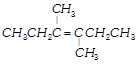The density of a cube is measured by measuring its mass and length of its sides. If the maximum error in the measurement of mass and length are 4% and 3% respectively, the maximum error in the measurement of density will be
A)
7%
done
clear
B)
9%
done
clear
C)
12%
done
clear
D)
13%
done
clear
View Answer play_arrow
The activity of a radioactive sample is measured as 9750 counts per minute at t = 0 and as 975 counts per minute at t = 5 minutes. The decay constant is approximately
A)
0.922 per minute
done
clear
B)
0.691 per minute
done
clear
C)
0.461 per minute
done
clear
D)
0.230 per minute
done
clear
View Answer play_arrow
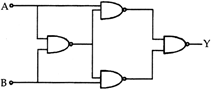
Truth table for system of four NAND gates as shown in figure is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
A closed organ pipe and an open organ pipe are tuned to the same fundamental frequency. What is the ratio of their lengths?
A)
1 : 2
done
clear
B)
2 : 1
done
clear
C)
2 : 3
done
clear
D)
4 : 3
done
clear
View Answer play_arrow
The radius R of the soap bubble is doubled under isothermal condition. If T be the surface tension of soap bubble, the work done in doing so is given by
A)
\[32\pi {{R}^{2}}T\]
done
clear
B)
\[24\pi {{R}^{2}}T\]
done
clear
C)
\[8\pi {{R}^{2}}T\]
done
clear
D)
\[4\pi {{R}^{2}}T\]
done
clear
View Answer play_arrow
The masses of the three wires of copper are in the ratio of 1: 3: 5 and their lengths are in the ratio of 5: 3: 1. The ratio of their electrical resistance is
A)
1: 3: 5
done
clear
B)
5: 3: 1
done
clear
C)
1: 25: 125
done
clear
D)
125: 15: 1
done
clear
View Answer play_arrow
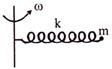
A particle of mass m is fixed to one end of a light spring of force constant k and unstretched length \[\ell \] The system is rotated about the other end of the spring with an angular velocity \[\omega ,\] in gravity free space. The increase in length of the spring will be
A)
\[\frac{m{{\omega }^{2}}\ell }{k}\]
done
clear
B)
\[\frac{m{{\omega }^{2}}\ell }{k-m{{\omega }^{2}}}\]
done
clear
C)
\[\frac{m{{\omega }^{2}}\ell }{k+m{{\omega }^{2}}}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
Two equal masses \[{{m}_{1}}\] and \[{{m}_{2}}\] moving along the same straight line with velocities \[+\,3\,m/s\] and \[-\,5\,m/s\] respectively, collide elastically. Their velocities after the collision will be respectively
A)
\[-\,3\,m/s\] & \[+\,5\,m/s\]
done
clear
B)
\[+\,4\,m/s\] for both
done
clear
C)
\[-\,4\,m/s\]& \[+\,4\,m/s\]
done
clear
D)
\[-\,5\,m/s\] &\[+\,3\,m/s\]
done
clear
View Answer play_arrow
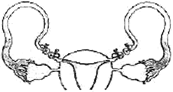
One end of uniform wire of length L and of weight W is attached rigidly to a point in the roof and a weight \[{{W}_{1}}\] is suspended from its lower end. If s is the area of cross section of the wire, the stress in the wire at a height \[\left( \frac{3L}{4} \right)\] from its lower end is
A)
\[\frac{{{W}_{1}}}{s}\]
done
clear
B)
\[\left[ {{W}_{1}}+\frac{W}{4} \right]s\]
done
clear
C)
\[\left[ {{W}_{1}}+\frac{3W}{4} \right]/s\]
done
clear
D)
\[\frac{{{W}_{1}}+W}{s}\]
done
clear
View Answer play_arrow
The maximum electric field that can be held in air without producing ionisation of air is \[{{10}^{7}}V/m.\] The maximum potential therefore, to which a conducting sphere of radius 0.10 m can be charged in air is
A)
\[{{10}^{9}}V\]
done
clear
B)
\[{{10}^{8}}V\]
done
clear
C)
\[{{10}^{7}}V\]
done
clear
D)
\[{{10}^{6}}V\]
done
clear
View Answer play_arrow
The moment of inertia of a regular circular disc of mass 0.4 kg and radius 100 cm about the axis perpendicular to the plane of the disc and passing through its centre is
A)
\[0.002\,\,kg\,{{m}^{2}}\]
done
clear
B)
\[0.02\,\,kg\,{{m}^{2}}\]
done
clear
C)
\[2\,\,kg\,{{m}^{2}}\]
done
clear
D)
\[0.2\,\,kg\,{{m}^{2}}\]
done
clear
View Answer play_arrow
Two cars of masses \[{{m}_{1}}\] and \[{{m}_{2}},\] are moving in circles of radii \[{{r}_{1}}\] and \[{{r}_{2}},\] respectively. Their speeds are such that they make complete circles in the same time (The ratio of their centripetal acceleration is
A)
\[{{m}_{1}}{{r}_{1}}:{{m}_{2}}{{r}_{2}}\]
done
clear
B)
\[{{m}_{1}}:{{m}_{2}}\]
done
clear
C)
\[{{r}_{1}}:{{r}_{2}}\]
done
clear
D)
\[1:1\]
done
clear
View Answer play_arrow
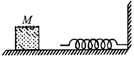
The block of mass M moving on the frictionless horizontal surface collides with the spring of spring constant k and compresses it by length L. The maximum momentum of the block after collision is
A)
\[\frac{k{{L}^{2}}}{2M}\]
done
clear
B)
\[\sqrt{Mk}\]
done
clear
C)
\[\frac{M{{L}^{2}}}{k}\]
done
clear
D)
zero
done
clear
View Answer play_arrow
Figure shows a small mass connected to a string, which is attached to a vertical post. If the ball is released when the string is horizontal as shown, the magnitude of the total acceleration of the mass as a function of the angle 9 is:
A)
\[g\,\,\sin \,\theta \]
done
clear
B)
\[g\,\,\cos \,\theta \]
done
clear
C)
\[g\sqrt{3{{\cos }^{2}}\theta +1}\]
done
clear
D)
\[g\sqrt{3{{\sin }^{2}}\theta +1}\]
done
clear
View Answer play_arrow
The angular velocities of three bodies in simple harmonic motion are \[{{\omega }_{1}},\]\[{{\omega }_{2}},\]\[{{\omega }_{3}}\] with their respective amplitudes as \[{{A}_{1}},\]\[{{A}_{2}},\]\[{{A}_{3}}.\] If all the three bodies have same mass and velocity, then
A)
\[{{A}_{1}}{{\omega }_{1}}={{A}_{2}}{{\omega }_{2}}={{A}_{3}}{{\omega }_{3}}\]
done
clear
B)
\[{{A}_{1}}^{2}{{\omega }_{1}}={{A}_{2}}^{2}{{\omega }_{2}}={{A}_{3}}^{2}{{\omega }_{3}}\]
done
clear
C)
\[{{A}_{1}}{{\omega }_{1}}^{2}={{A}_{2}}{{\omega }_{2}}^{2}={{A}_{3}}{{\omega }_{3}}^{2}\]
done
clear
D)
\[{{A}_{1}}^{2}{{\omega }_{1}}^{2}={{A}_{2}}^{2}{{\omega }_{2}}^{2}={{A}_{3}}^{2}{{\omega }_{3}}^{2}\]
done
clear
View Answer play_arrow
A pulley of radius 2 m is rotated about its axis by a force \[F=(20t-5{{t}^{2}})\]newton (where t is measured in seconds) applied tangentially. If the moment of inertia of the pulley about its axis of rotation is \[10\,\,kg-{{m}^{2}}\] the number of rotations made by the pulley before its direction of motion is reversed, is:
A)
more than 3 but less than 6
done
clear
B)
more than 6 but less than 9
done
clear
C)
more than 9
done
clear
D)
less than 3
done
clear
View Answer play_arrow
A point charge of magnitude \[+1\mu C\] is fixed at (0, 0, 0). An isolated uncharged spherical conductor, is fixed with its center at (4, 0, 0). The potential and the induced electric field at the centre of the sphere is:
A)
\[1.8\times {{10}^{5}}V\] and\[-5.625\times {{10}^{6}}V/m\]
done
clear
B)
0V and 0V/m
done
clear
C)
\[2.25\times {{10}^{5}}V\]and\[-5.625\times {{10}^{6}}V/m\]
done
clear
D)
\[2.25\times {{10}^{5}}V\]and \[0\,V/m\]
done
clear
View Answer play_arrow
We have a galvanometer of resistance \[25\,\Omega .\] It is shunted by a \[2.5\,\Omega \] wire. The part of total current that flows through the galvanometer is
A)
\[\frac{I}{{{I}_{{}^\circ }}}=\frac{2}{11}\]
done
clear
B)
\[\frac{I}{{{I}_{{}^\circ }}}=\frac{1}{11}\]
done
clear
C)
\[\frac{I}{{{I}_{{}^\circ }}}=\frac{4}{11}\]
done
clear
D)
\[\frac{I}{{{I}_{{}^\circ }}}=\frac{3}{11}\]
done
clear
View Answer play_arrow
Consider a thermodynamic system. If \[\Delta U\] represents the increase in its internal energy and W the work done by the system, which of the following statements is true?
A)
\[\Delta U=-\]W in an adiabatic process
done
clear
B)
\[\Delta U=\]W in an isothermal process
done
clear
C)
\[\Delta U=-\]W in an isothermal process
done
clear
D)
\[\Delta U=\]W in an adiabatic process
done
clear
View Answer play_arrow
The electric field at a distance r from the centre in the space between two concentric metallic spherical shells of radii \[{{r}_{1}}\] and \[{{r}_{2}}\] carrying charge \[{{Q}_{1}}\] and \[{{Q}_{2}}\]is \[({{r}_{1}}<r<{{r}_{2}})\]
A)
\[\frac{{{Q}_{1}}+{{Q}_{2}}}{4\pi {{\in }_{0}}{{({{r}_{1}}+{{r}_{2}})}^{2}}}\]
done
clear
B)
\[\frac{{{Q}_{1}}+{{Q}_{2}}}{4\pi {{\in }_{0}}{{r}^{2}}}\]
done
clear
C)
\[\frac{{{Q}_{1}}}{4\pi {{\in }_{0}}{{r}^{2}}}\]
done
clear
D)
\[\frac{{{Q}_{2}}}{4\pi {{\in }_{0}}{{r}^{2}}}\]
done
clear
View Answer play_arrow
Light of wavelength \[200\overset{{}^\circ }{\mathop{A}}\,\] fall on aluminium surface. Work function of aluminium is 4.2 eV. What is the kinetic energy of the fastest emitted photoelectrons?
A)
2 eV
done
clear
B)
1 eV
done
clear
C)
4 eV
done
clear
D)
0.2 eV
done
clear
View Answer play_arrow
Which of the following waves have the maximum wavelength?
A)
Microwaves
done
clear
B)
UV rays
done
clear
C)
Radio waves
done
clear
D)
X - rays
done
clear
View Answer play_arrow
Impedance of circuit when a resistance R and an inductor of inductance L are connected in series in an A. C. circuit of frequency v, is
A)
\[\sqrt{{{R}^{2}}+2{{\pi }^{2}}{{v}^{2}}{{L}^{2}}}\]
done
clear
B)
\[\sqrt{{{R}^{2}}+4{{\pi }^{2}}{{v}^{2}}{{L}^{2}}}\]
done
clear
C)
\[\sqrt{R+4{{\pi }^{2}}{{v}^{2}}{{L}^{2}}}\]
done
clear
D)
\[\sqrt{R+2{{\pi }^{2}}{{v}^{2}}{{L}^{2}}}\]
done
clear
View Answer play_arrow
If the wavelength of the first line of the Balmer series in the hydrogen spectrum is \[\lambda ,\] then the wavelength of the first line of the Lyman series is
A)
\[(27/5)\lambda \]
done
clear
B)
\[(5/27)\lambda \]
done
clear
C)
\[(32/27)\lambda \]
done
clear
D)
\[(27/32)\lambda \]
done
clear
View Answer play_arrow
The effective capacitance of combination of equal capacitors between points A and B shown in figure is
A)
\[C\]
done
clear
B)
\[2C\]
done
clear
C)
\[3C\]
done
clear
D)
\[\frac{C}{2}\]
done
clear
View Answer play_arrow
Which one of the following is NOT a correct statement about semi-conductors?
A)
The electrons and holes have different mobilities in a semi-conductor
done
clear
B)
In an n-type semi-conductor, the Fermi level lies closer to the conduction band edge
done
clear
C)
Silicon is a direct band gap semi-conductor
done
clear
D)
Silicon has diamond structure
done
clear
View Answer play_arrow
The sprinkling of water reduces slightly the temperature of a closed room because temperature of water is less than that of the room
A)
specific heat of water is high
done
clear
B)
water has large latent heat of vaporization
done
clear
C)
water is a bad conductor of heat
done
clear
D)
done
clear
View Answer play_arrow
Two bodies of masses m and 4m are moving with equal kinetic energies. The ratio of their linear momenta will be
A)
1 : 4
done
clear
B)
4 : 1
done
clear
C)
1 : 2
done
clear
D)
2 : 1
done
clear
View Answer play_arrow
Masses of 1 kg each are placed 1 m, 2 m, 4 m, 8 m, ..... from a point P. The gravitational field intensity at P due to these masses is
A)
\[G\]
done
clear
B)
\[G\]
done
clear
C)
\[4G\]
done
clear
D)
\[\frac{4G}{3}\]
done
clear
View Answer play_arrow
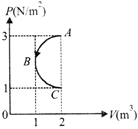
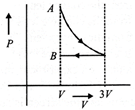
In P- Vdiagram shown in figure ABC is a semicircle. The work done in the process ABC is
A)
\[4\,\,J\]
done
clear
B)
\[\frac{-\pi }{2}J\]
done
clear
C)
\[\frac{\pi }{2}J\]
done
clear
D)
zero
done
clear
View Answer play_arrow
The electric field associated with an e.m. wave in vacuum is given by \[\overrightarrow{E}=\overrightarrow{i}\,\,40\cos (kz-6\times {{10}^{8}}t),\] where E, z and t are in volt/m, meter and seconds respectively. The value of wave vector k is
A)
\[2\,\,{{m}^{-1}}\]
done
clear
B)
\[0.5\,{{m}^{-1}}\]
done
clear
C)
\[6\,{{m}^{-1}}\]
done
clear
D)
\[3\,{{m}^{-1}}\]
done
clear
View Answer play_arrow
A bullet is fired with a speed of 1500 m/s in order to hit a target 100 m away If \[g=10m/{{s}^{2}}.\]The gun should be aimed
A)
15 cm above the target
done
clear
B)
10 cm above the target
done
clear
C)
2.2 cm above the target
done
clear
D)
directly towards the target
done
clear
View Answer play_arrow
The point charges \[Q\] and \[-2Q\] are placed at some distance apart. If the electric field at the location of\[Q\] is \[E.\] Then, the electric field at the location of \[-2Q\] will be
A)
\[-\frac{3E}{2}\]
done
clear
B)
\[-E\]
done
clear
C)
\[-\frac{E}{2}\]
done
clear
D)
\[-2E\]
done
clear
View Answer play_arrow
A cell of internal resistance r is connected to an external resistance R. The current will be maximum in R, if
A)
\[R=r\]
done
clear
B)
\[R<r\]
done
clear
C)
\[R>r\]
done
clear
D)
\[R={r}/{2}\;\]
done
clear
View Answer play_arrow
A vessel of depth x is half filled with oil of refractive index \[{{\mu }_{1}}\] and the other half is filled with water of refractive index \[{{\mu }_{2.}}\] The apparent depth of the vessel when viewed from above is
A)
\[\frac{x({{\mu }_{1}}+{{\mu }_{2}})}{2{{\mu }_{1}}{{\mu }_{2}}}\]
done
clear
B)
\[\frac{x{{\mu }_{1}}{{\mu }_{2}}}{2({{\mu }_{1}}+{{\mu }_{2}})}\]
done
clear
C)
\[\frac{x{{\mu }_{1}}{{\mu }_{2}}}{({{\mu }_{1}}+{{\mu }_{2}})}\]
done
clear
D)
\[\frac{2x({{\mu }_{1}}+{{\mu }_{2}})}{{{\mu }_{1}}{{\mu }_{2}}}\]
done
clear
View Answer play_arrow
A cylindrical block of area of cross-section A and of material of density p is placed in a liquid of density one-third of density of block. The block compresses a spring and compression in the spring is one-third of the length of the block. If acceleration due to gravity is g, the spring constant of the spring is
A)
\[\rho Ag\]
done
clear
B)
\[2\rho Ag\]
done
clear
C)
\[2\rho Ag/3\]
done
clear
D)
\[\rho Ag/3\]
done
clear
View Answer play_arrow
In A.C. circuit in which inductance and capacitance are joined in series, current is found to be maximum when the value of inductance is 0.5 H and the value of capacitance is \[8\mu F.\] The angular frequency of applied alternating voltage will be
A)
\[400\,Hz\]
done
clear
B)
\[5000\,Hz\]
done
clear
C)
\[2\times {{10}^{5}}\,Hz\]
done
clear
D)
\[500\,Hz\]
done
clear
View Answer play_arrow
Angle between magnetic meridian and geographical meridian is called
A)
angle of dip
done
clear
B)
angle of declination
done
clear
C)
angle of depression
done
clear
D)
both [a] and [b]
done
clear
View Answer play_arrow
Heat is required to change 1 kg of ice of\[-20{}^\circ C\] into steam. \[{{Q}_{1}}\] is the heat needed to warm the ice from \[-20{}^\circ C\] to \[0{}^\circ C,\]\[{{Q}_{2}}\] is the heat needed to melt the ice, \[{{Q}_{3}}\] is the heat needed to warm the water from \[0{}^\circ C\] to \[100{}^\circ C\] and\[{{Q}_{4}}\] is the heat needed to vaporize the water. Then
A)
\[{{Q}_{4}}>{{Q}_{3}}>{{Q}_{2}}>{{Q}_{1}}\]
done
clear
B)
\[{{Q}_{4}}>{{Q}_{3}}>{{Q}_{1}}>{{Q}_{2}}\]
done
clear
C)
\[{{Q}_{4}}>{{Q}_{2}}>{{Q}_{3}}>{{Q}_{1}}\]
done
clear
D)
\[{{Q}_{4}}>{{Q}_{2}}>{{Q}_{1}}>{{Q}_{3}}\]
done
clear
View Answer play_arrow
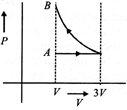
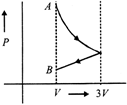
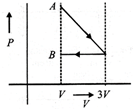
One mole of an ideal gas goes from an initial state A to final state B via two processes: It first undergoes isothermal expansion from volume V to 3 V and then its volume is reduced from 3 V to V at constant pressure. The correct P- V diagram representing the two processes is :
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
A current carrying wire in the neighbourhood produces
A)
electric and magnetic field
done
clear
B)
magnetic field only
done
clear
C)
no field
done
clear
D)
electric field only
done
clear
View Answer play_arrow
10 gm of ice cubes at \[0{}^\circ C\] are released in a tumbler (water equivalent 55 g) at \[40{}^\circ C.\] Assuming that negligible heat is taken from the surroundings, the temperature of water in the tumbler becomes nearly \[(L=80{cal}/{g}\;)\]
A)
\[31{}^\circ C\]
done
clear
B)
\[22{}^\circ C\]
done
clear
C)
\[19{}^\circ C\]
done
clear
D)
\[15{}^\circ C\]
done
clear
View Answer play_arrow
The internal energy of an ideal gas increases during an isothermal process when the gas is
A)
expanded by adding more molecules to it
done
clear
B)
expanded by adding more heat to it
done
clear
C)
expanded against zero pressure
done
clear
D)
compressed by doing work on it
done
clear
View Answer play_arrow
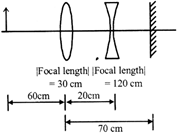
A convex lens, of focal length 30 cm, a concave lens of focal length 120 cm, and a plane mirror are arranged as shown. For an object kept at a distance of 60 cm from the convex lens, the final image, formed by the combination, is a real image, at a distance of:
A)
60 cm from the convex lens
done
clear
B)
60 cm from the concave lens
done
clear
C)
70 cm from the convex lens
done
clear
D)
70 cm from the concave lens
done
clear
View Answer play_arrow
In Young's double-slit experiment, the separation between the slits is halved and the distance between the slits and the screen is doubled. The fringe width is
A)
unchanged
done
clear
B)
halved
done
clear
C)
doubled
done
clear
D)
quadrupled
done
clear
View Answer play_arrow
The number of antibonding electron pairs in \[O_{2}^{2-}\] molecular ion on the basis of molecular orbital theory are (at. no. O=8)
A)
2
done
clear
B)
3
done
clear
C)
4
done
clear
D)
5
done
clear
View Answer play_arrow
Consider the following reaction:
\[4N{{H}_{3}}(g)+5{{O}_{2}}(g)\xrightarrow{Pt(s)}4NO(g)+6{{H}_{2}}O(g)\] (i) Rate of reaction with respect to \[N{{H}_{3}}\]will be \[-\frac{1}{4}\frac{\Delta [N{{H}_{3}}]}{\Delta t}\] (ii) For the given reaction \[-\frac{1}{5}\frac{\Delta [{{O}_{2}}]}{\Delta t}=\frac{-1}{6}\frac{\Delta [{{H}_{2}}O]}{\Delta t}\] (iii) For the given reaction \[-\frac{1}{4}\frac{\Delta [N{{H}_{3}}]}{\Delta t}=\frac{1}{4}\frac{\Delta [NO]}{\Delta t}\] (iv) For the given reaction, Rate\[=-\frac{1}{4}\frac{\Delta [N{{H}_{3}}]}{\Delta t}=-\frac{1}{5}\frac{\Delta [{{O}_{2}}]}{\Delta t}\] \[=\frac{4\Delta [NO]}{\Delta t}=\frac{6\Delta [{{H}_{2}}O]}{\Delta t}\] Which of the following are the correct statements.
A)
(i) and (iv)
done
clear
B)
(i) and (iii)
done
clear
C)
(ii) and (iv)
done
clear
D)
All are correct
done
clear
View Answer play_arrow
Read the following statements carefully.
(i) According to a convention cell potential of hydrogen electrode (S.H.E.) is considered to be zero at all temperatures. (ii) e.m.f. of the cell \[Pt(s)/{{H}_{2}}(g,1\,\,bar)/{{H}^{+}}(aq,1M)||Z{{n}^{2+}}(aq,1M)/Zn\] is \[-0.76\]This negative value indicates that \[Z{{n}^{2+}}\]ion reduces less easily then \[{{H}^{+}}\] ions. (iii) Copper does not dissolve in HCl but dissolves in \[HN{{O}_{3}}\] as in nitric acid it gets oxidised by nitrate ion. (iv) Inert metals like Pt or Au are used in certain electrodes i.e., these metals does not participate in reaction but provide surface for oxidation and reduction reactions. (v) Fluorine has the highest electrode potential thereby making it strongest oxidising agent whereas lithium with lowest electrode potential is the weakest oxidising and strongest reducing agent. Which of the following are the correct statements above?
A)
(iv) and (v)
done
clear
B)
(i), (ii) and (iv)
done
clear
C)
(iii), (iv) and (v)
done
clear
D)
All are correct
done
clear
View Answer play_arrow
The transition metals have a less tendency to form ions due to:
A)
High ionisation energy.
done
clear
B)
Low heat of hydration of ions.
done
clear
C)
High heat of sublimation.
done
clear
D)
All of the above.
done
clear
View Answer play_arrow
\[2{{C}_{6}}{{H}_{5}}CHO\xrightarrow[{{H}_{2}}O]{O{{H}^{-}}}{{C}_{6}}{{H}_{5}}C{{H}_{2}}OH+{{C}_{6}}{{H}_{5}}CO{{O}^{-}}\] Which of the following statements are correct regarding the above reduction of benzaldehyde to benzyl alcohol?
(i) One hydrogen is coming from \[{{H}_{2}}O\] as \[{{H}^{+}}\] and another from \[{{C}_{6}}{{H}_{5}}CHO\] as \[{{H}^{-}}\] (ii) One hydrogen is coming from \[{{H}_{2}}O\] as \[{{H}^{-}}\] and another from \[{{C}_{6}}{{H}_{5}}CHO\] as \[{{H}^{+}}\] (iii) One hydrogen from \[{{H}_{2}}O\] and another from\[{{C}_{6}}{{H}_{5}}CHO,\] both in the form of\[{{H}^{-}}\] (iv) The reduction is an example of disproportionation reaction
A)
(i), (ii) and (iii)
done
clear
B)
(i) and (iv)
done
clear
C)
(ii),(iii) and (iv)
done
clear
D)
(iii) and (iv)
done
clear
View Answer play_arrow
Read the following statements and choose the correct option.
(i) The numerical value of the equilibrium constant for a reaction indicates the extent of the reaction. (ii) An equilibrium constant give information about the rate at which the equilibrium is reached. (iii) If \[{{K}_{c}}>{{10}^{3}},\] products predominate over reactants, i.e., if \[{{K}_{c}}\] is very large, the reaction proceeds nearly to completion. (iv) If \[{{K}_{c}}<{{10}^{-3}},\] reactants predominate over products, i.e., if \[{{K}_{c}}\] is very small, the reaction proceeds rarely.
A)
(i), (ii) and (iv) are correct
done
clear
B)
(i), (iii) and (iv)
done
clear
C)
(i), (ii) and (iii) are correct
done
clear
D)
only (iii) is correct
done
clear
View Answer play_arrow
The important antioxidant used in food is:
A)
BHT
done
clear
B)
BHC
done
clear
C)
BTX
done
clear
D)
All the three
done
clear
View Answer play_arrow
Brine is electrolysed by using inert electrodes. The reaction at anode is _____.
A)
\[C{{l}^{-}}(aq)\xrightarrow{{}}\frac{1}{2}C{{l}_{2}}(g)+{{e}^{-}};\]\[E_{cell}^{{}^\circ }=1.36V\]
done
clear
B)
\[2{{H}_{2}}O(1)\xrightarrow{{}}{{O}_{2}}(g)+4{{H}^{+}}4{{e}^{-}};\]\[E_{cell}^{{}^\circ }=1.23V\]
done
clear
C)
\[N{{a}^{+}}(aq)+{{e}^{-}}\xrightarrow{{}}Na(s);\]\[E_{cell}^{{}^\circ }=2.71V\]
done
clear
D)
\[{{H}^{+}}(aq)+{{e}^{-}}\xrightarrow{{}}\frac{1}{2}{{H}_{2}}(g);\]\[E_{cell}^{{}^\circ }=0.00V\]
done
clear
View Answer play_arrow
Geometrical isomerism is not shown by
A)
done
clear
B)
\[{{C}_{2}}{{H}_{5}}-\underset{H}{\mathop{\underset{|}{\mathop{C}}\,}}\,=\underset{H}{\mathop{\underset{|}{\mathop{C}}\,}}\,-C{{H}_{2}}I\]
done
clear
C)
\[C{{H}_{2}}=C(CI)C{{H}_{3}}\]
done
clear
D)
\[C{{H}_{3}}-CH=CH-CH=C{{H}_{2}}\]
done
clear
View Answer play_arrow
Schotten-Baumann reaction is a reaction of phenols with:
A)
Benzoyl chloride and sodium hydroxide.
done
clear
B)
Acetyl chloride and sodium hydroxide.
done
clear
C)
Salicylic acid and cone. \[{{H}_{2}}S{{O}_{4}}.\]
done
clear
D)
Acetyl chloride and cone. \[{{H}_{2}}S{{O}_{4}}.\]
done
clear
View Answer play_arrow
Which of the following statements are correct?
(i) Ionic product of water \[({{K}_{w}})=[{{H}^{+}}][O{{H}^{-}}]={{10}^{-14}}{{M}^{2}}\] (ii) At \[298K[{{H}^{+}}]=[O{{H}^{-}}]={{10}^{-7}}\] (iii) \[{{K}_{w}}\] does not depends upon temperature (iv) Molarity of pure water = 55.55M
A)
(i), (ii) and (iii)
done
clear
B)
(i), (ii) and (iv)
done
clear
C)
(i) and (iv)
done
clear
D)
(ii) and (iii)
done
clear
View Answer play_arrow
According to the adsorption theory of catalysis, the speed of the reaction increases because
A)
in the process of adsorption, the activation energy of the molecules becomes large.
done
clear
B)
adsorption produces heat which increases the speed of the reaction.
done
clear
C)
adsorption lowers the activation energy of the reaction.
done
clear
D)
the concentration of product molecules at the active centres of the catalyst becomes high due to adsorption.
done
clear
View Answer play_arrow
Consider the following reaction at \[1000{}^\circ C\] A.\[Zn(s)+\frac{1}{2}{{O}_{2}}(g)\to ZnO(s);\]\[\Delta G{}^\circ =-360kJ\,mo{{l}^{-1}}\] B. \[C\left( gr \right)+\frac{1}{2}{{O}_{2}}(g)\to CO(g);\]\[\Delta G{}^\circ =-460kJ\,mo{{l}^{-1}}\] Choose the correct statement at \[1000{}^\circ C\]
A)
zinc can be oxidised by carbon monoxide.
done
clear
B)
zinc oxide can be reduced by graphite.
done
clear
C)
both statements [a] and [b] are true.
done
clear
D)
carbon monoxide can be reduced by zinc.
done
clear
View Answer play_arrow
Consider the following complex \[[Co{{(N{{H}_{3}})}_{5}}C{{O}_{3}}]Cl{{O}_{4.}}\] The coordination number, oxidation number, number of d-electrons and number of unpaired d-electrons on the metal are respectively
A)
6, 3, 6, 0
done
clear
B)
7, 2, 7, 1
done
clear
C)
7, 1, 6, 4
done
clear
D)
6, 2, 7, 3
done
clear
View Answer play_arrow
Match List I (Reaction) with List II (Reagent) and select the correct answer using the codes given below the lists:
List I List II I. Etard reaction A. Alcoholic KOH II. Hydroxylation B. Anhydrous\[AlC{{l}_{3}}\] III. Dehydro-halogenation C. Chromyl chloride IV. Friedel-Crafts reaction D. Dilute alkaline \[KMn{{O}_{4}}\]
Codes:
A)
I\[\to \]A, II\[\to \]B, III\[\to \]C, IV\[\to \]B
done
clear
B)
I\[\to \]D, II\[\to \]C, III\[\to \]A, IV\[\to \]B
done
clear
C)
I\[\to \]C, II\[\to \]D, III\[\to \]A, IV\[\to \]B
done
clear
D)
I\[\to \]B, II\[\to \]A, III\[\to \]D, IV\[\to \]C
done
clear
View Answer play_arrow
The solubility product of AgI at \[25{}^\circ C\] is \[1.0\times {{10}^{-16}}mo{{l}^{2}}{{L}^{-2}}.\] The solubility of AgI in\[{{10}^{-4}}N\] solution of KI at \[25{}^\circ C\] is approximately (in \[mol\,{{L}^{-1}}\])
A)
\[1.0\times {{10}^{-12}}\]
done
clear
B)
\[1.0\times {{10}^{-10}}\]
done
clear
C)
\[1.0\times {{10}^{-8}}\]
done
clear
D)
\[1.0\times {{10}^{-16}}\]
done
clear
View Answer play_arrow
Match the columns
Column-I Column-II [A] \[\frac{x}{m}=k{{c}^{{1}/{n}\;}}\] (p) Adsorption varies directly with pressure [B] \[\log \frac{x}{m}=\log k+\frac{1}{n}\log p\left( \frac{1}{n}=0 \right)\] (q) Adsorption from solution phase [C] \[\log \frac{x}{m}=\log k+\frac{1}{n}\log p\left( \frac{1}{n}=1 \right)\] (r) Freudlich isotherm cannot be explained [D] \[\frac{x}{m}=k{{p}^{{1}/{n}\;}}\] (s) Adsorption is (high pressure) independent of pressure
A)
A\[\to \](q), B\[\to \](s), C\[\to \](p), D\[\to \](r)
done
clear
B)
A\[\to \](q), B\[\to \](p), C\[\to \](s), D\[\to \](r)
done
clear
C)
A\[\to \](r), B\[\to \](p), C\[\to \](s), D\[\to \](q)
done
clear
D)
A\[\to \](r), B\[\to \](s), C\[\to \](p), D\[\to \](q)
done
clear
View Answer play_arrow
Which one of the following is the correct statement?
A)
\[{{B}_{2}}{{H}_{6}}.2N{{H}_{3}}\] is known as 'inorganic benzene?.
done
clear
B)
Boric acid is a protonic acid.
done
clear
C)
Beryllium exhibits coordination number of six.
done
clear
D)
Chlorides of both beryllium and aluminium have bridged chloride structures in solid phase.
done
clear
View Answer play_arrow
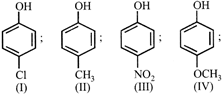
Arrange the following compounds in order of decreasing acidity:
A)
\[II>IV>I>III\]
done
clear
B)
\[I>II>III>IV\]
done
clear
C)
\[III>I>II>IV\]
done
clear
D)
\[IV>III>I>II\]
done
clear
View Answer play_arrow
Teflon, styron and neoprene are all:
A)
Copolymers
done
clear
B)
Condensation polymers
done
clear
C)
Homopolymers
done
clear
D)
Monomers
done
clear
View Answer play_arrow
Values of dissociation constant, \[{{K}_{a}}\] are given as follows:
Acid \[{{K}_{a}}\] HCN \[6.2\times {{10}^{-10}}\] HF \[7.2\times {{10}^{-4}}\] \[HN{{O}_{2}}\] \[4.0\times {{10}^{-4}}\]
Correct order of increasing base strength of the base \[C{{N}^{-,}}\]\[{{F}^{-}}\] and \[NO_{2}^{-}\] will be:
A)
\[{{F}^{-}}<C{{N}^{-}}<NO_{2}^{-}\]
done
clear
B)
\[NO_{2}^{-}<C{{N}^{-}}<{{F}^{-}}\]
done
clear
C)
\[{{F}^{-}}<NO_{2}^{-}<C{{N}^{-}}\]
done
clear
D)
\[NO_{2}^{-}<{{F}^{-}}<C{{N}^{-}}\]
done
clear
View Answer play_arrow
Which of the following statement is correct if the intermolecular forces in liquids A, B and C are in the order\[A<B<C?\]
A)
B evaporates more readily than A
done
clear
B)
B evaporates less readily than C
done
clear
C)
A and B evaporate at the same rate
done
clear
D)
A evaporates more readily than C
done
clear
View Answer play_arrow
The reducing power of divalent species decreases in the order
A)
\[Ge>Sn>Pb\]
done
clear
B)
\[Sn>Ge>Pb\]
done
clear
C)
\[Pb>Sn>Ge\]
done
clear
D)
None.
done
clear
View Answer play_arrow
Sodium Carbonate cannot be used in place of \[{{(N{{H}_{4}})}_{2}}C{{O}_{3}}\] for the identification of \[C{{a}^{2+}},\]\[B{{a}^{2+}}\] and \[S{{r}^{2+}}\]ions (in group V) during mixture analysis because :
A)
\[M{{g}^{2+}}\] ions will also be precipitated.
done
clear
B)
Concentration of\[C{{O}_{3}}^{2-}\] ions is very low.
done
clear
C)
Sodium ions will react with acid radicals.
done
clear
D)
\[N{{a}^{+}}\] ions will interfere with the detection of \[C{{a}^{2+}},\]\[B{{a}^{2+}},\]\[S{{r}^{2+}}\] ions.
done
clear
View Answer play_arrow
Which of the following statements is not correct regarding aniline?
A)
It is less basic than ethylamine.
done
clear
B)
It can be steam-distilled.
done
clear
C)
It reacts with sodium to give hydrogen.
done
clear
D)
It is soluble in water.
done
clear
View Answer play_arrow
Which of the following equilibria will shift to right side on increasing the temperature?
A)
\[CO(g)+{{H}_{2}}O(g)C{{O}_{2}}(g)+{{H}_{2}}(g)\]
done
clear
B)
\[2S{{O}_{2}}(g)+{{O}_{2}}(g)2S{{O}_{3}}(g)\]
done
clear
C)
\[{{H}_{2}}O(g){{H}_{2}}(g)+\frac{1}{2}{{O}_{2}}(g)\]
done
clear
D)
\[4HCl(g)+{{O}_{2}}(g)2{{H}_{2}}O(g)+2C{{l}_{2}}(g)\]
done
clear
View Answer play_arrow
An aqueous solution of sucrose, \[{{C}_{12}}{{H}_{22}}{{O}_{11}},\] containing 34.2 g has an osmotic pressure of 2.38 atmospheres at \[17{}^\circ C.\] For an aqueous solution of glucose, \[{{C}_{6}}{{H}_{12}}{{O}_{6}}\] to be isotonic with this solution, it would have
A)
34.2 g/lit
done
clear
B)
17.1 g/lit
done
clear
C)
18.0 g/lit
done
clear
D)
36.0 g/lit of glucose
done
clear
View Answer play_arrow
Prevention of corrosion of iron by Zn coating is called:
A)
Galvanization
done
clear
B)
Cathodic protection
done
clear
C)
Electrolysis
done
clear
D)
Photoelectrolysis
done
clear
View Answer play_arrow
Aromatic character of benzene is proved by:
A)
Resonance theory.
done
clear
B)
Aromatic sextet theory.
done
clear
C)
Orbital theory.
done
clear
D)
All the above.
done
clear
View Answer play_arrow
RNA is different from DNA because RNA contains
A)
ribose sugar and thymine.
done
clear
B)
ribose sugar and uracil.
done
clear
C)
deoxyribose sugar and thymine.
done
clear
D)
deoxyribose sugar and uracil.
done
clear
View Answer play_arrow
The de Broglie wavelength of a tennis ball of mass 60 g moving with a velocity of 10 m/s is approximately
A)
\[{{10}^{-33}}\text{metres}\]
done
clear
B)
\[{{10}^{-31}}\text{metres}\]
done
clear
C)
\[{{10}^{-16}}\text{metres}\]
done
clear
D)
\[-N{{H}_{2}}\] Planck's constant, \[h=6.63\times {{10}^{-34}}Js\]
done
clear
View Answer play_arrow
AB, \[{{A}_{2}}\] and \[{{B}_{2}}\] are diatomic molecules. If the bond enthalpies of \[{{A}_{2}},\] AB and \[{{B}_{2}}\] are in the ratio 1:1:0.5 and enthalpy of formation of AB from \[{{A}_{2}}\]and \[{{B}_{2}}\] is \[-100kJ\,mo{{l}^{-1}}.\] What is the bond energy of \[{{A}_{2}}:\]
A)
\[200\,kJ\,mo{{l}^{-1}}\]
done
clear
B)
\[100\,kJ\,mo{{l}^{-1}}\]
done
clear
C)
\[300\,kJ\,mo{{l}^{-1}}\]
done
clear
D)
done
clear
View Answer play_arrow
Beryllium shows diagonal relationship with aluminium. Which of the following similarity is incorrect:
A)
Be forms beryllates and Al forms aluminates.
done
clear
B)
Be \[{{(OH)}_{2}}\] like \[Al{{(OH)}_{3}}\] is basic.
done
clear
C)
Be like Al is rendered passive by \[HN{{O}_{3}}.\]
done
clear
D)
\[B{{e}_{2}}C\] like \[A{{l}_{4}}{{C}_{3}}\] yields methane on hydrolysis.
done
clear
View Answer play_arrow
and
are
A)
Resonating structures
done
clear
B)
Tautomers
done
clear
C)
Geometrical isomers
done
clear
D)
Optical isomers
done
clear
View Answer play_arrow
The fatty acid which shows reducing property is
A)
Acetic acid
done
clear
B)
Ethanoic acid
done
clear
C)
Oxalic acid
done
clear
D)
Formic acid
done
clear
View Answer play_arrow
If the nitrogen atom had electronic configuration \[1{{s}^{7}}\] it would have energy lower than that of the normal ground state configuration \[1{{s}^{2}}2{{s}^{2}}2{{p}^{3}}\] because the electrons would be closer to the nucleus. Yet \[1{{s}^{7}}\] is not observed. It violates
A)
Heisenberg's uncertainty principle
done
clear
B)
Hund's rule
done
clear
C)
Pauli exclusion principle
done
clear
D)
Bohr postulate of stationary orbits
done
clear
View Answer play_arrow
For orthorhombic system axial ratios are\[a\ne b\ne c\] and the axial angles are
A)
\[\alpha =\beta =\gamma \ne 90{}^\circ \]
done
clear
B)
\[\alpha =\beta =\gamma =90{}^\circ \]
done
clear
C)
\[\alpha =\gamma =90{}^\circ ,\]\[\beta \ne 90{}^\circ \]
done
clear
D)
\[\alpha \ne \beta \ne \gamma =90{}^\circ \]
done
clear
View Answer play_arrow
When cyclohexane is poured on water, it floats, because
A)
cyclohexane is in ?boat? form.
done
clear
B)
cyclohexane is in ?chair? form.
done
clear
C)
cyclohexane is in ?crown? form.
done
clear
D)
cyclohexane is less dense than water.
done
clear
View Answer play_arrow
\[(n-1){{d}^{10}}n{{s}^{2}}\] is the general electronic configuration of
A)
Fe, Co, Ni
done
clear
B)
Cu, Ag, Au
done
clear
C)
Zn, Cd, Hg
done
clear
D)
Se, Y, La
done
clear
View Answer play_arrow
Which is colourless in water?
A)
\[T{{i}^{3+}}\]
done
clear
B)
\[{{V}^{3+}}\]
done
clear
C)
\[C{{u}^{3+}}\]
done
clear
D)
\[S{{c}^{3+}}\]
done
clear
View Answer play_arrow
Structurally biodegrdable detergent should contain
A)
normal alkyi chain.
done
clear
B)
branched alkyi chain.
done
clear
C)
phenyl side chain.
done
clear
D)
cyclohexyl side chain.
done
clear
View Answer play_arrow
Which of the following crystals does not exhibit Frenkel defect?
A)
AgBr
done
clear
B)
AgCl
done
clear
C)
KBr
done
clear
D)
ZnS
done
clear
View Answer play_arrow
A reaction which is of first order w.r.t. reactant A, has a rate constant \[6\,{{\min }^{-1}}.\] If we start with \[[A]=0.5\,mol\,{{L}^{-1}},\]when would \[[A]\] reach the value of \[0.5\,mol\,{{L}^{-1}}\]
A)
0.384 min
done
clear
B)
0.15 min
done
clear
C)
3 min
done
clear
D)
3.84 min
done
clear
View Answer play_arrow
Which pair of elements belongs to same group?
A)
Elements with atomic no.17and 38
done
clear
B)
Elements with atomic no. 20 and 40
done
clear
C)
Elements with atomic no. 17 and 53
done
clear
D)
Elements with atomic no. 11 and 33
done
clear
View Answer play_arrow
Which of the following statements about low density polythene is FALSE?
A)
Its synthesis requires dioxygen or a peroxide initiator as a catalyst.
done
clear
B)
It is used in the manufacture of buckets, dust-bins etc.
done
clear
C)
Its synthesis requires high pressure.
done
clear
D)
It is a poor conductor of electricity.
done
clear
View Answer play_arrow
Which plays an important role in the dispersal of spores in Funarial
A)
Operculum
done
clear
B)
Capsule
done
clear
C)
Peristome and annulus
done
clear
D)
Sporogonium
done
clear
View Answer play_arrow
Read the following five statements (i - v) and answer the question.
(i) In Equisetum, the female gametophyte is retained on the parent sporophyte. (ii) In Ginkgo, male gametophyte is not independent. (iii) The sporophyte in Riccia is more developed than that in Polytrichum. (iv) Sexual reproduction in Volvox is isogamous. (v) The spores of slime molds lack cell walls. How many of the above statements are correct?
A)
Two
done
clear
B)
Three
done
clear
C)
Four
done
clear
D)
One
done
clear
View Answer play_arrow
The fluidity of membranes in a plant in cold weather may be maintained by
A)
increasing the number of phospholipids with unsaturated hydrocarbon tails
done
clear
B)
increasing the proportion of integral proteins
done
clear
C)
increasing concentration of cholesterol in membrane
done
clear
D)
increasing the number of phospholipids with saturated hydrocarbon tail
done
clear
View Answer play_arrow
Addition of a solute to pure water causes
A)
negative water potential.
done
clear
B)
more negative water potential.
done
clear
C)
positive water potential.
done
clear
D)
more positive water potential.
done
clear
View Answer play_arrow
Which one of the following process help the water- soluble inorganic nutrients go down into the soil horizon and get precipitated as unavailable salts?
A)
Fragmentation
done
clear
B)
Leaching
done
clear
C)
Catabolism
done
clear
D)
Humification
done
clear
View Answer play_arrow
Siliceous frustules of diatoms being indestructible, piled up at the bottom of ocean and formed a thick bed over billions of years. Such a thick bed is known as
A)
red sea
done
clear
B)
diatomaceous earth
done
clear
C)
pseudorocks
done
clear
D)
red tides
done
clear
View Answer play_arrow
The first dicarboxylic acid in Krebs' cycle is
A)
isocitric acid
done
clear
B)
pyruvic acid
done
clear
C)
oxalo acetic acid
done
clear
D)
\[\alpha \]-ketoglutaric acid
done
clear
View Answer play_arrow
\[{{N}_{2}}+8{{e}^{-}}+8{{H}^{+}}+16\,ATP\to \]\[2N{{H}_{4}}+{{H}_{2}}+16ADP+16Pi\] The above equation refers to
A)
ammonification
done
clear
B)
nitrification
done
clear
C)
nitrogen fixation
done
clear
D)
denitrification
done
clear
View Answer play_arrow
A pressure that is responsible for pushing up water to small height in the stem is called
A)
positive root pressure
done
clear
B)
turgor pressure
done
clear
C)
pressure gradient
done
clear
D)
negative root pressure
done
clear
View Answer play_arrow
A cell cannot divide if it does not cross
A)
Hayflick limit
done
clear
B)
cytokinesis
done
clear
C)
restriction point
done
clear
D)
\[{{G}_{0}}\] - phase
done
clear
View Answer play_arrow
A vascular bundle in which the protoxylem is pointing to the periphery is called
A)
endarch
done
clear
B)
exarch
done
clear
C)
radial
done
clear
D)
closed
done
clear
View Answer play_arrow
Which one of the following micro-organisms is used for production of citric acid in industries?
A)
Penicillium citrinum
done
clear
B)
Aspergillus niger
done
clear
C)
Rhizopus nigricans
done
clear
D)
Lactobacillus bulgaris
done
clear
View Answer play_arrow
Emasculation is not required when flowers are
A)
bisexual
done
clear
B)
intersexual
done
clear
C)
unisexual
done
clear
D)
either [a] or [b]
done
clear
View Answer play_arrow
Which of the following pairs is not correctly matched?
A)
Anabaena - Cyanobacteria
done
clear
B)
Amoeba - Protozoa
done
clear
C)
Gonyaulax - Dinoflagellates
done
clear
D)
Albugo - Chrysophytes
done
clear
View Answer play_arrow
Which part of the coconut produces coir?
A)
Seed coat
done
clear
B)
Mesocarp
done
clear
C)
Epicarp
done
clear
D)
Pericarp
done
clear
View Answer play_arrow
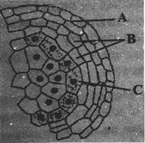
The given diagram shows microsporangium of a mature anther. Identify A, B and C.
A)
A-Middle layer, B-Endothecium, C-Tapetum
done
clear
B)
A-Endothecium, B-Tapetum, C-Middle layer
done
clear
C)
A-Endothecium, B-Middle layer, C-Tapetum
done
clear
D)
A-Tapetum, B-Middle layer, C-Endothecium
done
clear
View Answer play_arrow
The productivity of a crop declines when leaves begin to wilt mainly because
A)
the chlorophyll of wilting leaves decomposes.
done
clear
B)
flaccid mesophyll cells are incapable of photosynthesis.
done
clear
C)
stomata close, preventing \[C{{O}_{2}}\] from entering the leaf.
done
clear
D)
photolysis, the water-splitting step of photosynthesis, cannot occur when there is a water deficiency.
done
clear
View Answer play_arrow
Which one of the following is not a function of an ecosystem?
A)
Energy flow
done
clear
B)
Decomposition
done
clear
C)
Productivity
done
clear
D)
Stratification
done
clear
View Answer play_arrow
The process in which mature differentiated cells reverse to meristematic activity to form callus is called
A)
dedifferentiation
done
clear
B)
differentiation
done
clear
C)
redifferentiation
done
clear
D)
None of the above
done
clear
View Answer play_arrow
Which one of the following statement is true?
A)
The greater the Biological Oxygen Demand (BOD) of waste water, more is its polluting potential.
done
clear
B)
The greater the BOD of waste water, less is its polluting potential.
done
clear
C)
The lesser the BOD of waste water, more is its polluting potential.
done
clear
D)
The lesser the BOD of waste water, less is its polluting potential.
done
clear
View Answer play_arrow
Most of the net primary productivity in an ecosystem is used for what purpose?
A)
respiration by primary consumers
done
clear
B)
respiration by secondary consumers
done
clear
C)
growth by primary consumers
done
clear
D)
growth by secondary consumers
done
clear
View Answer play_arrow
Endemic plants and animals are those which are
A)
cosmopolitan in distribution.
done
clear
B)
restricted to certain area.
done
clear
C)
found in arctic region.
done
clear
D)
gregarious in habit.
done
clear
View Answer play_arrow
Micro-propagation is
A)
propagation of microbes in vitro
done
clear
B)
propagation of plants in vitro
done
clear
C)
propagation of cells in vitro
done
clear
D)
growing plants on smaller scale
done
clear
View Answer play_arrow
Final electron acceptor in oxidative phosphorylation is
A)
hydrogen
done
clear
B)
dehydrogenase
done
clear
C)
cytochrome
done
clear
D)
oxygen
done
clear
View Answer play_arrow
In a CAM plant, the concentration of organic acid
A)
increases during the day.
done
clear
B)
decreases during the day.
done
clear
C)
increases during night.
done
clear
D)
decreases or increases during day.
done
clear
View Answer play_arrow
The phenomenon ofplasmolysis is evident when cells are kept in
A)
hypotonic solution
done
clear
B)
hypertonic solution
done
clear
C)
isotonic solution
done
clear
D)
None of the above
done
clear
View Answer play_arrow
Quantasomes are found in
A)
mitochondria
done
clear
B)
ehloroplast
done
clear
C)
lysosome
done
clear
D)
endoplasmic reticulum
done
clear
View Answer play_arrow
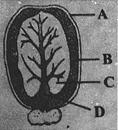
Identify the parts labelled as A, B, C and D in the given figure and select the correct option from the codes given below.
A)
A - Seed coat B - Scutellum C - Epicotyl D - Hypocotyl
done
clear
B)
A - Seed coat B - Scutellum C - Hypocotyl D - Epicotyl
done
clear
C)
A - Seed coat B - Cotyledon C - Endosperm D - Hypocotyl
done
clear
D)
A - Seed coat B - Endosperm C - Cotyledon D - Hypocotyl
done
clear
View Answer play_arrow
Which one of the following is a correct statement?
A)
Pteridophyte gametophyte has a protonemal and leafy stage
done
clear
B)
In gymnosperms, female gametophyte is free-living
done
clear
C)
Antheridiophores and archegoniophores are present in pteridophytes
done
clear
D)
Origin of seed habit can be traced in pteridophytes
done
clear
View Answer play_arrow
Function of suspensor of embryo is
A)
absorption of nourishment.
done
clear
B)
push the embryo into nutritive endosperm region.
done
clear
C)
formation of secondary embryos.
done
clear
D)
all of the above
done
clear
View Answer play_arrow
Pineapple (ananas) fruit develops from
A)
a multipistillate syncarpous flower
done
clear
B)
a cluster of compactly borne flowers on a common axis
done
clear
C)
a multilocular monocarpellary flower
done
clear
D)
a unilocular polycarpellary flower
done
clear
View Answer play_arrow
In a raceme, the flowers are-
A)
Arranged in a basipetal succession
done
clear
B)
Arranged in acropetal succession
done
clear
C)
Separate sexes
done
clear
D)
Of the same sex
done
clear
View Answer play_arrow
The common bottle cork is a product of
A)
Dermatogen
done
clear
B)
Phellogen
done
clear
C)
Xyiem
done
clear
D)
Vascular cambium
done
clear
View Answer play_arrow
Telomere and eukaryotic chromosome possesses short segments of
A)
guanine rich repeats.
done
clear
B)
thymine rich repeats.
done
clear
C)
cytosine rich repeats.
done
clear
D)
adenine rich repeats.
done
clear
View Answer play_arrow
What mechanism explains the movement of sucrose from source to sink?
A)
Evaporation of water and active transport of sucrose from sink.
done
clear
B)
Osmotic movement of water into the sucrose loaded sieve tube cells creating a higher hydrostatic pressure into the source than in the sink.
done
clear
C)
Tension created by differences in hydrostatic pressure in the source and sink.
done
clear
D)
Active transport of sucrose through the sieve tube membranes driven by proton pump.
done
clear
View Answer play_arrow
Which one of the following pairs is not correctly matched:
A)
Manas Sanctuary - Assam
done
clear
B)
Kanha National park - Madhya Pradesh
done
clear
C)
Dachigam Sanctuary - West Bengal
done
clear
D)
Bandipur Sanctuary - Karnataka
done
clear
View Answer play_arrow
A taxon facing an extremely high risk of extinction in wild in the immediate future is called
A)
critical endangered
done
clear
B)
endangered
done
clear
C)
vulnerable
done
clear
D)
extinct in wild
done
clear
View Answer play_arrow
Compensation point is
A)
where there is neither photosynthesis nor respiration.
done
clear
B)
when rate of photosynthesis is equal to the rate of respiration.
done
clear
C)
when entire food synthesized into photosynthesis remain utilized.
done
clear
D)
when there is enough water just to meet the requirements of plant.
done
clear
View Answer play_arrow
Which of the following hormones does not naturally occur in plants?
A)
2, 4-D (2, 4-dichloropheoxy acetic acid)
done
clear
B)
IAA
done
clear
C)
ABA
done
clear
D)
GA
done
clear
View Answer play_arrow
Hydroponics or soilless culture helps is knowing
A)
essentiality of an element
done
clear
B)
deficiency symptoms caused by an element
done
clear
C)
toxicity caused by an element
done
clear
D)
all of these
done
clear
View Answer play_arrow
A trace element essential for plant growth and radioisotope, which is used in cancer therapy is
A)
cobalt
done
clear
B)
calcium
done
clear
C)
sodium
done
clear
D)
iron
done
clear
View Answer play_arrow
Choose odd w.r.t. collenchyma.
A)
Found in hypodermis of dicot stem
done
clear
B)
Living mechanical tissue
done
clear
C)
Absent in monocots
done
clear
D)
Thickening of wall is due to deposition of cellulose and lignin
done
clear
View Answer play_arrow
Water will be absorbed by root hairs when
A)
concentration of salt in the soil is high.
done
clear
B)
concentration of solutes in the cell sap is high.
done
clear
C)
plant is rapidly respiring.
done
clear
D)
they are separated from soil by a permeable membrane.
done
clear
View Answer play_arrow
Which of the following types of phytohormones resemble the nucleic acids in some structural aspects?
A)
Auxin
done
clear
B)
Cytokinin
done
clear
C)
Gibberellin
done
clear
D)
ABA
done
clear
View Answer play_arrow
Which of the following statement(s) is/are incorrect?
(i) Proton channel of oxysome / complex V / ATP synthase is located in \[{{F}_{1}}.\] (ii) Metabolic water is produced in terminal oxidation / produced in respiration. (iii) CoQ accepts electron from NADH dehydrogenase (complex I) and also can accept electron from \[FAD{{H}_{2}}/\] succinate Q-reductase / complex II. (iv) Cytochrome c is a small protein attached to outer surface of the inner mitochondrial membrane and acts as mobile carrier for transfer of electrons between complex I (Cyt be, complex) and III. (v) Complex IV refers to cytochrome c oxidase (cyt a, \[{{a}_{3}}\] and 2 Cu per centre). (vi) If a cell is treated with a drug that inhibits ATP synthase, the pH of mitochondrial matrix will increase.
A)
(i), (ii) and (iii)
done
clear
B)
(iii), (v) and (vi)
done
clear
C)
(i) and (iv)
done
clear
D)
Only (iii)
done
clear
View Answer play_arrow
Which one of following feature is possessed by Crustaceans and not by insects?
A)
Paired limbs
done
clear
B)
Two pairs of antenna
done
clear
C)
Chitinous exoskeleton
done
clear
D)
Bilateral symmetry
done
clear
View Answer play_arrow
Which one is not a vestigial organ?
A)
Wings of kiwi
done
clear
B)
Coccyx in man
done
clear
C)
Pelvic girdle of python
done
clear
D)
Flipper of seal
done
clear
View Answer play_arrow
During inspiration, the diaphragm
A)
relaxes to become dome-shaped
done
clear
B)
contracts and flattens
done
clear
C)
expands
done
clear
D)
shows no change
done
clear
View Answer play_arrow
Cirrhosis of liver is caused by the chronic intake of
A)
Opium
done
clear
B)
Alcohol
done
clear
C)
Tobacco (Chewing)
done
clear
D)
Cocaine
done
clear
View Answer play_arrow
Which of the following sets contains polysaccharides?
A)
Glucose, fructose, lactose
done
clear
B)
Starch, glycogen, cellulose
done
clear
C)
Sucrose, maltose, cellulose
done
clear
D)
Galactose, starch, sucrose
done
clear
View Answer play_arrow
Which one of the following combinations of organisms are responsible for the formation and flavour of yoghurt?
A)
Lactobacillus bulgaricus and Streptococcus thermophiles
done
clear
B)
Rhizobium meliloti and Azotobacter
done
clear
C)
Bacillus subtilis and Escherichia coli
done
clear
D)
Bacillus megathermus and Xanthomonas species
done
clear
View Answer play_arrow
Baculoviruses are excellent candidates for
A)
species-specific narrow spectrum pesticidal applications.
done
clear
B)
species-specific broad spectrum pesticidal applications.
done
clear
C)
species-specific narrow spectrum insecticidal applications.
done
clear
D)
species-specific broad spectrum insecticidal applications.
done
clear
View Answer play_arrow
Select the correct statement.
A)
Genetic engineering works only on animals and has not yet been successfully used on plants.
done
clear
B)
There are no risks associated with DNA technology.
done
clear
C)
The first step in PCR is heat which is used to separate both the strands of target DNA.
done
clear
D)
DNA from one organism will not bond to DNA from another animal.
done
clear
View Answer play_arrow
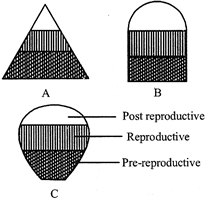
Study the age pyramids given below and select the correct statements regarding these.
A)
A is a triangle shaped age pyramid, where pre-reproductive stage is very large as compared to the reproductive and post reproductive stages of the population. This type of age structure indicates that the population would increase rapidly.
done
clear
B)
B is an inverted bell shaped age pyramid, where number of pre-reproductive and reproductive individuals is almost equal. This type of age structure indicates that the population is stable.
done
clear
C)
C is an urn shaped age pyramid, where more number of older people are present. This type of age structure indicates that the population is declining.
done
clear
D)
All of these
done
clear
View Answer play_arrow
The lactic acid generated during muscle contraction is converted to glycogen in
A)
Muscle
done
clear
B)
Kidney
done
clear
C)
Pancreas
done
clear
D)
Liver
done
clear
View Answer play_arrow
Thromboplastin is secreted by
A)
platelets
done
clear
B)
lymphocytes
done
clear
C)
helper T-cells
done
clear
D)
mast cells
done
clear
View Answer play_arrow
B-DNA which is right-handed double helix contains ______ base pairs per turn of the helix and each turns is ______ long.
A)
\[10,\]\[3.4\,\overset{{}^\circ }{\mathop{A}}\,\]
done
clear
B)
\[10,\]\[34\,\overset{{}^\circ }{\mathop{A}}\,\]
done
clear
C)
\[11,\]\[20\,\overset{{}^\circ }{\mathop{A}}\,\]
done
clear
D)
\[11,\]\[34\,\overset{{}^\circ }{\mathop{A}}\,\]
done
clear
View Answer play_arrow
Match Column-1 with Column-11 and select the correct option from the codes given below.
Column-I Column-II A. Van Kupffer cell (i) Islets of Langerhans B. \[\beta \]-cells (ii) Liver sinusoids C. Oxyntic cells (iii) Thyroid gland D. Paneth cells (iv) Stomach (v) Small intestine
A)
A\[\to \](i); B\[\to \](v); C\[\to \](iii); D\[\to \](iv)
done
clear
B)
A\[\to \](iii); B\[\to \](iv); C\[\to \](ii); D\[\to \](v)
done
clear
C)
A\[\to \](iv); B\[\to \](ii); C\[\to \](i); D\[\to \](iii)
done
clear
D)
A\[\to \](ii); B\[\to \](i); C\[\to \](iv); D\[\to \](v)
done
clear
View Answer play_arrow
Splitting of centromere and hence separation of chromatids occur during
A)
anaphase of mitosis
done
clear
B)
anaphase of meiosis-I
done
clear
C)
anaphase of meiosis-II
done
clear
D)
both [a] and [c]
done
clear
View Answer play_arrow
Which one of the following combination is mismatched?
A)
Glycocalyx - may be capsule or slime layer
done
clear
B)
Pili - Reproduction
done
clear
C)
Cell wall - Protective, determines shape, prevents from bursting
done
clear
D)
Flagella, Pili and Fimbriae - Surface structures of bacterial cell
done
clear
View Answer play_arrow
The most basic amino acid is
A)
Arginine
done
clear
B)
Histidine
done
clear
C)
Glycine
done
clear
D)
Glutamine
done
clear
View Answer play_arrow
Poison glands of snake are modified
A)
Linguals
done
clear
B)
Sublinguals
done
clear
C)
Maxillaries
done
clear
D)
Parotids
done
clear
View Answer play_arrow
Which of the following statements are wrong?
(i) Leucocytes disintegrate in the spleen and liver. (ii) RBC, WBC and blood platelets are produced by bone marrow. (iii) Neutrophils bring about destruction and detoxification of toxins of protein origin. (iv) The important function of lymphocytes is to produce antibodies.
A)
(i) and (ii) only
done
clear
B)
(i) and (iv) only
done
clear
C)
(i) and (iii) only
done
clear
D)
(ii) and (iii) only
done
clear
View Answer play_arrow
Which one single organism or the pair of organisms is correctly assigned to its taxonomic group?
A)
Paramecium and Plasmodium belong to the same kingdom as that of Penicillium
done
clear
B)
Lichen is a composite organism formed from the symbiotic association of an algae and a protozoan
done
clear
C)
Yeast used in making bread and beer is a fungus
done
clear
D)
Nostoc and Anabaena are examples of Protista
done
clear
View Answer play_arrow
Bee dances are meant for
A)
Courtship
done
clear
B)
Communication
done
clear
C)
Recreation
done
clear
D)
Instinct
done
clear
View Answer play_arrow
Which of the following are correct?
A)
Sponges: Cellular level of organization
done
clear
B)
Cnidaria: Tissue level of organization
done
clear
C)
Platyhelminthes: Organ level of organization
done
clear
D)
Annelids, Arthropods, Molluscs, Echinoderms and Chordates: Organ system level of organization.
done
clear
View Answer play_arrow
Which one of the following techniques is safest for the detection of cancers?
A)
Magnetic resonance imaging (MRI)
done
clear
B)
Radiography (X-ray)
done
clear
C)
Computed tomography (CT)
done
clear
D)
Histopathological studies
done
clear
View Answer play_arrow
Select the correct option showing the life cycle of Plasmodium.
A)
Sporozoites (human)\[\to \]RBCs\[\to \]liver cells \[\to \]gametocytes in blood\[\to \]blood meal, bite (female mosquito)\[\to \]multiply (mosquito)\[\to \]sporozoites (mosquito)
done
clear
B)
Sporozoites (human)\[\to \]liver cells\[\to \]RBCs\[\to \] gametocytes in blood\[\to \]blood meal, bite (female mosquito)\[\to \]multiply (mosquito)\[\to \]sporozoites (mosquito)
done
clear
C)
Gametocytes (mosquito)\[\to \]bite\[\to \] gametocytes (human)\[\to \]RBCs\[\to \]multiply\[\to \] sporozoites blood meal (human)\[\to \]bite \[\to \]sporozoites (female mosquito)\[\to \]multiply (mosquito)\[\to \]gametocytes (mosquito)
done
clear
D)
Sporozoites (human)\[\to \]liver cells\[\to \] gametocytes in blood\[\to \]blood meal, bite(female mosquito)\[\to \]multiply (mosquito)\[\to \]sporozoites (mosquito)
done
clear
View Answer play_arrow
Which of the following technique is used for the separation of DNA fragments?
A)
Gel electrophoresis
done
clear
B)
Chromatography
done
clear
C)
Transformation
done
clear
D)
Transduction
done
clear
View Answer play_arrow
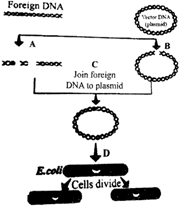
The flow chart given below represents the process of recombinant DNA technology. Identify A, B,C and D.
A)
A-Restriction endonuclease, B-Restriction exonuclease, C- DNA ligase, D- Transformation
done
clear
B)
A-Restriction endonuclease, B-Restriction endonuclease, C-DNA ligase, D-Transformation
done
clear
C)
A-Restriction endonuclease, B-Restriction endonuclease, C-Hydrolase, D-Transformation
done
clear
D)
A-Restriction endonuclease, B-Restriction endonuclease, C-Hydrolase, D-Transduction
done
clear
View Answer play_arrow
Which one of the following statement is correct?
A)
Warm and moist environment favour decomposition whereas low temperature and anaerobiosis inhibit decomposition
done
clear
B)
Warm and moist environment inhibit decomposition whereas low temperature and anaerobiosis favour decomposition
done
clear
C)
Warm and anaerobiosis favour decomposition whereas low temperature favours decomposition
done
clear
D)
Warm and low temperature inhibit decomposition whereas anaerobiosis favours decomposition
done
clear
View Answer play_arrow
Which of the following statement(s) is/are true?
(i) Biowar is the use of biological weapons against humans and /or their crops and animals. (ii) Bioethics is the unauthorized use of bioresources and traditional knowledge related to bioresources for commercial benefits. (iii) Biopatent is exploitation of bioresources of other nations without proper authorisation.
A)
(ii) only
done
clear
B)
(i) only
done
clear
C)
(i) and (ii) only
done
clear
D)
(i) and (iii) only
done
clear
View Answer play_arrow
The species that invade a bare area are called
A)
keystone species
done
clear
B)
extinct species
done
clear
C)
pioneer species
done
clear
D)
rare species
done
clear
View Answer play_arrow
If receptor molecule is removed from target organ for hormone action, the target organ will
A)
continue to respond but require higher concentration of hormone
done
clear
B)
continue to respond but in opposite away
done
clear
C)
continue to respond without any difference
done
clear
D)
not respond to hormone
done
clear
View Answer play_arrow
Osteoporosis is caused by
A)
\[C{{a}^{2+}}\] deficiency
done
clear
B)
Hypersecretion of calcitonin
done
clear
C)
\[{{K}^{+}}\] deficiency
done
clear
D)
Hypersecretion of Parathormone
done
clear
View Answer play_arrow
Which hormone is related to mineral metabolism but is not a peptide / protein in nature?
A)
Parathyroid hormone (PTH)
done
clear
B)
Atrial natriuretic (ANF)
done
clear
C)
Aldosterone
done
clear
D)
All of these
done
clear
View Answer play_arrow
Mark the incorrect statement
A)
The ear ossicle attached to tympanic membrane is malleus
done
clear
B)
Opsin (of Rhodopsin) develops from vitamin A
done
clear
C)
The pressure on ear drum is equalized by eustachian tube
done
clear
D)
Otolith organ consists of saccule and utricle
done
clear
View Answer play_arrow
The contractile protein of skeletal muscle involving ATPase activity is
A)
Myosin
done
clear
B)
Actin
done
clear
C)
Troponin
done
clear
D)
Tropomyosin
done
clear
View Answer play_arrow
Endocrine glands have ____ to carry their secretions to the specific organ.
A)
capillaries
done
clear
B)
tubules
done
clear
C)
no ducts
done
clear
D)
ducts
done
clear
View Answer play_arrow
The functional unit of mammalian kidney is
A)
ureter
done
clear
B)
urinary bladder
done
clear
C)
urethra
done
clear
D)
nephron
done
clear
View Answer play_arrow
The condition of excess urea in blood is known as
A)
Polyuria
done
clear
B)
Haematuria
done
clear
C)
Uraemia
done
clear
D)
Diuresis
done
clear
View Answer play_arrow
Chordae tendinae are found in
A)
joints of legs
done
clear
B)
atria of heart
done
clear
C)
ventricles of brain
done
clear
D)
ventricles of heart
done
clear
View Answer play_arrow
The pattern of contraction and relaxation of the heart is referred to as
A)
blood pressure
done
clear
B)
arterial flow
done
clear
C)
blood flow
done
clear
D)
cardiac cycle
done
clear
View Answer play_arrow
Chemosensitive area of respiratory centre in medulla is affected is
A)
less \[C{{O}_{2}}\] and \[{{H}^{+}}\] ions
done
clear
B)
less \[{{O}_{2}}\] and \[{{H}^{+}}\] ions
done
clear
C)
excess \[C{{O}_{2}}\] and \[{{H}^{+}}\] ions
done
clear
D)
excess \[{{O}_{2}}\] and \[{{H}^{+}}\] ions
done
clear
View Answer play_arrow
Which of the following statements are true?
(i) The blood transports \[C{{O}_{2}}\] comparatively easily because of its higher solubility. (ii) Approximately 8-9% of \[C{{O}_{2}}\] is transported being dissolved in the plasma of blood. (iii) The carbon dioxide produced by the tissues, diffuses passively into the blood stream and passes into red blood corpuscles and react with water to form \[{{H}_{2}}C{{O}_{3}}.\] (iv) The oxyhaemoglobin \[(Hb{{O}_{2}})\] of the erythrocytes is basic. (v) The chloride ions' diffuse from plasma into the erythrocytes to maintain ionic balance.
A)
(i), (iii) and (v) are true, (ii) and (iv) are false
done
clear
B)
(i), (iii) and (v) are false, (ii) and (iv) are true
done
clear
C)
(i), (ii) and (iv) are true, (iii) and (v) are false
done
clear
D)
(i), (ii) and (iv) are false, (iii) and (v) are true
done
clear
View Answer play_arrow
In a mRNA molecule, untranslated regions (UTRs) are present at
A)
5' - end (before start codon)
done
clear
B)
3' - end (after stop codon)
done
clear
C)
both [a] and [b]
done
clear
D)
no UTRs present in Mrna
done
clear
View Answer play_arrow
What is the figure given below showing in particular?
A)
Ovarian cancer
done
clear
B)
Uterine cancer
done
clear
C)
Tubectomy
done
clear
D)
Vasectomy
done
clear
View Answer play_arrow
Identify the human developmental stage shown below as well as the related right place of its occurrence in a normal pregnant woman, and select the right option for the two together.
A)
Develeopmental stage - Late morula Site of occurrence - Middle part of fallopian
done
clear
B)
Develeopmental stage - Blastula Site of occurrence - End part of fallopian tube
done
clear
C)
Develeopmental stage - Blastocyst Site of occurrence - Uterine wall
done
clear
D)
Develeopmental stage - 8 celled morula Site of occurrence - Starting point of fallopian tube
done
clear
View Answer play_arrow
Which one of the following statements is correct with respect to immunity?
A)
Preformed antibodies need to be injected to treat the bite by a viper snake.
done
clear
B)
The antibodies against small pox pathogen are produced by T - lymphocytes.
done
clear
C)
Antibodies are protein molecules, each of which has four light chains.
done
clear
D)
Rejection of a kidney graft is the function of B-lymphocytes.
done
clear
View Answer play_arrow
A)
dominant X-linked
done
clear
B)
recessive X-linked
done
clear
C)
dominant Y-linked
done
clear
D)
recessive Y-linked
done
clear
View Answer play_arrow