A) \[ds{{p}^{2}}\]
B) \[s{{p}^{2}}\]
C) \[s{{p}^{3}}\]
D) \[s{{p}^{3}}{{d}^{2}}\]
Correct Answer: A
Solution :
\[{{[Ni{{(CN)}_{4}}]}^{2-}}\] In this complex, oxidation state of \[Ni\]is \[+2~\] \[[x-4=-2\,\,or\,\,x=+2]\] \[N{{i}^{r}}=[Ar]3{{d}^{8}}\]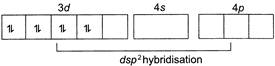
You need to login to perform this action.
You will be redirected in
3 sec
