A) \[zero\]
B) \[0.88\]
C) \[1.33\]
D) \[2\]
Correct Answer: C
Solution :
We know that carbonate ion has following resonating structures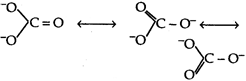 Bond order \[=\frac{total\,\,number\,\,of\,\,bons\,\,between\,\,atoms}{total\,\,number\,\,of\,\,resonating\,\,structures}\]\[=1.33\]
Bond order \[=\frac{total\,\,number\,\,of\,\,bons\,\,between\,\,atoms}{total\,\,number\,\,of\,\,resonating\,\,structures}\]\[=1.33\]
You need to login to perform this action.
You will be redirected in
3 sec
