| Two solutions A and B have pH values of 3.0 and 10.5 respectively. Which of these will turn: |
| (i) Blue litmus solution to red and |
| (ii) Phenolphthalein from colourless to pink? |
| Justify your answer in each case. |
| Write one point of difference between each of the following: |
| (i) A hydrated salt and an anhydrous salt |
| (ii) Washing soda and soda ash |
| (ii) Baking soda and Baking powder |
| (a) Name any one metal each which can be extracted by: |
| (i) reduction with carbon |
| (ii) electrolytic reduction |
| (iii) reduction with aluminium |
| (iv) reduction with heat alone |
| (b) Write a chemical equation for any of the above four parts. |
| State which of the following chemical reactions will take place or not, giving suitable reason for each: |
| (a) \[Z{{n}_{(S)}}+CuS{{O}_{4(aq)}}\to ZnS{{O}_{4(aq)}}+\text{ }C{{u}_{(S)}}\] |
| (b) \[F{{e}_{(S)}}+ZnS{{O}_{4(aq)}}\to FeS{{O}_{4(aq)}}+Z{{n}_{(S)}}\] |
| (c) \[Z{{n}_{(S)}}+FeS{{O}_{4(aq)}}\to ZnS{{O}_{4(aq)}}+F{{e}_{(S)}}\] |
| Write one function of each of the following components of the transport system in human beings: |
| (a) Blood vessels |
| (b) Lymph |
| (c) Heart |
| Give reason for the following: |
| (i) Why are copper and aluminium wires used as connecting wires? |
| (ii) Why is tungsten used for filaments of electric lamps? |
| (iii) Why is lead-tin alloy used for fuse wires? |
| You have been appointed as the 'eco club in charge' of your school. You have to take care of the maintenance and conservation of the environment. |
| (i) Suggest any three ways by which you will carry on your duties. |
| (ii) Write any three qualities that you would like your school mates to develop for environment conservation. |
| (a) Define corrosion. What name is given to the corrosion of iron? |
| (b) Name the colour of coating formed on silver and copper articles, when exposed to air? |
| (c) List two damages caused by corrosion and suggest how corrosion can be prevented. |
| (a) Define indicator. Name two indicators obtained from plants. |
| (b) Write balanced chemical equation for the reaction that takes place when sodium oxide reacts with water. How will this solution behave towards phenolphthalein and red litmus paper? |
| (c) State what happen when sodium hydroxide, solution reacts with dilute hydrochloric acid. What is this reaction called? |
| (a) State the form in which the following an stored: |
| (i) Unused carbohydrates in plants. |
| (ii) The energy derived from food in humans. |
| (b) Describe the process of nutrition in amoeba with the help of diagram. |
| (a) Write two points of difference between electric energy and electric power. |
| (b) Out of 60 W and 40 W lamps, which one has a higher electrical resistance when in use. |
| (c) What is commercial unit of electric energy? Convert it into Joules. |
| (a) How is the strength of the magnetic field at a point near a straight conductor related to the strength of the electric current flowing in the conductor? |
| (b) With the help of a diagram describe an activity to show that a straight conductor carrying current produces a magnetic field around it. State the rule which may be used to determine the direction of magnetic field thus produced. |
| (c) Why do two magnetic field lines never intersect each other? Explain. |
| (a) What is meant by heating effect of electric current? Give two applications of heating effect of current. |
| (b) State the law relating heat dissipated in an electric circuit with the current potential difference and time. |
| (c) 50 J of heat is produced each second in a \[\mathbf{2}\,\mathbf{\Omega }\] resistor. Find this potential difference across the resistor. |
| The two colours seen at the extreme ends of the pH chart are: |
| (a) Red and blue |
| (b) Red and green |
| (c) Green and blue |
| (d) Orange and green |
| Four students A, B, C and D determine the pH of water, lemon juice and dil. sodium bicarbonate solution. They recorded their observations and arranged them in descending order of pH values as follows: | |
| Student | Solutions |
| (A) | Water, lemon juice, sodium bicarbonate solution |
| (B) | Water, sodium bicarbonate solution, lemon juice |
| (C) | Lemon juice, water, sodium bicarbonate solution |
| (D) | Sodium bicarbonate solution, water, lemon juice |
| The correct sequence is of the student: | |
| (a) (A) (b) (B) | |
| (c) (C) (d) (D) | |
| Zinc metal is added to dilute hydrochloric acid. The gas evolved is: |
| (a) Oxygen |
| (b) Hydrogen |
| (c) Chlorine |
| (d) Carbon dioxide |
| Four metal rods labelled as P, Q, R and S along with their corresponding colours are shown below. Which of these rod could be made up of aluminium? |
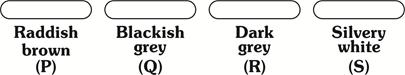 |
| (a) P (b) Q |
| (c) R (d) S |
| A solution of \[\mathbf{FeS}{{\mathbf{O}}_{\mathbf{4}}}\] in water is: |
| (a) colourless (b) blue |
| (c) light green (d) brown |
| To find the equivalent resistance of two resistors connected in series, the connection of ammeter is correct in the circuit: |
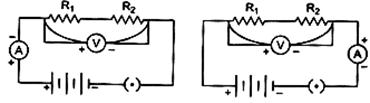 |
| (a) Circuit A (b) Circuit B |
| (c) Both the circuits (d) Neither of the two circuits |
| A student found that when a resistance of \[\mathbf{3}\,\mathbf{\Omega }\] was joined with 3 V battery as per figure shown below, the current flowing through it was 1 A. He then joined another resistance of 6\[\mathbf{6}\,\mathbf{\Omega }\] in parallel with \[\mathbf{3}\,\mathbf{\Omega }\] resistance. The reading in the ammeter will now be: |
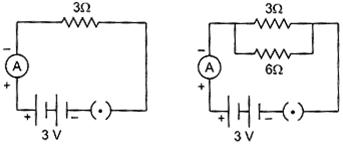 |
| (a) 9 A (b) 1.5 A |
| (c) 1 A (d) 6 A |
| A student covered a leaf from a de-starched plant with a black paper strip and kept it in the garden outside his house in fresh air. In the evening, he tested the covered portion of the leaf for the presence of starch. By doing so the student was trying to show that. |
| (a) \[C{{O}_{2}}\] is given out during respiration |
| (b) \[C{{O}_{2}}\] is necessary for photosynthesis |
| (c) Chlorophyll is necessary for photosynthesis |
| (d) Light is necessary for photosynthesis |
| In the experiment to show that ?\[\mathbf{C}{{\mathbf{O}}_{\mathbf{2}}}\] is released during respiration?, the small test tube of KOH solution is suspended inside the conical flask to absorb the: |
| (a) air of the flask |
| (b) moisture of the flask |
| (c) \[{{O}_{2}}\] of the flask |
| (d) \[C{{O}_{2}}\] of the flask released by the seeds |
| While demonstrating decomposition reaction in laboratory the teacher heated ferrous sulphate crystals in a hard glass dry boiling tube. |
| (i) What change in the colour of ferrous sulphate crystals you will observe? |
| (ii) What type of smell of the gases coming out of the boiling tube would you feel? |
You need to login to perform this action.
You will be redirected in
3 sec
