A) \[trans{{[Co{{(en)}_{2}}C{{l}_{2}}]}^{+}}\]
B) \[cis{{[Co(en){{(N{{H}_{3}})}_{6}}]}^{2+}}\]
C) \[{{[Co{{(N{{H}_{3}})}_{6}}]}^{3+}}\]
D) \[{{[Fe{{(CN)}_{6}}]}^{3-}}\]
Correct Answer: B
Solution :
Usually optical isomerism is exhibited by \[[M{{(aa)}_{3}}]\]or\[[M{{(aa)}_{2}}{{X}_{2}}]\]or\[[M(aa){{X}_{2}}1/2]\]type complexes. (Here M = metal atom, aa = bidentate ligand, X and Y = unidentate ligand). Hence,\[cis{{[(en){{(N{{H}_{3}})}_{2}}C{{l}_{2}}]}^{+}}\]exhibits optical isomerism. Its optically active forms are as: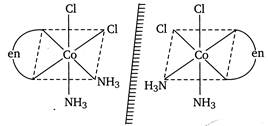 Only cis form of te+rahedral complexes of the type\[{{[M{{(aa)}_{2}}{{x}_{2}}]}^{+}}\]is optically active.
Only cis form of te+rahedral complexes of the type\[{{[M{{(aa)}_{2}}{{x}_{2}}]}^{+}}\]is optically active.
You need to login to perform this action.
You will be redirected in
3 sec
