A) 1
B) 2
C) 3
D) 4
Correct Answer: A
Solution :
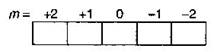
| n = 3 |
| l = 2 |
| m = + 2 |
| \[s=\,\,\pm \,1/2\] |
| These values of quantum numbers are possible for an orbital of 3d sub-shell as the possible value of m for l = 2 is |
 |
You need to login to perform this action.
You will be redirected in
3 sec
