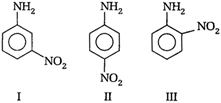
A) I > II > III
B) I > III > II
C) III > II > I
D) III > I > II
E) III \[\simeq \]II > I
Correct Answer: A
Solution :
I > II > III is the correct order of basicity. ? \[N{{O}_{2}}\] is an electron withdrawing group, thus nitro-anilines are less basic than aniline. ? (III) is less basic than (II) because \[-N{{O}_{2}}\] is closer and exerts a stronger electron withdrawing or \[-I\]-effect.You need to login to perform this action.
You will be redirected in
3 sec
