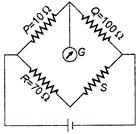
A) \[0.7\,\,\Omega \]
B) \[70\,\,\Omega \]
C) \[7\,\,\Omega \]
D) \[700\,\,\Omega \]
Correct Answer: D
Solution :
From Einsteins photoelectric equation \[{{E}_{K}}=hv-W\] where W is work function of metal, hv is the energy of the photon absorbed by the electron in the metal and \[{{E}_{K}}\]is maximum kinetic energy. Given, \[W=2\,eV=2\times 1.6\times {{10}^{-19}}V\] \[=3.2\times {{10}^{-19}}J\] \[\therefore \] \[{{E}_{K}}=\frac{6.6\times {{10}^{-34}}\times 3\times {{10}^{8}}}{5000\times {{10}^{-10}}}-3.2\times {{10}^{-19}}\] \[{{E}_{K}}=7.6\times {{10}^{-20}}J=\frac{7.6\times {{10}^{-20}}}{1.6\times {{10}^{-19}}}=0.47\,eV\] Note: Maximum kinetic energy of photo electrons does not depend upon the intensity of incident light.You need to login to perform this action.
You will be redirected in
3 sec
