sodium vapour lamp is an example of:
A)
absorption spectrum
done
clear
B)
emissive spectrum
done
clear
C)
continuous spectrum
done
clear
D)
bond spectrum
done
clear
View Answer play_arrow
question_answer 23) If 1000 droplets each of potential 1 volt and radius r are mixed to form a big drop. Then the potential of the drop as compared to small droplets, will be:
A)
1000 volt
done
clear
B)
800 volt
done
clear
C)
100 volt
done
clear
D)
20 volt
done
clear
View Answer play_arrow
question_answer 24) If a magnet of length 10 cm and pole strength 40 am is placed at an angle\[45{}^\circ \]in an uniform induction field intensity\[2\times {{10}^{-4}}T\]. The couple acting on it will be:
A)
\[0.5656\times {{10}^{-3}}Nm\]
done
clear
B)
\[0.656\times {{10}^{-4}}N-m\]
done
clear
C)
\[0.5656\times {{10}^{-5}}Nm\]
done
clear
D)
\[0.5656\times {{10}^{-4}}N-m\]
done
clear
View Answer play_arrow
question_answer 25) Kinetic energy of an electron accelerated in a potential difference of 1000 volt, will be:
A)
\[1.6\times {{10}^{-21}}\text{J}\]
done
clear
B)
\[1.6\times {{10}^{-23}}\text{J}\]
done
clear
C)
\[1.6\times {{10}^{-10}}\text{J}\]
done
clear
D)
\[1.6\times {{10}^{-16}}\text{J}\]
done
clear
View Answer play_arrow
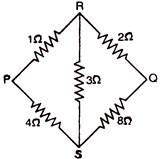
question_answer 26)
The equivalent resistance \[R{{p}_{Q}}\] between point \[p\] and \[Q\] will be:
A)
\[8\,\Omega \]
done
clear
B)
\[2.4\,\Omega \]
done
clear
C)
\[4.5\,\Omega \]
done
clear
D)
\[3\,\Omega \]
done
clear
View Answer play_arrow
question_answer 27) Two waves are said to be coherent, if they have:
A)
same frequency, phase and amplitude
done
clear
B)
different phase and frequency and amplitude
done
clear
C)
same phase and different amplitude
done
clear
D)
same frequency but different amplitude
done
clear
View Answer play_arrow
question_answer 28) In gases of diatomic molecules, the ratio of two specific heats of gases \[\frac{{{C}_{p}}}{{{C}_{\upsilon }}}\] is :
A)
1.21
done
clear
B)
1.33
done
clear
C)
2.80
done
clear
D)
1.40
done
clear
View Answer play_arrow
question_answer 29) Velocity time curve for body projected vertically upward is:
A)
hyperbola
done
clear
B)
ellipse
done
clear
C)
parabola
done
clear
D)
straight line
done
clear
View Answer play_arrow
question_answer 30) A bomb is fired from a cannon with a velocity of of 1000 m/s making an angle of\[30{}^\circ \] with the horizontal. The time taken by the bomb to reach the highest point, will be:
A)
51 sec
done
clear
B)
25.5 sec
done
clear
C)
61 sec
done
clear
D)
21 sec
done
clear
View Answer play_arrow
question_answer 31) A black body at a temperature of 900 K emits energy at a rate, which is proportional to:
A)
\[{{(900)}^{3}}\]
done
clear
B)
\[{{(900)}^{4}}\]
done
clear
C)
(900)
done
clear
D)
\[{{(900)}^{2}}\]
done
clear
View Answer play_arrow
question_answer 32) Number of electron emitted by a surface exposed to light is directly proportional to the:
A)
wavelength of light
done
clear
B)
frequency of light
done
clear
C)
intensity of light
done
clear
D)
velocity of light
done
clear
View Answer play_arrow
question_answer 33) The radium of a circular path of an electron moving in the magnetic field to its path is equal to:
A)
\[\frac{m\upsilon }{Be}\]
done
clear
B)
\[\frac{me}{B}\]
done
clear
C)
\[\frac{mE}{B}\]
done
clear
D)
\[\frac{Be}{m\upsilon }\]
done
clear
View Answer play_arrow
question_answer 34) A moving conductor coil produces an induced emf this is in accordance with :
A)
Ampere law
done
clear
B)
Coulombs law
done
clear
C)
Faradaya law
done
clear
D)
Lenzs Law
done
clear
View Answer play_arrow
question_answer 35) The coefficient of volumetric expansion of mercury is\[18\times {{10}^{-5}}{{/}^{0}}C.\] A thermometer bulb has value of\[{{10}^{-6}}{{m}^{3}}\]and crossectional of stem is \[0.002\text{ }c{{m}^{2}}\]assuming the bulb is tilled mercury at \[{}^\circ C\] the length of mercury at\[100{}^\circ C\] is:
A)
18 cm
done
clear
B)
4.5 cm
done
clear
C)
2.25 cm
done
clear
D)
9 cm
done
clear
View Answer play_arrow
question_answer 36) A hot electric iron has a resistance of 80\[\Omega \] and is used on a 200 volt source. The electrical energy spent, if used for 2 hrs, is:
A)
8000Wh
done
clear
B)
2000Wh
done
clear
C)
1000Wh
done
clear
D)
500Wh
done
clear
View Answer play_arrow
question_answer 37) A source is moving towards an observer with a speed of 20 m/s and having frequency of 240 Hz. The observer now moving towards the source with a speed of 20 m/s. Apparent frequency heard by the observer, is (velocity of sound = 340 m/s):
A)
270 Hz
done
clear
B)
540 Hz
done
clear
C)
135 Hz
done
clear
D)
370 Hz
done
clear
View Answer play_arrow
question_answer 38) A bomb is dropped from an aeroplane moving horizontally at constant speed, when air resistance is taken into consideration. Then the bomb :
A)
falls on the earth the exactly below the aeroplane
done
clear
B)
tails on earth behind aeroplane
done
clear
C)
falls with aeroplane
done
clear
D)
falls on earth ahead the aeroplane
done
clear
View Answer play_arrow
question_answer 39) A car of mass 1500 kg is moving with a speed of 12.5 m/s on a circular path of radius 20 cm on a level road. The coefficient of friction between the car and the road, so that the car does not slip, will be:
A)
0.8
done
clear
B)
0.4
done
clear
C)
0.2
done
clear
D)
0.1
done
clear
View Answer play_arrow
question_answer 40) If force \[\vec{F}=4\hat{i}+5\hat{j}\] and displace\[\vec{s}=3\hat{i}+10\hat{k}\] then the work done will be :
A)
\[4\times 6\]
done
clear
B)
\[6\times 3\]
done
clear
C)
\[5\times 6\]
done
clear
D)
\[4\times 3\]
done
clear
View Answer play_arrow
question_answer 41) Beilstein test is used for the detection of:
A)
N
done
clear
B)
S
done
clear
C)
\[Cl\]
done
clear
D)
Na
done
clear
View Answer play_arrow
question_answer 42) The mass of a compound of molecular formula \[{{C}_{60}}{{H}_{122}}\]is:
A)
\[1.4\times {{10}^{-21}}g\]
done
clear
B)
\[3.7\times {{10}^{-23}}g\]
done
clear
C)
\[4.1\times {{10}^{23}}g\]
done
clear
D)
\[4.7\times {{10}^{23}}g\]
done
clear
View Answer play_arrow
question_answer 43) The best water soluble hydroxide is:
A)
\[Ca{{(OH)}_{2}}\]
done
clear
B)
\[Mg{{(OH)}_{2}}\]
done
clear
C)
\[Sr{{(OH)}_{2}}\]
done
clear
D)
\[Ba{{(OH)}_{2}}\]
done
clear
View Answer play_arrow
question_answer 44) In a period the elements are arranged in sequence of:
A)
increasing charge on the nucleus
done
clear
B)
decreasing charge on the nucleus
done
clear
C)
equal charge on the nucleus
done
clear
D)
constant charge on the nucleus
done
clear
View Answer play_arrow
question_answer 45) The most favourable condition for the manufacture of\[N{{H}_{3}}\]is:
A)
high temperature and high pressure
done
clear
B)
low temperature and low pressure
done
clear
C)
high temperature and low pressure
done
clear
D)
low temperature and high pressure
done
clear
View Answer play_arrow
question_answer 46) The number of sodium atoms present in 2.3 g of sodium are:
A)
\[6.023\times {{10}^{22}}\]
done
clear
B)
\[6.012\times {{10}^{23}}\]
done
clear
C)
\[2.323\times {{10}^{20}}\]
done
clear
D)
\[2.323\times {{10}^{23}}\]
done
clear
View Answer play_arrow
question_answer 47) At\[25{}^\circ C\]the average kinetic energy of an ideal gas per molecule in S.I. unit\[[N=6.02\times {{10}^{23}}]\]is:
A)
\[6.17\times {{10}^{-21}}J\]
done
clear
B)
\[61.7\times {{10}^{-23}}J\]
done
clear
C)
\[6.17\times {{10}^{-20}}J\]
done
clear
D)
\[6.17\times {{10}^{-20}}J\]
done
clear
View Answer play_arrow
question_answer 48) The Bohrs orbit radius for the hydrogen atom \[(n=1)\]is approximately\[0.53\overset{o}{\mathop{\text{A}}}\,\]. The radius for the first excite state\[(n=2)\]orbit is:
A)
\[0.27\overset{o}{\mathop{\text{A}}}\,\]
done
clear
B)
\[1.27\overset{o}{\mathop{\text{A}}}\,\]
done
clear
C)
\[2.12\overset{o}{\mathop{\text{A}}}\,\]
done
clear
D)
\[3.12\overset{o}{\mathop{\text{A}}}\,\]
done
clear
View Answer play_arrow
question_answer 49) Radioactive lead, si^207 has a half life of 8 hours. Starting from 1 mg of\[_{82}P{{b}^{207}}\]how much will remain after 24 hours?
A)
\[\frac{1}{2}mg\]
done
clear
B)
\[\frac{1}{4}mg\]
done
clear
C)
\[\frac{1}{8}mg\]
done
clear
D)
\[\frac{1}{6}mg\]
done
clear
View Answer play_arrow
question_answer 50) The most readily absorpped gas on activated charcoal is:
A)
sulphur dioxide
done
clear
B)
nitrogen
done
clear
C)
hydrogen
done
clear
D)
oxygen
done
clear
View Answer play_arrow
question_answer 51) The reaction of alkyl halide and sodium metal in presence of dry ether is known as:
A)
Wurtzs reaction
done
clear
B)
Clemmensen reaction
done
clear
C)
Cannizzaros reaction
done
clear
D)
Kolbes reaction
done
clear
View Answer play_arrow
question_answer 52) The number of unpaired electrons in \[1{{s}^{2}},2{{s}^{2}}2{{p}^{3}}\]is:
A)
1
done
clear
B)
2
done
clear
C)
3
done
clear
D)
5
done
clear
View Answer play_arrow
question_answer 53) \[_{95}A{{m}^{241}}+X{{\xrightarrow[{}]{{}}}_{97}}B{{k}^{243}}+Y\] The value of X and Y must be:
A)
\[X{{=}_{2}}H{{e}^{4}},Y={{2}_{0}}{{n}^{1}}\]
done
clear
B)
\[X=2e,Y=a\]
done
clear
C)
\[X=a,Y=2e\]
done
clear
D)
\[X={{2}_{0}}{{n}^{1}},Y=2{{a}^{4}}\]
done
clear
View Answer play_arrow
question_answer 54) On heating\[MgC{{l}_{2}}.6{{H}_{2}}O,\]the product obtained is:
A)
\[MgCl\]
done
clear
B)
\[MgO\]
done
clear
C)
\[MgC{{l}_{2}}2{{H}_{2}}O\]
done
clear
D)
\[MgC{{l}_{2}}\]
done
clear
View Answer play_arrow
question_answer 55) The conversion of benzaldehyde into benzyl alcohol takes place by:
A)
Fitting reaction
done
clear
B)
Wurtz fitting reaction
done
clear
C)
Wurtz reaction
done
clear
D)
Cannizzaros reaction
done
clear
View Answer play_arrow
question_answer 56) The compound which does not have carboxyl group is:
A)
benzoic acid
done
clear
B)
picric acid
done
clear
C)
methanoic acid
done
clear
D)
ethanoic acid
done
clear
View Answer play_arrow
question_answer 57) A liquid which decompose at its normal pressure can be purified by:
A)
steam distillation
done
clear
B)
sublimation
done
clear
C)
fractional distillation
done
clear
D)
vacuum distillation
done
clear
View Answer play_arrow
question_answer 58) Which is not effected by temperature?
A)
Molality
done
clear
B)
Molarity
done
clear
C)
Normality
done
clear
D)
Formality
done
clear
View Answer play_arrow
question_answer 59) Nesslers reagent is:
A)
\[H{{g}_{2}}{{I}_{2}}+N{{H}_{4}}OH\]
done
clear
B)
\[{{K}_{2}}Hg{{I}_{4}}+KOH\]
done
clear
C)
\[KHg{{I}_{4}}+N{{H}_{4}}OH\]
done
clear
D)
\[KHg{{I}_{2}}+KOH\]
done
clear
View Answer play_arrow
question_answer 60) If the initial concentration of reactants in certain reaction is doubled, the half life period of the reaction doubles, the order of reaction is:
A)
3rd
done
clear
B)
1st
done
clear
C)
2nd
done
clear
D)
zero
done
clear
View Answer play_arrow
question_answer 61) A gas has a vapour density 11.2. The volume occupies by 1 g of the gas at NTP is:
A)
1L
done
clear
B)
11.2 L
done
clear
C)
22.4 L
done
clear
D)
4 L
done
clear
View Answer play_arrow
question_answer 62) The highest first ionisation potential is of:
A)
carbon
done
clear
B)
boron
done
clear
C)
oxygen
done
clear
D)
nitrogen
done
clear
View Answer play_arrow
question_answer 63) By adding 25 ml of\[3\text{ }M\text{ }HCl\]to 75 ml of \[0.05M\text{ }HCl,\]the molarity of\[HCl\]in the resulting solution is:
A)
0.7875
done
clear
B)
0.9346
done
clear
C)
1.2476
done
clear
D)
1.6684
done
clear
View Answer play_arrow
question_answer 64) If\[n=3,\text{ }l=0\]and\[m=0,\]then atomic number is:
A)
12, 13
done
clear
B)
13, 14
done
clear
C)
10, 11
done
clear
D)
11, 12
done
clear
View Answer play_arrow
question_answer 65) On heating lead nitrate produce:
A)
\[{{N}_{2}}O\]
done
clear
B)
\[NO\]
done
clear
C)
\[N{{O}_{2}}\]
done
clear
D)
\[{{N}_{2}}{{O}_{5}}\]
done
clear
View Answer play_arrow
question_answer 66) lodoform reaction is not given by:
A)
acetayehyde
done
clear
B)
acetone
done
clear
C)
propanpl
done
clear
D)
ethanol
done
clear
View Answer play_arrow
question_answer 67) \[{{K}_{4}}[Fe{{(CN)}_{6}}]\]is used for detecting:
A)
\[Z{{n}^{2+}}\]ion
done
clear
B)
\[C{{u}^{2+}}\]ion
done
clear
C)
\[F{{e}^{2+}}\]ion
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 68) On hydrolysis maltose produce:
A)
galactose
done
clear
B)
mannose
done
clear
C)
glucose
done
clear
D)
fructose
done
clear
View Answer play_arrow
question_answer 69) Paracetamol is an:
A)
antibiotic
done
clear
B)
antipyretic
done
clear
C)
antimalarial
done
clear
D)
analgesic
done
clear
View Answer play_arrow
question_answer 70) Rhombic and monoclinic sulphur are at equilibrium at the transition temperature\[(95.5{}^\circ C)\]. The enthalpy of transition at\[95{}^\circ C\] is\[353.8Jmo{{l}^{-1}}\]. The entropy of transition is:
A)
\[0.46\text{ }J{{K}^{-1}}mo{{l}^{-1}}\]
done
clear
B)
\[0.96\text{ }J{{K}^{-1}}mo{{l}^{-1}}\]
done
clear
C)
\[1.12J{{K}^{-1}}mo{{l}^{-1}}\]
done
clear
D)
\[1.42\text{ }J{{K}^{-1}}mo{{l}^{-1}}\]
done
clear
View Answer play_arrow
question_answer 71) Benzaldehyde gives a positive test with:
A)
Tollens reagent
done
clear
B)
Fehlings solution
done
clear
C)
Benedicts solution
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 72) One mole of an ideal gas at\[25{}^\circ C\]is subjected to expand reversibly ten times of its initial volume. The change in entropy of expansion is:
A)
\[12.13J{{K}^{-1}}mo{{l}^{-1}}\]
done
clear
B)
\[15.17J{{K}^{-1}}mo{{l}^{-1}}\]
done
clear
C)
\[17.15\text{ }J{{K}^{-1}}mo{{l}^{-1}}\]
done
clear
D)
\[19.15J{{K}^{-1}}mo{{l}^{-1}}\]
done
clear
View Answer play_arrow
question_answer 73) Which is not a colligative property?
A)
Osmotic pressure
done
clear
B)
Optical activity
done
clear
C)
Elevation in boiling point
done
clear
D)
Depression in freezing point
done
clear
View Answer play_arrow
question_answer 74) Acetylene contains bonds:
A)
\[4\sigma \]and\[1\pi \]
done
clear
B)
\[3\sigma \]and\[2\pi \]
done
clear
C)
\[5\sigma \]and\[2\pi \]
done
clear
D)
\[5\sigma \]and\[3\pi \]
done
clear
View Answer play_arrow
question_answer 75) The volume of 2.8 gm of carbon monoxide at \[27{}^\circ C\]and 0.821 atm pressure is [R = 0.0821 litre atm \[mo{{l}^{-1}}{{K}^{-1}}\]]:
A)
0.5 litre
done
clear
B)
1.51itrc
done
clear
C)
3 litre
done
clear
D)
6 litre
done
clear
View Answer play_arrow
question_answer 76) \[{{O}_{2}}\] molecule is:
A)
ferromagnetic
done
clear
B)
paramagnetic
done
clear
C)
diamagnetic
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 77) The correct order of electronegativity of halogens is:
A)
\[Cl>Br>I>F\]
done
clear
B)
\[Br>I>Cl>F\]
done
clear
C)
\[I>Br>Cl>F\]
done
clear
D)
\[F>Cl>Br>I\]
done
clear
View Answer play_arrow
question_answer 78) In sulphur detection of an organic compound, sodium nitropruside solution is added to sodium extract. Formation of violet colour is due to:
A)
\[N{{a}_{3}}Fe{{(CN)}_{6}}\]
done
clear
B)
\[N{{a}_{3}}[Fe{{(CN)}_{5}}NOSNa]\]
done
clear
C)
\[N{{a}_{3}}[Fe{{(CN)}_{5}}NS]\]
done
clear
D)
\[Fe{{(CNS)}_{3}}\]
done
clear
View Answer play_arrow
question_answer 79) Epsom salt is:
A)
\[MgS{{O}_{4}}.2{{H}_{2}}O\]
done
clear
B)
\[MgS{{O}_{4}}.7{{H}_{2}}O\]
done
clear
C)
\[BaS{{O}_{4}}.2{{H}_{2}}O\]
done
clear
D)
\[CaS{{O}_{4}}.2{{H}_{2}}O\]
done
clear
View Answer play_arrow
question_answer 80) Cobalt is present in:
A)
vitamin \[{{B}_{1}}\]
done
clear
B)
vitamin \[{{B}_{2}}\]
done
clear
C)
vitamin \[{{B}_{6}}\]
done
clear
D)
vitamin \[{{B}_{12}}\]
done
clear
View Answer play_arrow
question_answer 81) The solubility product of\[AgBr\]solution is \[4\times {{10}^{-13}}\] moles/litre. The Ag1 ion concentration in it is \[1\times {{10}^{-6}}\] moles/litre. The Br- ion concentration is:
A)
\[1\times {{10}^{-6}}\]
done
clear
B)
\[2\times {{10}^{-6}}\]
done
clear
C)
\[2\times {{10}^{-7}}\]
done
clear
D)
\[4\times {{10}^{-7}}\]
done
clear
View Answer play_arrow
question_answer 82) The number of water molecules present in Mohrs salt is:
A)
5
done
clear
B)
6
done
clear
C)
8
done
clear
D)
10
done
clear
View Answer play_arrow
question_answer 83) The rate of diffusion of hydrogen gas is:
A)
two times than He
done
clear
B)
four times than He
done
clear
C)
1.4 times than He
done
clear
D)
similar to He
done
clear
View Answer play_arrow
question_answer 84) The number of essential amino acid in human being is:
A)
10
done
clear
B)
12
done
clear
C)
14
done
clear
D)
16
done
clear
View Answer play_arrow
question_answer 85) The monomers of terelene is:
A)
ethylene glycol and terephthalic acid
done
clear
B)
adipic acid and hexamethylene diamine
done
clear
C)
ethylene glycol and phthalic acid
done
clear
D)
adipic acid and vinyl chloride
done
clear
View Answer play_arrow
question_answer 86) The product formed by exposure of ether to oxygen and sunlight:
A)
alkane
done
clear
B)
alkene
done
clear
C)
oxide
done
clear
D)
peroxide
done
clear
View Answer play_arrow
question_answer 87) The hydrolysis of\[PC{{l}_{3}}\]produce:
A)
\[{{H}_{3}}P{{O}_{3}}\]
done
clear
B)
\[{{H}_{3}}P{{O}_{4}}\]
done
clear
C)
\[HP{{O}_{3}}\]
done
clear
D)
\[POC{{l}_{3}}\]
done
clear
View Answer play_arrow
question_answer 88) Milk is:
A)
gel
done
clear
B)
sol
done
clear
C)
suspension
done
clear
D)
emulsion
done
clear
View Answer play_arrow
question_answer 89) The oxidation number of nitrogen in nitric acid is:
A)
+4
done
clear
B)
+5
done
clear
C)
\[-4\]
done
clear
D)
\[-5\]
done
clear
View Answer play_arrow
question_answer 90) Industrially methanol is produced from :
A)
\[C{{H}_{3}}COOH\]
done
clear
B)
\[C{{H}_{3}}COC{{H}_{3}}\]
done
clear
C)
\[C{{H}_{3}}CHO\]
done
clear
D)
\[CO+{{H}_{2}}\]
done
clear
View Answer play_arrow
question_answer 91) The standard e.m.f. for the cell reaction, \[2C{{u}^{2+}}_{(aq)}\xrightarrow[{}]{{}}C{{u}_{(s)}}+C{{u}^{2+}}(aq)\] is +0.36V at 298 K. The equilibrium constant of the reaction is:
A)
\[5\times {{10}^{6}}\]
done
clear
B)
\[1.4\times {{10}^{12}}\]
done
clear
C)
\[7.4\times {{10}^{12}}\]
done
clear
D)
\[1.2\times {{10}^{6}}\]
done
clear
View Answer play_arrow
question_answer 92) The standard e.m.f. of the cell, \[Cd(s)|CdC{{l}_{2}}(aq)(0.1M)||AgCl(s)|Ag(s)\]in which the cell reaction is: \[Cd(s)+2AgCl(s)\xrightarrow[{}]{{}}2Ag(s)+C{{d}^{2+}}(aq)\] \[+2C{{l}^{-}}(aq)\] is 0.6915 V at 0°C and 0.6753 V at\[25{}^\circ C\]. The enthalpy change of the reaction at\[25{}^\circ C\]is:
A)
\[-176kJ\]
done
clear
B)
\[-234.7kJ\]
done
clear
C)
\[+123.5\text{ }kJ\]
done
clear
D)
\[-167.26\text{ }kJ\]
done
clear
View Answer play_arrow
question_answer 93) Which of the following statement is true?
A)
The relative lowering of vapour pressure of a solution is equal to the mole fraction of the solute present in the solution
done
clear
B)
Passage of solute molecules towards solution side through semipermeable membrane is osmosis.
done
clear
C)
The boiling point of a solution is always lower than the solvent
done
clear
D)
The boiling point of a liquid is the temperature at which its vapour pressure becomes equal to 260 mm
done
clear
View Answer play_arrow
question_answer 94) The deviation from the ideal gas behaviour of a gas can be expressed as:
A)
\[Z=\frac{P}{VRT}\]
done
clear
B)
\[Z=\frac{PV}{nRT}\]
done
clear
C)
\[Z=\frac{nRT}{PV}\]
done
clear
D)
\[Z=\frac{VR}{PT}\]
done
clear
View Answer play_arrow
question_answer 95) Which of the following statement is not true?
A)
The pressure of a gas is due to collision of the gas molecules with the walls of the container
done
clear
B)
The molecular velocity of any gas is proportional to the square root of the absolute temperature
done
clear
C)
The rate of diffusion of a gas is directly proportional to the density of the gas at constant pressure
done
clear
D)
Kinetic energy of an ideal gas is directly proportional to the absolute temperature
done
clear
View Answer play_arrow
question_answer 96) The unit of second order reaction rate constant is:
A)
\[{{L}^{-1}}.\text{ }mol.\text{ }{{s}^{-1}}\]
done
clear
B)
\[{{L}^{2}}.\text{ }mo{{l}^{-2}}.\text{ }{{s}^{-1}}\]
done
clear
C)
\[L.\text{ }mol.\text{ }{{s}^{-1}}\].
done
clear
D)
\[{{s}^{-1}}\]
done
clear
View Answer play_arrow
question_answer 97) Hess law states that:
A)
the standard enthalpy of an overall reaction is the sum of the enthalpy changes in individual reactions
done
clear
B)
enthalpy of formation of a compound is same as the enthalpy of decomposition of the compound into constituent elements, but with opposite sign
done
clear
C)
at constant temperature the pressure of a gas is inversely proportional to its volume
done
clear
D)
the mass of a gas dissolved per litre of a solvent is proportional to the pressure of the gas in equilibrium with the solution
done
clear
View Answer play_arrow
question_answer 98) The half-life of a reaction is halved as the initial concentration of the reactant is doubled. The order of the reaction is
A)
0.5
done
clear
B)
1
done
clear
C)
2
done
clear
D)
0
done
clear
View Answer play_arrow
question_answer 99) One gram of [A] decays by p-emission to 0.125 g in 200 years. The half-life period of the reaction is:
A)
0.014 years
done
clear
B)
6.66 years
done
clear
C)
66.6 years
done
clear
D)
666 years
done
clear
View Answer play_arrow
question_answer 100) Isotopes are:
A)
atoms of different elements having same mass number
done
clear
B)
atoms of same elements having same mass number
done
clear
C)
atoms of same elements having different mass number
done
clear
D)
atoms of different elements having same number of neutrons
done
clear
View Answer play_arrow
question_answer 101) How many tissue systems are suggested by Sachs:
A)
two
done
clear
B)
three
done
clear
C)
four
done
clear
D)
five
done
clear
View Answer play_arrow
question_answer 102) The function of vessel is :
A)
conduction of food
done
clear
B)
conduction of water & minerals
done
clear
C)
to help in transpiration
done
clear
D)
to help in respiration
done
clear
View Answer play_arrow
question_answer 103) When vegetative cells of sporophyte form an embryo it is called :
A)
Apospory
done
clear
B)
Apogamy
done
clear
C)
Apomixis
done
clear
D)
Adventive polyembryony
done
clear
View Answer play_arrow
question_answer 104) Which of the following is used for ripening of fruits ?
A)
Auxin
done
clear
B)
ABA
done
clear
C)
Ethylene gas
done
clear
D)
Cytokinin
done
clear
View Answer play_arrow
question_answer 105) Induction of flowering in plants by low temperature treatment is called :
A)
Vernalization
done
clear
B)
Photoperiodism
done
clear
C)
Cryobiology
done
clear
D)
Chilozology
done
clear
View Answer play_arrow
question_answer 106) The enzyme restriction endonucleases:
A)
cuts single strand of DNA
done
clear
B)
joins the strands of DNA
done
clear
C)
cuts double strand of DNA
done
clear
D)
cuts RNA strand
done
clear
View Answer play_arrow
question_answer 107) Kufri Pukhraj is a hybrid clone of:
A)
Rice
done
clear
B)
Potato
done
clear
C)
Grape
done
clear
D)
Wheat
done
clear
View Answer play_arrow
question_answer 108) Climatology is the science of:
A)
biotic factors
done
clear
B)
climatic factors
done
clear
C)
edaphic factors
done
clear
D)
topographic factors
done
clear
View Answer play_arrow
question_answer 109) Aril is the edible part of:
A)
Apple
done
clear
B)
Grapes
done
clear
C)
Orange
done
clear
D)
Litchi
done
clear
View Answer play_arrow
question_answer 110) Pyruvic acid is formed during :
A)
Krebs cycle
done
clear
B)
Glycolysis
done
clear
C)
Omithine cycle
done
clear
D)
Calvin cycle
done
clear
View Answer play_arrow
question_answer 111) Phagocytosis was observed first time by:
A)
Huxley
done
clear
B)
Strassburger
done
clear
C)
Matchnikoff
done
clear
D)
Haeckel
done
clear
View Answer play_arrow
question_answer 112) Poisonous sea-snakes possess a :
A)
compressed tail
done
clear
B)
printed tail
done
clear
C)
depressed tail
done
clear
D)
conical tail
done
clear
View Answer play_arrow
question_answer 113) When seed germinates, while the fruit is attached to parent plant, it is called:
A)
apomixes
done
clear
B)
vivipary
done
clear
C)
parthenocarpy
done
clear
D)
parthenogenesis
done
clear
View Answer play_arrow
question_answer 114) Blue-green alga are included in:
A)
protista
done
clear
B)
fungi
done
clear
C)
prokaryotes
done
clear
D)
bryophytes
done
clear
View Answer play_arrow
question_answer 115) Echidna is found in:
A)
India
done
clear
B)
Africa
done
clear
C)
Malaya
done
clear
D)
Australia
done
clear
View Answer play_arrow
question_answer 116) Cellulose is a :
A)
Monosaccharide
done
clear
B)
Disaccharide
done
clear
C)
Polysaccharide
done
clear
D)
Lipid
done
clear
View Answer play_arrow
question_answer 117) The stored food material of muscles is :
A)
Starch
done
clear
B)
Glycogen
done
clear
C)
Lipid
done
clear
D)
Protein
done
clear
View Answer play_arrow
question_answer 118) Urease enzyme was obtained from :
A)
Urine
done
clear
B)
Canavalia
done
clear
C)
Ficus fruit
done
clear
D)
Pancreas
done
clear
View Answer play_arrow
question_answer 119) Which of the following is involved in transfer of electrons in photosynthesis ?
A)
phytochrome
done
clear
B)
cytochrome
done
clear
C)
photohormone
done
clear
D)
desmosome
done
clear
View Answer play_arrow
question_answer 120) Plant growth is :
A)
limited
done
clear
B)
unlimited
done
clear
C)
diffused
done
clear
D)
irretic
done
clear
View Answer play_arrow
question_answer 121) Hormone produced during leaf fall is :
A)
IAA
done
clear
B)
ABA
done
clear
C)
Florigen
done
clear
D)
KN
done
clear
View Answer play_arrow
question_answer 122) Climacteric fruit shows :
A)
high respiration rate at ripening
done
clear
B)
sudden change in taste
done
clear
C)
sudden change in colour & shape
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 123) The joint of radio-ulna with upper arm is:
A)
higne joint
done
clear
B)
gliding joint
done
clear
C)
socket joint
done
clear
D)
pivot joint
done
clear
View Answer play_arrow
question_answer 124) Rough endoplasmic reticulum differs from smooth walled endoplasmic reticulum due to the presence of:
A)
DNA
done
clear
B)
ribosome
done
clear
C)
Ergastic substance
done
clear
D)
nucleus
done
clear
View Answer play_arrow
question_answer 125) Apoenzyme is:
A)
protein
done
clear
B)
vitamin
done
clear
C)
carbohydrate
done
clear
D)
amino acid
done
clear
View Answer play_arrow
question_answer 126) The basic unit of classification of plants & animals is:
A)
genus
done
clear
B)
species
done
clear
C)
variety
done
clear
D)
sub species
done
clear
View Answer play_arrow
question_answer 127) Sex organs in Funaria develop :
A)
is axil of leaf
done
clear
B)
at tip of gametophore
done
clear
C)
inside capsule
done
clear
D)
in protonema
done
clear
View Answer play_arrow
question_answer 128) Edible part of cauliflower is :
A)
bud
done
clear
B)
leaves
done
clear
C)
inflorescence
done
clear
D)
fruit
done
clear
View Answer play_arrow
question_answer 129) Which of the following has no stomata ?
A)
Hydrophytes
done
clear
B)
Submerged hydrophytes
done
clear
C)
Xerophytes
done
clear
D)
Mesophytes
done
clear
View Answer play_arrow
question_answer 130) Living fossil is :
A)
Pinus
done
clear
B)
Cycas
done
clear
C)
Ginkgo
done
clear
D)
Ephedra
done
clear
View Answer play_arrow
question_answer 131) The poisonous fluid present in the nematocyst of Hydra is :
A)
venom
done
clear
B)
toxin
done
clear
C)
hypnotoxin
done
clear
D)
haematin
done
clear
View Answer play_arrow
question_answer 132) Which of the following substance is at its lowest level in fish food ?
A)
Actin
done
clear
B)
Myosin
done
clear
C)
Cholesterol
done
clear
D)
Tissue fluid
done
clear
View Answer play_arrow
question_answer 133) Cumulus covers:
A)
ovary
done
clear
B)
ovum
done
clear
C)
zygote
done
clear
D)
embryo
done
clear
View Answer play_arrow
question_answer 134) The disorders like alkaptonuria and phenylkctonuria are referred as:
A)
acquired disease
done
clear
B)
infection disease
done
clear
C)
congential disease
done
clear
D)
all the above
done
clear
View Answer play_arrow
question_answer 135) Which of the following is a communicable disease ?
A)
Hypertension
done
clear
B)
Diabetes
done
clear
C)
Kwasniorkor
done
clear
D)
Diphtheria
done
clear
View Answer play_arrow
question_answer 136) The development of adult characteristics in a moulting insect is promoted by :
A)
thyroxine
done
clear
B)
ecdysone
done
clear
C)
pheromone
done
clear
D)
juvanile hormone
done
clear
View Answer play_arrow
question_answer 137) Who discovered that malaria is transmitted by mosquitoes ?
A)
Ronald Ross
done
clear
B)
Lancisi
done
clear
C)
Grnssi
done
clear
D)
Laveron
done
clear
View Answer play_arrow
question_answer 138) Which of the following is largest gland in adult man ?
A)
Thymus
done
clear
B)
Liver
done
clear
C)
Thyroid
done
clear
D)
Pancreas
done
clear
View Answer play_arrow
question_answer 139) Digestion of glucose takes pake in :
A)
colon
done
clear
B)
caucus
done
clear
C)
small intestine
done
clear
D)
large intestine
done
clear
View Answer play_arrow
question_answer 140) Sensation of stomach pain is due to :
A)
exteroceptors
done
clear
B)
interoceptors
done
clear
C)
teloreceptors
done
clear
D)
proprioceptors
done
clear
View Answer play_arrow
question_answer 141) Example of ophiveroidea is :
A)
Star fish
done
clear
B)
Brittle star
done
clear
C)
Sea urchin
done
clear
D)
Sand dwellers
done
clear
View Answer play_arrow
question_answer 142) Flatworms are:
A)
acoelomates
done
clear
B)
coclomates
done
clear
C)
haemocoelomates
done
clear
D)
pseudocoelomates
done
clear
View Answer play_arrow
question_answer 143) Which of the following class of Protozoa is totally parasitic ?
A)
Ciliata
done
clear
B)
Sporozoa
done
clear
C)
Sarcodina
done
clear
D)
Mastigophora
done
clear
View Answer play_arrow
question_answer 144) Which of the following is colourless Hydra ?
A)
H.fusca
done
clear
B)
H. oligactes
done
clear
C)
H. virides
done
clear
D)
H. vulgaris
done
clear
View Answer play_arrow
question_answer 145) During hibernation period, frogs respiration is:
A)
cutaneous
done
clear
B)
pulmonary
done
clear
C)
pharyngeal
done
clear
D)
buccopharyngeal
done
clear
View Answer play_arrow
question_answer 146) Anti-malarial drug quinine is extracted from bark of:
A)
Mango
done
clear
B)
Cinnamon
done
clear
C)
Cinchona
done
clear
D)
Commune
done
clear
View Answer play_arrow
question_answer 147) The technique of growing plants in water culture is called:
A)
Ikebana
done
clear
B)
Hydroponics
done
clear
C)
Tissue culture
done
clear
D)
Cell-culture
done
clear
View Answer play_arrow
question_answer 148) Which of the following plant shows multiple epidermis:
A)
Palm
done
clear
B)
Ficus
done
clear
C)
Croton
done
clear
D)
Cuciirbita
done
clear
View Answer play_arrow
question_answer 149) The correct sequence of electron acceptor in ATP synthesis:
A)
\[cyt.\text{ }a,{{a}_{3}},\text{ }b,\text{ }c\]
done
clear
B)
\[cyt.\text{ }b,\text{ }c,\text{ }a,\text{ }{{a}_{3}}\]
done
clear
C)
\[cyt.b,{{c}_{3}},\text{ }a,\text{ }{{a}_{3}}\]
done
clear
D)
\[cyt.\text{ }c,\text{ }b,a,{{a}_{3}}\]
done
clear
View Answer play_arrow
question_answer 150) Which of the following is parastic ?
A)
Nostoc
done
clear
B)
Polysiphonia
done
clear
C)
Chara
done
clear
D)
Cephaleuros
done
clear
View Answer play_arrow
question_answer 151) The edible plant part of mushroom is:
A)
basidiocarp
done
clear
B)
primary mycelium
done
clear
C)
secondary mycelium
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 152) What will happen if plant cells are placed in hypertonic solution?
A)
Turgid
done
clear
B)
Plasmolysed
done
clear
C)
Deplasmolysed
done
clear
D)
Lysed
done
clear
View Answer play_arrow
question_answer 153) Loss of water from tips of leaf is called:
A)
bleeding
done
clear
B)
gestation
done
clear
C)
respiration
done
clear
D)
transpiration
done
clear
View Answer play_arrow
question_answer 154) Pond is an example of ecosystem:
A)
artifical
done
clear
B)
natural
done
clear
C)
forest
done
clear
D)
grassland
done
clear
View Answer play_arrow
question_answer 155) Which of the following occurs in fresh as well as marine water ?
A)
Spirogyra
done
clear
B)
Clai lophorn
done
clear
C)
Oedogonium
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 156) Big bang theory was proposed by:
A)
Kant
done
clear
B)
Miller
done
clear
C)
Lemaitre
done
clear
D)
Darwin
done
clear
View Answer play_arrow
question_answer 157) Opium (aphim) is obtained from :
A)
Crotalaria
done
clear
B)
Coffea
done
clear
C)
Ramwifia
done
clear
D)
Papaver sonmiferum
done
clear
View Answer play_arrow
question_answer 158) Which is man made cereal?
A)
Triticum
done
clear
B)
Hordeum
done
clear
C)
Triticale
done
clear
D)
Elusin
done
clear
View Answer play_arrow
question_answer 159) Branched conidiophores are present in :
A)
Rhizopus
done
clear
B)
Penicillium
done
clear
C)
Ustilago
done
clear
D)
Aspegrillus
done
clear
View Answer play_arrow
question_answer 160) Crown gall is caused by :
A)
Mycoplasma
done
clear
B)
Candida
done
clear
C)
Agrobacterium
done
clear
D)
Yeast
done
clear
View Answer play_arrow
question_answer 161) The membrane surrounding cell vacuole is called:
A)
tonoplast
done
clear
B)
cell wall
done
clear
C)
plasma membrane
done
clear
D)
cell membrane
done
clear
View Answer play_arrow
question_answer 162) National Botanical Research Institute (NBRI) is situated at:
A)
Lucknow
done
clear
B)
Bombay
done
clear
C)
Calcutta
done
clear
D)
Delhi
done
clear
View Answer play_arrow
question_answer 163) Nissles granules are found in :
A)
liver cells
done
clear
B)
nerve cells
done
clear
C)
kidney
done
clear
D)
heart
done
clear
View Answer play_arrow
question_answer 164) Hutchinson system of classification is :
A)
Natural
done
clear
B)
Artifical
done
clear
C)
Phylogenetic
done
clear
D)
Binomial
done
clear
View Answer play_arrow
question_answer 165) Thennal pollution is caused by :
A)
power plants
done
clear
B)
industries
done
clear
C)
automobiles
done
clear
D)
all the above
done
clear
View Answer play_arrow
question_answer 166) Pure line breed refers to :
A)
homozygosity, independent assortmant only
done
clear
B)
hetirozygosity and linkage
done
clear
C)
heterozygosity only
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 167) Two free ribonucleotide units are interlinked with:
A)
peptide bond
done
clear
B)
covalent bond
done
clear
C)
hydrogen bond
done
clear
D)
phosphodiester bond
done
clear
View Answer play_arrow
question_answer 168) Which of the following is related to increase in girth of plant ?
A)
Shoot apex
done
clear
B)
Root apex
done
clear
C)
Lateral meristem
done
clear
D)
Intercalary meristem of stamen
done
clear
View Answer play_arrow
question_answer 169) Monoadelphous character is found in :
A)
Brassicaceae
done
clear
B)
Malvaceae
done
clear
C)
Poaceae
done
clear
D)
Solanaceae
done
clear
View Answer play_arrow
question_answer 170) If an organism is crossed with homozygous recessive individual, it is called :
A)
back cross
done
clear
B)
test cross
done
clear
C)
simple cross
done
clear
D)
complex cross
done
clear
View Answer play_arrow
question_answer 171) Humus is:
A)
dead and decayed organic matter
done
clear
B)
living matter
done
clear
C)
fertilizers
done
clear
D)
living animal/plants/microbes
done
clear
View Answer play_arrow
question_answer 172) Bryophytes lack true :
A)
roots
done
clear
B)
stem
done
clear
C)
leaves
done
clear
D)
all the above
done
clear
View Answer play_arrow
question_answer 173) Scurvy is caused due to deficiency of vitamin:
A)
B complex
done
clear
B)
C
done
clear
C)
K
done
clear
D)
D
done
clear
View Answer play_arrow
question_answer 174) Which of the following has most reduced form of stem ?
A)
Rhizome
done
clear
B)
Stolons
done
clear
C)
Runner
done
clear
D)
Bulb
done
clear
View Answer play_arrow
question_answer 175) Binomial nomenclature indicates :
A)
genus and species
done
clear
B)
genus and family
done
clear
C)
species and variety
done
clear
D)
phylum and genus
done
clear
View Answer play_arrow
question_answer 176) Which is haploid in case of Cycas ?
A)
Stem
done
clear
B)
Root
done
clear
C)
Endosperm
done
clear
D)
All the above
done
clear
View Answer play_arrow
question_answer 177) Tetradynamous stamens are- found in :
A)
Brassica
done
clear
B)
Pisum
done
clear
C)
Hibiscus
done
clear
D)
Cucurbita
done
clear
View Answer play_arrow
question_answer 178) Pollinia are present in family :
A)
Brassicaceae
done
clear
B)
Cucurbitaceae
done
clear
C)
Asclepiadaceae
done
clear
D)
Malvaceae
done
clear
View Answer play_arrow
question_answer 179) Nuclear endosperm is present in :
A)
Banana
done
clear
B)
Coconut
done
clear
C)
Phoenix
done
clear
D)
Mango
done
clear
View Answer play_arrow
question_answer 180) Genetic code is :
A)
universal
done
clear
B)
non-ambiguous
done
clear
C)
triplet
done
clear
D)
all the above
done
clear
View Answer play_arrow
question_answer 181) Cytoplasmic inheritance is due to :
A)
mitochondria
done
clear
B)
Kappa particles
done
clear
C)
both a and b
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 182) Hurlers disease is caused by the absence of:
A)
mitochondria
done
clear
B)
lysosomes
done
clear
C)
lomosomes
done
clear
D)
mesosomes
done
clear
View Answer play_arrow
question_answer 183) Which of the following is bounded by double membrane:
A)
Chloroplast
done
clear
B)
Mitochondria
done
clear
C)
Nucleus
done
clear
D)
All the above
done
clear
View Answer play_arrow
question_answer 184) The process of formation of glycogen from glucose is called ?
A)
Glycogenesis
done
clear
B)
Lipogenesis
done
clear
C)
Gluconeogensis
done
clear
D)
Glycogenolysis
done
clear
View Answer play_arrow
question_answer 185) The amount of energy given by one molecule of ATP is :
A)
7.6 kcal
done
clear
B)
7.3 kcal
done
clear
C)
6.86 kcal
done
clear
D)
7.06 kcal
done
clear
View Answer play_arrow
question_answer 186) Which of the following is enucleate at maturity ?
A)
Cortical cell
done
clear
B)
Cambial cell
done
clear
C)
Meristematic cell
done
clear
D)
Sieve tube cell
done
clear
View Answer play_arrow
question_answer 187) The cambuim which produces cork is called as:
A)
Phelloderm
done
clear
B)
Phellogen
done
clear
C)
Periblem
done
clear
D)
Plerome
done
clear
View Answer play_arrow
question_answer 188) Casparian strips are characteristic feature of endodermis and made up of :
A)
cutin
done
clear
B)
pectin
done
clear
C)
suberin
done
clear
D)
cellulose
done
clear
View Answer play_arrow
question_answer 189) Root pressure is measured by :
A)
Manometer
done
clear
B)
Potometer
done
clear
C)
Auxanometer
done
clear
D)
Osmometer
done
clear
View Answer play_arrow
question_answer 190) Substrate for photorespiration is :
A)
Glycolate
done
clear
B)
Glucose
done
clear
C)
Lipid
done
clear
D)
Sucrose
done
clear
View Answer play_arrow
question_answer 191) In aquaria, green plants are grown for:
A)
\[C{{O}_{2}}\]
done
clear
B)
\[{{O}_{2}}\]
done
clear
C)
beauty
done
clear
D)
all the above
done
clear
View Answer play_arrow
question_answer 192) Which of the following is releated with transfer of food material ?
A)
Xylem
done
clear
B)
Collenchyma
done
clear
C)
Phloem
done
clear
D)
Parenchyma
done
clear
View Answer play_arrow
question_answer 193) Vitamin containing cobalt is:
A)
\[A\]
done
clear
B)
\[{{B}_{6}}\]
done
clear
C)
\[{{B}_{1}}\]
done
clear
D)
\[{{B}_{12}}\]
done
clear
View Answer play_arrow
question_answer 194) Which cells do not divide after birth?
A)
Nerve cells
done
clear
B)
Germ cells
done
clear
C)
Muscle cells
done
clear
D)
Osteocytes
done
clear
View Answer play_arrow
question_answer 195) Main function of cerebellum is :
A)
Balancing
done
clear
B)
Sight
done
clear
C)
Hearing
done
clear
D)
Memory
done
clear
View Answer play_arrow
question_answer 196) Meiosis occurs in:
A)
root cells
done
clear
B)
stem cells
done
clear
C)
leaf cells
done
clear
D)
pollen mother cells
done
clear
View Answer play_arrow
question_answer 197) Genetic material of TMV is:
A)
RNA
done
clear
B)
DNA
done
clear
C)
DNAandRNA
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 198) Bacteria with flagella all over body is called :
A)
peritrichous
done
clear
B)
monotrichous
done
clear
C)
lophotrichous
done
clear
D)
amphitrichous
done
clear
View Answer play_arrow
question_answer 199) The largest cell organelle is :
A)
mitochondria
done
clear
B)
chloroplast
done
clear
C)
golgibody
done
clear
D)
chromosome
done
clear
View Answer play_arrow
question_answer 200) Which of the following is unicellular?
A)
Yeast
done
clear
B)
E. coli
done
clear
C)
Acetabularia
done
clear
D)
All the above
done
clear
View Answer play_arrow