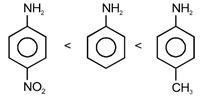
| The correct increasing order of basic strength for the following compounds is [NEET-2017] |
| (i) |
| (ii) |
| (iii) |
A) II < I < III
B) II < III < I
C) III < I < II
D) III < II < I
Correct Answer: A
Solution :
| [a] \[-N{{O}_{2}}\]has strong -R effect and \[-C{{H}_{3}}\] shows +R effect. |
| \[\therefore \]Order of basic strength is |
 |
You need to login to perform this action.
You will be redirected in
3 sec
