A) A > B > C
B) A > C > B
C) C > A > B
D) C > B > A
Correct Answer: C
Solution :
\[C>A>B\]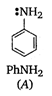
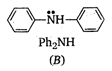
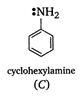 In diphenylamine, there are two electron withdrawing phenyl groups with -I effect while there is one such group in aniline. Therefore, diphenylamine is a weaker base than aniline which, in turn, is less basic than cyclohexyl amine in which the electron pair on nitrogen can be easily donated to a Lewis acid.
In diphenylamine, there are two electron withdrawing phenyl groups with -I effect while there is one such group in aniline. Therefore, diphenylamine is a weaker base than aniline which, in turn, is less basic than cyclohexyl amine in which the electron pair on nitrogen can be easily donated to a Lewis acid.
You need to login to perform this action.
You will be redirected in
3 sec
