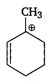
A)
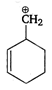
B)
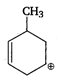
C)
D) All are equally
Correct Answer: A
Solution :
The stability of carbocation increases with increasing the number of alkyl groups attached to positively charged carbon atom or with increasing the number of hyperconjugative structures. Thus, \[{{1}^{o}}\] carbocation \[<{{2}^{o}}\] carbocation \[<{{3}^{o}}\] carbocation. Allyl and benzyl carbocation are stabilised by resonance. Hence,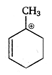 is most stable because it is \[{{3}^{o}}\] allylic carbocation.
is most stable because it is \[{{3}^{o}}\] allylic carbocation.
You need to login to perform this action.
You will be redirected in
3 sec
