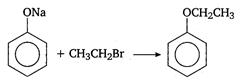
 the type of reaction is
the type of reaction is
A) electrophilic substitution
B) nucleophilic substitution
C) free radical substitution
D) None of the above
Correct Answer: B
Solution :
Electrophilic substitution reaction Electrophilic substitution involves the attack by an electrophile. eg, \[C{{H}_{3}}C{{H}_{2}}MgBr\xrightarrow[-MgBr]{}\overset{\oplus }{\mathop{C}}\,{{H}_{3}}C{{H}_{2}}\]\[\xrightarrow{{{H}^{+}}}C{{H}_{3}}C{{H}_{3}}\] Nucleophilic substitution reaction Many substitution reactions, especially at the saturated carbon atom in aliphatic compounds such as alkyl halides, are brought about by nucleophilic reagents or nucleophiles. \[R-X+\underset{nucleophile}{\mathop{O{{H}^{-}}}}\,\xrightarrow{{}}R-OH+\underset{\begin{smallmatrix} leaving \\ group \end{smallmatrix}}{\mathop{{{X}^{\Theta }}}}\,\] Such substitution reactions are called nucleophilic substitution reactions. eg,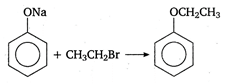 Free radical substitution Free radical substitution reactions involve the attack by a free radical. eg, \[C{{H}_{4}}+C{{l}_{2}}\,\,\xrightarrow[light]{UV}C{{H}_{3}}Cl+HCl\]
Free radical substitution Free radical substitution reactions involve the attack by a free radical. eg, \[C{{H}_{4}}+C{{l}_{2}}\,\,\xrightarrow[light]{UV}C{{H}_{3}}Cl+HCl\]
You need to login to perform this action.
You will be redirected in
3 sec
