A) \[C{{H}_{3}}COONa\]
B) \[N{{H}_{4}}Cl\]
C) \[NaCl\]
D) \[C{{H}_{3}}COON{{H}_{4}}\]
Correct Answer: B
Solution :
\[N{{H}_{4}}Cl\]gives an acidic solution in water\[N{{H}_{4}}ClN{{H}_{4}}+C{{l}^{-}}\]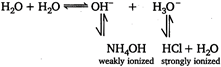
You need to login to perform this action.
You will be redirected in
3 sec
