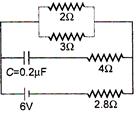
A) zero
B) 0.6 A
C) 0.9 A
D) 1.5A
Correct Answer: C
Solution :
From Bohrs atomic relation \[{{r}_{n}}=\frac{{{\varepsilon }_{0}}\,{{n}^{2}}{{h}^{2}}}{\pi \,m\,Z{{e}^{2}}}\] or \[{{r}_{n}}\propto {{n}^{2}}\]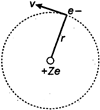 where h is Plancks constant, Z is atomic number and m is mass. Given, \[{{r}_{1}}=0.53\,\,\overset{o}{\mathop{A}}\,,\,\,{{n}_{1}}=1,\,{{n}_{2}}=4\] \[\therefore \] \[\frac{{{r}_{1}}}{{{r}_{2}}}=\frac{{{n}^{2}}}{{{n}_{2}}^{2}}\] \[\frac{0.53}{{{r}_{2}}}=\frac{1}{16}\] \[\Rightarrow \] \[{{r}_{2}}=0.53\times 16=8.48\,\overset{o}{\mathop{A}}\,\]
where h is Plancks constant, Z is atomic number and m is mass. Given, \[{{r}_{1}}=0.53\,\,\overset{o}{\mathop{A}}\,,\,\,{{n}_{1}}=1,\,{{n}_{2}}=4\] \[\therefore \] \[\frac{{{r}_{1}}}{{{r}_{2}}}=\frac{{{n}^{2}}}{{{n}_{2}}^{2}}\] \[\frac{0.53}{{{r}_{2}}}=\frac{1}{16}\] \[\Rightarrow \] \[{{r}_{2}}=0.53\times 16=8.48\,\overset{o}{\mathop{A}}\,\]
You need to login to perform this action.
You will be redirected in
3 sec
