Three spheres have radii 7 cm, 10 cm and 12 cm respectively, which sphere exerts maximum pressure on earth?
A)
First
done
clear
B)
Second
done
clear
C)
Third
done
clear
D)
All exert same pressure
done
clear
View Answer play_arrow
If in a wire of Young's modulus Y, longitudinal strain X is produced, then the potential energy stored in its unit volume will be
A)
\[0.5 Y\,\,{{X}^{2}}\]
done
clear
B)
\[0.5 {{Y}^{2}}\,\,X\]
done
clear
C)
\[2Y\,\,{{X}^{2}}\]
done
clear
D)
\[Y\,{{X}^{2}}\]
done
clear
View Answer play_arrow
A spring of force constant k is cut into three equal parts. The force constant of each part would be
A)
3 k
done
clear
B)
2 k
done
clear
C)
k
done
clear
D)
\[\frac{k}{3}\]
done
clear
View Answer play_arrow
If the tension on a wire is removed at once, then
A)
it will break
done
clear
B)
its temperature will reduce
done
clear
C)
its temperature will increase
done
clear
D)
there will be no change in its temperature
done
clear
View Answer play_arrow
Distance between two successive atomic planes of a calcite crystals is 0.3 nm. The minimum angle for Bragg scattering of 0.3 A X-rays will be
A)
\[1.43{}^\circ \]
done
clear
B)
\[1.56{}^\circ \]
done
clear
C)
\[2.86{}^\circ \]
done
clear
D)
\[30{}^\circ \]
done
clear
View Answer play_arrow
If in a vernier calliper, one main scale division is equal to M units and nth division of main scale coincides with \[{{\left( n +1 \right)}^{th}}\] division of vernier scale, then least count of vernier is
A)
\[\frac{nM}{n+1}\]
done
clear
B)
\[\frac{nM}{M+1}\]
done
clear
C)
\[\frac{M}{n+1}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
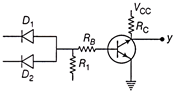
The following circuit diagram represent
A)
AND
done
clear
B)
OR
done
clear
C)
NAND
done
clear
D)
NOT
done
clear
View Answer play_arrow
In an L-R circuit, time constant is that time in which current grows from zero to the value where \[{{I}_{0}}\] is steady state current.
A)
\[0.63\,\,{{I}_{0}}\]
done
clear
B)
\[0.50\,\,{{I}_{0}}\]
done
clear
C)
\[0.37\,\,{{I}_{0}}\]
done
clear
D)
\[{{I}_{0}}\]
done
clear
View Answer play_arrow
An alternating current is given by\[\operatorname{I}={{I}_{1}}\ cos\,\,\omega t\,\,+\,\,{{I}_{2}}\,sin\,\,\omega t\]. The root mean square current is given by
A)
\[\frac{{{\left( {{I}_{1}}+{{I}_{2}} \right)}^{2}}}{2}\]
done
clear
B)
\[\frac{\left( {{I}_{1}}+{{I}_{2}} \right)}{\sqrt{2}}\]
done
clear
C)
\[\sqrt{\frac{\left( I_{1}^{2}+I_{2}^{2} \right)}{2}}\]
done
clear
D)
\[\frac{\sqrt{I_{1}^{2}-I_{2}^{2}}}{2}\]
done
clear
View Answer play_arrow
Three waves A, B and C of frequency 1600 kHz, 5 MHz and 60 MHz, respectively are to be transmitted from one place to another. Which of the following mode is the most appropriate mode of communication?
A)
A is transmitted via space wave while B and C are transmitted via sky wave
done
clear
B)
A is transmitted via ground wave, B via sky wave and C via space wave
done
clear
C)
B and C are transmitted via ground wave while A is transmitted via sky wave
done
clear
D)
B is transmitted via ground wave while A and C are transmitted via space wave
done
clear
View Answer play_arrow
The output is low when either of the input is high, then this represents which of the following gates?
A)
OR
done
clear
B)
NOR
done
clear
C)
AND
done
clear
D)
NAND
done
clear
View Answer play_arrow
An electromagnetic wave passing through vacuum is described by\[\operatorname{E} = {{E}_{0}} sin\,(kx\,-\,\omega t) B= {{B}_{0}} sin\,(kx - \omega t)\]. The correct equation is
A)
\[{{\operatorname{E}}_{0}}\omega \,\,=\,\,{{B}_{0}}k\]
done
clear
B)
\[{{\operatorname{E}}_{0}}k\,\,=\,\,{{B}_{0}}\omega \]
done
clear
C)
\[{{\operatorname{E}}_{0}}{{B}_{0}}\,\,=\,\,\omega k\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
If critical angle of a certain medium is\[si{{n}^{-1}}\,\left( \frac{3}{5} \right)\]. The polarising angle of the medium is
A)
\[{{\tan }^{-1}}\,\left( \frac{3}{4} \right)\]
done
clear
B)
\[{{\tan }^{-1}}\,\left( \frac{4}{3} \right)\]
done
clear
C)
\[{{\tan }^{-1}}\,\left( \frac{5}{3} \right)\]
done
clear
D)
\[{{\sin }^{-1}}\,\left( \frac{4}{5} \right)\]
done
clear
View Answer play_arrow
A copper rod is suspended in a non-homogeneous magnetic field region. The rod when in equilibrium will align itself
A)
in the region where magnetic field is strongest
done
clear
B)
in the region where magnetic field is weakest and parallel to direction of magnetic field there
done
clear
C)
in the direction in which it was originally suspended
done
clear
D)
in the region where magnetic field is weakest and perpendicular to the direction of magnetic field there
done
clear
View Answer play_arrow
The input resistance of a silicon transistor is\[50\,\Omega \]. Base current is changed by\[20\,\mu A\], which results in a change in collector current by 1 mA. This transistor is used as a common emitter amplifier with a load resistance of\[2\,\,k\Omega \]. The voltage gain of amplifier is
A)
4000
done
clear
B)
2000
done
clear
C)
1000
done
clear
D)
None of these
done
clear
View Answer play_arrow
A quantity M is related to three quantities x, y, z as \[M=\frac{{{x}^{1/2}}\,{{y}^{2}}}{{{z}^{1/3}}}\]. If in an experiment the percentage error in measurement of x, y, z are \[2%, \,3% \,and \,3%\] respectively, then error in M is
A)
\[2%\]
done
clear
B)
\[4%\]
done
clear
C)
\[6\,%\]
done
clear
D)
\[8\,%\]
done
clear
View Answer play_arrow
The magnetic flux through a circuit of resistance R changes by an amount \[\Delta \phi \] in a time\[\Delta t\]. Then, the total quantity of electric charge Q that passes at any point in the circuit during the time \[\Delta t\] is represented by
A)
\[Q=\frac{\Delta \phi }{R}\]
done
clear
B)
\[Q=\frac{1}{R}\,\frac{\Delta \phi }{\Delta t}\]
done
clear
C)
\[Q=R\,\frac{\Delta \phi }{\Delta t}\]
done
clear
D)
\[Q\,\,=\,\,\frac{\Delta \phi }{\Delta t}\]
done
clear
View Answer play_arrow
In an equal convex lens having radius of curvature R and focal length f. If \[f\,\,>\,\,R\], the refractive index \[\mu \] of the material of lens is
A)
\[0 < \mu < 1\]
done
clear
B)
\[1 < \mu < 1.5\]
done
clear
C)
\[\mu > 2\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
The value of universal gravitational constant (G) depends upon
A)
mass of the body
done
clear
B)
acceleration due to gravity
done
clear
C)
medium between the two bodies
done
clear
D)
None of these
done
clear
View Answer play_arrow
If a mixture contains two radioactive material \[{{\operatorname{X}}_{1}}\,\,and\,\,{{X}_{2}}\] with half-lives of 20 s and 10 s respectively. If at \[\operatorname{t}=0\] the mixture has 40 g of \[{{\operatorname{X}}_{1}}\] and 160 g of\[{{X}_{2}}\]. The amount of the two in the mixture will become equal after t
A)
80 s
done
clear
B)
40 s
done
clear
C)
60 s
done
clear
D)
20 s
done
clear
View Answer play_arrow
Even Carnot engine cannot give \[100\,%\] efficiency because we cannot
A)
find ideal sources
done
clear
B)
prevent radiation
done
clear
C)
eliminate friction
done
clear
D)
reach absolute zero temperature
done
clear
View Answer play_arrow
In the formula \[\operatorname{x}=3y{{z}^{2}}\] x and z have dimensions of capacitance and magnetic induction respectively. The dimensions of y should be
A)
\[[{{M}^{3}}\,{{L}^{2}}\,{{T}^{-\,4}}\,{{A}^{-\,4}}]\]
done
clear
B)
\[[{{M}^{-\,2}}\,{{L}^{-\,2}}\,{{T}^{3}}\,{{A}^{2}}]\]
done
clear
C)
\[[{{M}^{-\,3}}\,{{L}^{-\,2}}\,{{T}^{4}}\,{{A}^{4}}]\]
done
clear
D)
\[[{{M}^{-\,3}}\,{{L}^{-\,2}}\,{{T}^{2}}\,{{A}^{0}}]\]
done
clear
View Answer play_arrow
If 50 J of work must be done to move electric charge of 4 C from a place where potential is -10 V to another place where potential is V volt, the value of V is
A)
5 V
done
clear
B)
2.5 V
done
clear
C)
1 V
done
clear
D)
0 V
done
clear
View Answer play_arrow
A body of mass m is thrown with a velocity v making an angle \[45{}^\circ \] with horizontal. The magnitude of change in momentum between starting point and at maximum height is (neglect air resistance)
A)
\[\sqrt{2}\,m\,v\]
done
clear
B)
2m v
done
clear
C)
\[\frac{\sqrt{2}\,m\,v}{2}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
Ray optics is valid, when characteristic dimensions are
A)
much smaller than the wavelength of light
done
clear
B)
much larger than the wavelength of light
done
clear
C)
of the order of millimetre
done
clear
D)
of the same order as the wavelength of the light
done
clear
View Answer play_arrow
A metre scale is standing vertically on the earth's surface on one of its ends. It now falls on the earth without slipping. Find the velocity with which the free end of the scale strikes the earth \[(g =\,\,9.8 m/{{s}^{2}})\]
A)
10.8 m/s
done
clear
B)
5.4 m/s
done
clear
C)
2.7 m/s
done
clear
D)
None of these
done
clear
View Answer play_arrow
A 500 pF capacitor is charged by a 200 V supply. It is then disconnected from the supply and is connected to another uncharged 500 pF capacitor. The loss of electrostatic energy in this process is
A)
\[2\,\,\times \,\,1{{0}^{-\,6}}\,V\]
done
clear
B)
\[3\,\,\times \,\,1{{0}^{-\,6}}\,V\]
done
clear
C)
\[5\,\,\times \,\,1{{0}^{-\,6}}\,V\]
done
clear
D)
\[6\,\,\times \,\,1{{0}^{-\,6}}\,V\]
done
clear
View Answer play_arrow
Three bodies having equal masses m are placed at (a, 0), (0, 0) and,\[\left( \frac{a}{2},\,\,\frac{a\sqrt{3}}{2} \right)\]. The coordinates of centre of mass \[{{x}_{CM}}\] and \[{{y}_{CM}}\] are respectively
A)
\[\frac{a\sqrt{3}}{6},\,\,\frac{a}{2}\]
done
clear
B)
\[\frac{a}{2},\,\,\frac{a\sqrt{3}}{6}\]
done
clear
C)
\[\frac{a\sqrt{3}}{2},\,\,\frac{a}{6}\]
done
clear
D)
\[\frac{a}{6},\,\,\frac{a\sqrt{3}}{2}\]
done
clear
View Answer play_arrow
The height from the earth surface at which acceleration due to gravity is decreased by \[2\,%\] is \[\left( radius of the earth =6400 km \right)\]
A)
32 km
done
clear
B)
64 km
done
clear
C)
128 km
done
clear
D)
256 km
done
clear
View Answer play_arrow
A block of 4 kg is resting on a frictionless plane. If it is struck by a jet stream releasing water at the rate of 2 kg/s at a speed of 3 m/s, then the initial acceleration of block is
A)
\[1.5 m/{{s}^{2}}\]
done
clear
B)
\[1.5\,km/{{h}^{2}}\]
done
clear
C)
\[3 m/{{s}^{2}}\]
done
clear
D)
\[3\,\,km/{{h}^{2}}\]
done
clear
View Answer play_arrow
A ball falls freely from rest and the total distance covered by it in last second of its motion is equals to the distance covered by it in the first 3 s of its motion. The ball remains in air for \[\left[ Take\,\,g=10 m/{{s}^{2}} \right]\]
A)
2 s
done
clear
B)
4 s
done
clear
C)
5 s
done
clear
D)
7 s
done
clear
View Answer play_arrow
The primary winding of a transformer has 500 turns. The primary is connected to an AC supply 20 V, 50 Hz. The secondary will have an output of
A)
200 V, 50 Hz
done
clear
B)
400 V, 100 Hz
done
clear
C)
100 V, 50 Hz
done
clear
D)
150 V, 100 Hz
done
clear
View Answer play_arrow
If shunt of \[4\,\,\Omega \] is attached to a galvanometer, the deflection is reduced to \[\frac{1}{5}th\]. If an additional shunt of \[2\,\,\Omega \] is attached. The deflection will be reduce to
A)
\[\frac{1}{10}\,th of initial\]
done
clear
B)
\[\frac{1}{13}\,th of initial\]
done
clear
C)
remain same
done
clear
D)
None of these
done
clear
View Answer play_arrow
For transition from state \[n=3\,\,to\,\,n=1\]in a hydrogen like atom results in ultraviolet radiation, then infrared radiation will be obtained in the transition from
A)
\[4\,\,\to 3\]
done
clear
B)
\[4\,\,\to 2\]
done
clear
C)
\[2\,\,\to 1\]
done
clear
D)
\[3\,\,\to 2\]
done
clear
View Answer play_arrow
A parallel plate air capacitor is charged to a potential difference of V volt. After disconnecting the charging battery the distance between the plates of the capacitor is increased using an insulating handle. As a result potential difference between the plates
A)
decreases
done
clear
B)
increases
done
clear
C)
becomes zero
done
clear
D)
does not change
done
clear
View Answer play_arrow
Which of the following properties regarding diamagnetic substances is wrong?
A)
Diamagnetic materials have a small negative susceptibility
done
clear
B)
Diamagnetic materials do not have permanent magnetic moment
done
clear
C)
Diamagnetism can be explained in terms q\ magnetic moment
done
clear
D)
All are correct
done
clear
View Answer play_arrow
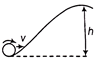
A solid spherical ball is rolling on a frictionless surface as shown in figure with a translational velocity v m/s. To climb the inclined surface, the velocity v should be
A)
2gh
done
clear
B)
\[\frac{10}{7}gh\]
done
clear
C)
\[\ge \,\,\sqrt{2gh}\]
done
clear
D)
\[\ge \,\,\sqrt{\frac{10}{7}gh}\]
done
clear
View Answer play_arrow
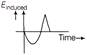
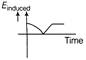
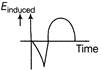
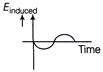
The variation of induced emf with time in a coil, if a short magnet is moved along its axis with a constant velocity (as shown in figure) is best represented by
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
The masses of neutron and proton are 1.0087 and 1.0073 amu respectively. If neutrons and protons combine form helium nucleus of mass 4.0015 amu, the binding energy of the helium nucleus will be
A)
28.4 MeV
done
clear
B)
20.8 MeV
done
clear
C)
27.3 MeV
done
clear
D)
14.2 MeV
done
clear
View Answer play_arrow
A uniform wire when connected directly across a 200 V line produces heat H per second. If the wire is divided into n parts and all parts are connected in parallel across a 200 V line. The heat produced per second will be
A)
H
done
clear
B)
nH
done
clear
C)
\[{{n}^{2}}H\]
done
clear
D)
\[\frac{H}{{{n}^{2}}}\]
done
clear
View Answer play_arrow
In a given Wheatstone's network\[\operatorname{P}=2\,\Omega ,\,\,Q=\,\,2\,\,\Omega ,\,\,R=2\,\,\Omega \,\,and\,\,5=3\,\Omega \], calculate the value of resistance with which S is to be shunted so that bridge get balanced.
A)
2
done
clear
B)
3
done
clear
C)
6
done
clear
D)
9
done
clear
View Answer play_arrow
In a Fraunhofer diffraction experiment at a single slit using light of wavelength 400 nm, the first minimum formed at an angle of 30°. What will be the direction 6 of the first secondary maximum?
A)
\[{{\sin }^{-\,1}}\left( \frac{3}{4} \right)\]
done
clear
B)
\[{{\sin }^{-\,1}}\left( \frac{2}{3} \right)\]
done
clear
C)
\[{{\sin }^{-\,1}}\left( \frac{1}{4} \right)\]
done
clear
D)
\[{{\sin }^{-\,1}}\left( \frac{4}{3} \right)\]
done
clear
View Answer play_arrow
A beaker contains water up to a height \[{{h}_{2}}\] and another non-miscible liquid upto height h 3 above water so that the total height of (water + non-miscible liquid) is \[({{h}_{1}}+{{h}_{2}})\]. Refractive index of water is \[{{\mu }_{1}}\] and that of non-miscible liquid is p, 2. \[{{\mu }_{2}}\]. The apparent shift in the position of bottom of the beaker when viewed from above is
A)
\[\left( 1-\frac{1}{{{\mu }_{1}}} \right){{h}_{2}}+\left( 1-\frac{1}{{{\mu }_{2}}} \right)\,{{h}_{1}}\]
done
clear
B)
\[\left( 1+\frac{1}{{{\mu }_{1}}} \right){{h}_{1}}+\left( 1+\frac{1}{{{\mu }_{2}}} \right)\,{{h}_{2}}\]
done
clear
C)
\[\left( 1-\frac{1}{{{\mu }_{1}}} \right){{h}_{1}}+\left( 1-\frac{1}{{{\mu }_{2}}} \right)\,{{h}_{2}}\]
done
clear
D)
\[\left( 1+\frac{1}{{{\mu }_{1}}} \right){{h}_{2}}+\left( 1+\frac{1}{{{\mu }_{2}}} \right)\,{{h}_{1}}\]
done
clear
View Answer play_arrow
A soap solution has a surface tension of\[2.8 \times 1{{0}^{2}}N/m\]. What will be the work done in blowing a bubble of radius 1 cm.
A)
\[40.2\,\,\times \,\,1{{0}^{-\,6}}\,J\]
done
clear
B)
\[70.3\,\,\times \,\,1{{0}^{-\,6}}\,J\]
done
clear
C)
\[40.2\,\,\times \,\,1{{0}^{-\,5}}\,J\]
done
clear
D)
\[70.3\,\,\times \,\,1{{0}^{-\,5}}\,J\]
done
clear
View Answer play_arrow
The electric field in a region is given by\[\operatorname{E}=x\,\hat{i} +y\hat{j}\,\,+\,\,z\,\hat{k}\]. Here x, y and z are constant. The net flux passing through a square are of side L parallel to y - z plane is
A)
\[{{x}^{2}}L\]
done
clear
B)
\[z{{L}^{2}}\]
done
clear
C)
\[y{{L}^{2}}\]
done
clear
D)
\[x{{L}^{2}}\]
done
clear
View Answer play_arrow
Which of the following is correct regarding following chemical reaction? \[3{{H}_{2}}\,\,+\,\,{{N}_{3}} \,\,2N{{H}_{3}}\]
A)
\[{{k}_{p}}>{{k}_{c}}\]
done
clear
B)
\[{{k}_{p}}<{{k}_{c}}\]
done
clear
C)
\[{{k}_{p}}={{k}_{c}}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
Antiaromaticity is a characteristic of a cyclic molecule with a u electron system that has higher energy due to the presence of 4 n electrons in it. Anti aromatic compounds are highly unstable and highly reactive. Which of the following annulene is anti-aromatic in nature?
A)
Benzene
done
clear
B)
Napthalene
done
clear
C)
Cyclobutadiene
done
clear
D)
Cyclooctatetraene
done
clear
View Answer play_arrow
\[[Cr{{\left( {{H}_{2}}O \right)}_{6}}]C{{l}_{3}}\] has a magnetic moment of 3.83 BM. The correct distribution of 3d electron in the chromium of the complex is
A)
\[3d_{xy}^{1},\,\,3d_{yz}^{1},\,\,3d_{xz}^{1}\]
done
clear
B)
\[3d_{xy}^{1},\,\,3d_{yz}^{1},\,\,3d_{{{z}^{2}}}^{1}\]
done
clear
C)
\[3d_{({{x}^{2}}-{{y}^{2}})}^{1},\,\,3d_{{{z}^{2}}}^{1},\,\,3d_{\,\,xz}^{1}\]
done
clear
D)
\[3d_{xy}^{1},\,\,3d_{({{x}^{2}}-{{y}^{2}})}^{1},\,\,3d_{\,\,yz}^{1}\]
done
clear
View Answer play_arrow
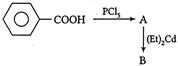
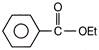
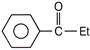
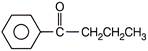
Benzoic acid reacts with\[PC{{l}_{5}}\], gives product A which further reacts with diethyl cadmium and gives B. The product B is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Antipyretic drugs are used to bring down body temperature during high fever and analgesics are used to reduce pain. Which of the following is an antipyretic as well as analgesic?
A)
Aspirin
done
clear
B)
Codeine
done
clear
C)
Phenol
done
clear
D)
Heroin
done
clear
View Answer play_arrow
The Itai-Itai diseases is named for the severe pains caused in the joints and spine. Which of the following causes Itai-Itai diseases?
A)
Cadmium
done
clear
B)
Mercury
done
clear
C)
Lead
done
clear
D)
Calcium
done
clear
View Answer play_arrow
Inverted sugar products tend to retain moisture and are less prone to crystallisation. Therefore, it valued by bakers. Invert sugar is
A)
chemically inactive form of sugar
done
clear
B)
equimolar mixture of glucose and fructose
done
clear
C)
mixture of glucose and sucrose
done
clear
D)
a variety of cane sugar
done
clear
View Answer play_arrow
Ziegler Natta catalyst is used in the synthesis of polymers of a-olefins. It is
A)
\[{{\operatorname{TiCl}}_{4}} + Al {{\left( {{C}_{2}}{{H}_{5}} \right)}_{3}}\]
done
clear
B)
\[{{\operatorname{TiCl}}_{3}} + Al {{\left( {{C}_{2}}{{H}_{5}} \right)}_{3}}\]
done
clear
C)
\[{{\operatorname{TiCl}}_{2}} + Al {{\left( {{C}_{2}}{{H}_{5}} \right)}_{3}}\]
done
clear
D)
\[{{\operatorname{AlCl}}_{3}}+Ti\,{{({{C}_{2}}{{H}_{5}})}_{3}}\]
done
clear
View Answer play_arrow
Cellulose is an important structural component of the primary cell wall of green plants. It is the most abundant polymer on earth. Cellulose is a linear polymer of
A)
\[\alpha -glucose\]
done
clear
B)
\[\beta -glucose\]
done
clear
C)
\[\alpha -fructose\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
The carbylamine reaction is a chemical test for detection of primary amines. Intermediate involved during carbylamine reaction is
A)
carbocation
done
clear
B)
carbanion
done
clear
C)
free radical
done
clear
D)
carbene
done
clear
View Answer play_arrow
Phenols are potentially very reactive towards electrophilic aromatic substitution. The hydroxyl group present in it is strongly activating, ortho/para-directing substituent. Phenol on reaction with \[B{{r}_{2}}\] (excess) produces
A)
\[\operatorname{o} and p- bromophenol\]
done
clear
B)
2, 4, 6 - tribromophenol
done
clear
C)
meta bromophenol
done
clear
D)
None of these
done
clear
View Answer play_arrow
Choose the correct choice regarding product of the following reaction
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Aromatic diazonium group can be replaced by various groups/atoms. In this context, match list I with list II and select the correct answer using the codes given below the lists.
List I List II A. \[{{H}_{3}}P{{O}_{2}}\] 1. Hydrogen B. Sandmeyer reaction 2. Hydroxyl group C. Boiling with water 3. Aryl group D. Gomberg reaction 4. Chlorine
Codes
A)
A-4, B-3, C-2, D-1
done
clear
B)
A-1, B-4, C-2, D-1
done
clear
C)
A-1, B-2, C-3, D-4
done
clear
D)
A-2, B-3, C-4, D-1
done
clear
View Answer play_arrow
Aniline when diazotised in cold and then treated with dimethylaniline gives a coloured compound, the product is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
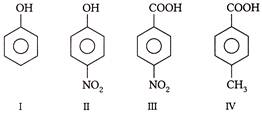
Choose the correct order of acidic strength of the following
A)
\[I<II<IU<IV\]
done
clear
B)
\[I<II <IV<III\]
done
clear
C)
\[I>II>IV<III\]
done
clear
D)
\[I<II>III<IV\]
done
clear
View Answer play_arrow
A flame test is an analytic procedure used in chemistry to detect the presence of certain elements, primarily metal ions, based on each elements characteristic emission spectrum. The colour of flamed in general also depends on temperature. Which of the following does not give flame test?
A)
Sr
done
clear
B)
Be
done
clear
C)
Ra
done
clear
D)
Li
done
clear
View Answer play_arrow
Choose the correct product among four options regarding following reaction
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
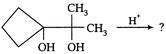
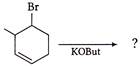
Product obtained on reaction of 3 chloro cyclopentene with \[AgN{{O}_{2}}\] is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
In solid\[N{{H}_{3}}\], each \[N{{H}_{3}}\] molecule had 6 other \[N{{H}_{3}}\] molecules as nearest neighbours. \[\Delta H\] of sublimation of \[N{{H}_{3}}\] at the melting point is 30.8 kJ/mole and the estimated AH of sublimation in the absence of hydrogen bonding is 14.4 kJ/mole. the strength of H-bond in solid \[N{{H}_{3}}\] is
A)
\[5.5 kJ mo{{l}^{-}}^{1}\]
done
clear
B)
\[16.4 kJ mo{{l}^{-}}^{1}\]
done
clear
C)
\[2.7 kJ mo{{l}^{-}}^{1}\]
done
clear
D)
\[-8.7 kJ mo{{l}^{-}}^{1}\]
done
clear
View Answer play_arrow
Boric acid is used in carom boards for smooth gliding of pawns because
A)
\[{{H}_{3}}B{{O}_{3}}\] molecules are loosely chemically bonded and hence it is soft
done
clear
B)
Its low density marked it fluffy
done
clear
C)
can be powdered to a very small grain size
done
clear
D)
\[\operatorname{H}-bodning\] in \[{{H}_{3}}B{{O}_{3}}\] give it a layered structure
done
clear
View Answer play_arrow
Lanthanide contraction is a term used to describe the greater than expected decrease in ionic radii of the elements in the lanthanide series. Which of the following trivalent ion of lanthanide has largest size?
A)
Ti
done
clear
B)
Hf
done
clear
C)
Zr
done
clear
D)
La
done
clear
View Answer play_arrow
Coordination compounds are the complex compounds of d-block elements. The d-block element form coordination compound due to
A)
small - ratio
done
clear
B)
large - ratio
done
clear
C)
small - ratio
done
clear
D)
None of these
done
clear
View Answer play_arrow
A black compound of manganese reacts with a halogen acid to give greenish yellow gas. When excess of this gas reacts with \[N{{H}_{3}}\], an unstable trihalide is formed. In this process the oxidation state of nitrogen changes from
A)
-3 to +3
done
clear
B)
-3 to zero
done
clear
C)
-3 to + 5
done
clear
D)
0 to -3
done
clear
View Answer play_arrow
Many things are known to increase the risk of cancer, including tobacco i.e., dietary factors, certain infections exposure to radiation and environmental pollutants. Which of the following coordination compound is used in treatment of cancer?
A)
\[{{[Pt {{(C{{H}_{3}})}_{2}} {{\left( N{{H}_{3}} \right)}_{6}}]}^{3\,+}}\]
done
clear
B)
\[cis\,[Pt {{(N{{H}_{3}})}_{2}} C{{l}_{2}}]\]
done
clear
C)
\[\left[ Pt {{\left( N{{H}_{3}} \right)}_{3}}Cl \right]\]
done
clear
D)
\[trans\,[Pt\,{{(N{{H}_{3}})}_{2}}\,C{{l}_{2}}]\]
done
clear
View Answer play_arrow
Atomic number of Mn, Fe and Co are 25, 26 and 27 respectively. Which of the following inner orbital octahedral complex ions are diamagnetic? (i) \[{{[CO{{(N{{H}_{3}})}_{6}}]}^{3\,+}}\] (ii) \[[Mn{{\left( CN \right)}_{6}}]{{\,}^{3}}^{-}\] (iii) \[[Fe{{\left( CN \right)}_{6}}]{{\,}^{4\,-}}\] (iv) \[[Fe{{\left( CN \right)}_{6}}]{{\,}^{3\,-}}\]
A)
(i) and (ii) only
done
clear
B)
(ii) and (iii) only
done
clear
C)
(i) and (iii) only
done
clear
D)
All of these
done
clear
View Answer play_arrow
The de-Broglie relations show that the wavelength is inversely proportional to the momention of a paricle. According to de-Broglie hypothesis all microscopic particle has
A)
particle like character
done
clear
B)
wave like character
done
clear
C)
both a and b
done
clear
D)
None of these
done
clear
View Answer play_arrow
Which of the following sets of quantum numbers is not possible?
A)
\[n=4,\,\,l=1,\,\,m=0\,\,\,s=+\frac{1}{2}\]
done
clear
B)
\[n=4,\,\,l=3,\,\,m=-\,3\,\,\,s=-\,\frac{1}{2}\]
done
clear
C)
\[n=4,\,\,l=0,\,\,m=0\,\,\,s=-\,\frac{1}{2}\]
done
clear
D)
\[n=4,\,\,l=1,\,\,m=+\,2\,\,\,s=-\,\frac{1}{2}\]
done
clear
View Answer play_arrow
A flux is chemical leaning agent, flowing agent or purifying agent. They may have more than one function at a time. They are used in both extractive metallurgy and metal joining. Which of the following is an example of acidic flux?
A)
\[Si{{O}_{2}}\]
done
clear
B)
CaO
done
clear
C)
MgO
done
clear
D)
None of these
done
clear
View Answer play_arrow
Which of the following statement is incorrect regarding carbonates and bicarbonates of calcium?
A)
Bicarbonate of calcium causes hardness to water.
done
clear
B)
Bicarbonates of calcium is converted back to insoluble carbonates of calcium.
done
clear
C)
Carbonates of calcium is insoluble in water containing\[C{{O}_{2}}\].
done
clear
D)
Caldum bicarbonates are produced by passing \[C{{O}_{2}}\] through a saturated solution of carbonates.
done
clear
View Answer play_arrow
The ionisation constant of chloroacetic acid is\[135 \times \,{{10}^{-}}^{3}\]. The pH of 0.1 M acid and its 0.1 M sodium salt solution are respectively
A)
1.94, 1.85
done
clear
B)
1.94, 7.94
done
clear
C)
7.94, 1.85
done
clear
D)
1.94, 8.25
done
clear
View Answer play_arrow
Arrange halides of group 13 elements in correct order of lewis acidic strength \[\left[ Hint X : halogen \right]\]
A)
\[{{\operatorname{BX}}_{3}} > AI{{X}_{3}} > Ga{{x}_{3}} > In{{X}_{3}}\]
done
clear
B)
\[B{{X}_{3}}<AI{{X}_{3}}<Ga{{X}_{3}}<In{{X}_{3}}\]
done
clear
C)
\[{{\operatorname{BX}}_{3}} < AI{{X}_{3}} > Ga{{X}_{3}} < In{{X}_{3}}\]
done
clear
D)
\[{{\operatorname{BX}}_{3}}<AI{{X}_{3}}>Go{{X}_{3}}>In{{X}_{3}}\]
done
clear
View Answer play_arrow
2 mol of He is mixed with 2 gm of\[{{H}_{2}}\]. The molar heat capacity at constant pressure for the mixture is
A)
\[\frac{17\,R}{6}\]
done
clear
B)
\[\frac{11\,R}{6}\]
done
clear
C)
4R
done
clear
D)
\[\frac{3\,R}{2}\]
done
clear
View Answer play_arrow
For a redox reaction given below the correct stoichiometric coefficient of \[MnO_{4}^{-},\,\,{{C}_{2}}O_{4}^{2\,-}\,\] and \[{{H}^{+}}\] are respectively. \[\operatorname{MnO}_{4}^{-}+{{C}_{2}}O_{4}^{2\,-}\,+\,\,{{H}^{+}}\,\xrightarrow{{}}\,\,M{{n}^{2}}\,\,+\,\,C{{O}_{2}}+{{H}_{2}}O\]
A)
2, 16, 5
done
clear
B)
2, 5, 16
done
clear
C)
16, 5, 2
done
clear
D)
5, 16, 2
done
clear
View Answer play_arrow
An electrolyte is a compound that ionizes when dissolved in suitable solvents such as water. The electrical conductance of an electrolytic solution in water depends upon
A)
size of its molecules
done
clear
B)
shape of its molecules
done
clear
C)
size of solvent molecules
done
clear
D)
extent of its ionisation
done
clear
View Answer play_arrow
The osmotic pressure of blood is 7.65 atm at \[37{}^\circ \] C. How much glucose should be used per litre for an intravenous injection that is to have the same osmotic pressure as blood?
A)
54.2 g
done
clear
B)
90 g
done
clear
C)
12 g
done
clear
D)
0.30 g
done
clear
View Answer play_arrow
Which of the following oxides behaves as conductor or insulator depending upon temperature?
A)
TiO
done
clear
B)
\[Si{{O}_{2}}\]
done
clear
C)
\[Ti{{O}_{3}}\]
done
clear
D)
MgO
done
clear
View Answer play_arrow
An aromatic compound 'X' with molecular formula \[{{\operatorname{C}}_{9}}{{H}_{10}}O\] gives the following chemical tests. It
I. forms 2, 4-DNP derivative
II. reduces Tollen?s reagent
III. undergoes Cannizarro reaction and
IV. On vigorous oxidation 1, 2-benzenedicarboxylic acid is obtained. X is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
What will be the value of \[\Delta S\] for conversion of liquid to vapour? Given that enthalpy of vapourisation of liquid diethylether and its boiling point are \[26 kJ mol{{\,}^{-}}^{1}\] and \[35\,C{}^\circ \]respectively.
A)
\[+\,84.420 J mo{{l}^{-}}^{1}\]
done
clear
B)
\[+\,84.4 J mo{{l}^{-}}^{1}\,{{K}^{-\,1}}\]
done
clear
C)
\[+\,8.442 J mo{{l}^{-}}^{1}\]
done
clear
D)
\[-\,84.4 J mo{{l}^{-}}^{1}\,{{K}^{-\,1}}\]
done
clear
View Answer play_arrow
Given that \[{{\operatorname{C}}_{diamond}}\,+{{O}_{2}}\,\,\xrightarrow{{}}\,\,C{{O}_{2}},\,\,\Delta H=395.3\,\,kJ/mol\] \[{{\operatorname{C}}_{graphite}}\,+{{O}_{2}}\,\,\xrightarrow{{}}\,\,C{{O}_{2}},\,\,\Delta H=-\,393.4\,\,kJ/mol\] Then for reaction\[{{C}_{graphite}}~\xrightarrow{{}}\,\,{{C}_{diamond}}\] What is the value of \[\Delta H\]?
A)
- 3.8
done
clear
B)
+ 3.8
done
clear
C)
- 1.9
done
clear
D)
+ 1.9
done
clear
View Answer play_arrow
When soap is used for cleaning, it couples with dirt and remove from the clothes. Aggregates of particles is known as
A)
micelles
done
clear
B)
sols
done
clear
C)
gels
done
clear
D)
emulsion
done
clear
View Answer play_arrow
The rate constant, the activation energy and arrhenius parameter of a chemical reaction at \[25{}^\circ C are 3.0 \times 1{{0}^{-}}^{4}{{s}^{-}}^{1}\], \[104.4 kJ mo{{l}^{-}}^{1}\,and 6.0 \times \,\,{{10}^{14}}{{s}^{-}}^{1}\] respectively. The value of rate constant at \[T\to \infty \] is
A)
\[2.0 \times 1{{0}^{18}}{{s}^{-}}^{1}\]
done
clear
B)
\[6.0 \times 1{{0}^{14}}{{s}^{-}}^{1}\]
done
clear
C)
\[\infty \]
done
clear
D)
\[3.6 \times 1{{0}^{30}}{{s}^{-}}^{1}\]
done
clear
View Answer play_arrow
Which of the following option show correct numeric value of osmotic pressure at a given temperature T K. pH of 0.1 M monobasic acid is measured to be 2?
A)
0.1 RT
done
clear
B)
0.11 RT
done
clear
C)
1.1 RT
done
clear
D)
0.01 RT
done
clear
View Answer play_arrow
A compound having molecular formula \[{{A}_{4}}{{O}_{6}}\]. If 10 g of \[{{A}_{4}}{{O}_{6}}\] has 5.72 g A, atomic mass of A is
A)
32 g/mol
done
clear
B)
37 g/mol
done
clear
C)
42 g/mol
done
clear
D)
98 g/mol
done
clear
View Answer play_arrow
3 moles of a perfect gas is expanded isothermally and reversibly from a volume of 1 L to a volume of 10 L at\[27{}^\circ C\]. The maximum work done is
A)
190 L atm
done
clear
B)
170.17 L atm
done
clear
C)
192.25 L atm
done
clear
D)
185.23 L atm
done
clear
View Answer play_arrow
Naturally occurring boron consists of two isotopes whose atomic weights are 10.01 and 11.01. The atomic weight of natural boron is 10.81. The percentage of each isotope in natural boron is
A)
20, 80
done
clear
B)
40, 60
done
clear
C)
60, 40
done
clear
D)
80, 20
done
clear
View Answer play_arrow
Consider the following statements. I. Lichen is a composite organism formed from the symbiotic association of an alga and a protozoan. II. The sporophyte in Riccia is more developed than that in Polytrichum. III. Gymnosperms are also called softwood spermatophytes because they lack xylem fibres, IV. Which of the statements given above are not correct?
A)
I, II and III
done
clear
B)
I and III
done
clear
C)
II and III
done
clear
D)
I and II
done
clear
View Answer play_arrow
Match the following columns.
Column I Column II A. Ichthyostega 1. Connecting link between reptile and mammals B. Seymouria 2. Connecting link between amphibians and reptile C. Lycaerops 3. Connecting link between fishes and amphibians
Codes
A)
A-1, B-2, C-3
done
clear
B)
A-1, B-3, C-2
done
clear
C)
A-3, B-2, C-1
done
clear
D)
A-3, B-1, C-2
done
clear
View Answer play_arrow
Match the following columns.
Column I Column II A. Barbiturates 1. Hallucinogens B. Methandone 2. Opiate C. Marijuana 3. Sedative
Codes
A)
A-1, B-2, C-3
done
clear
B)
A-1, B-3, C-2
done
clear
C)
A-3, B-2, C-1
done
clear
D)
A-3, B-1, C-2
done
clear
View Answer play_arrow
Isotopes popularly known to have been used in the study of photosynthesis are
A)
\[{{P}^{15}}\,and\,\,{{C}^{32}}\]
done
clear
B)
\[{{\operatorname{C}}^{11}}\,and\,\,{{P}^{15}}\]
done
clear
C)
\[{{\operatorname{C}}^{14}}\,and\,\,{{O}^{18}}\]
done
clear
D)
\[{{\operatorname{C}}^{16}}\,and\,\,{{N}^{15}}\]
done
clear
View Answer play_arrow
\[C{{O}_{2}}+{{H}_{2}}O\,\,\,B\,\,\,\,HCO_{3}^{-}+{{H}^{+}}\] Choose the correct option for A, B and C.
A)
A- Carbonic anhydrase B-\[{{H}_{2}}C{{O}_{3}}\], C- Carbonic hydratase
done
clear
B)
A- Carbonic anhydrase, B-\[HC{{O}_{3}}\], C- Carbonic hydratase
done
clear
C)
A- Carbonic anhydrase, B-\[HC{{O}_{3}}\], C- Carbonic hydratase
done
clear
D)
A- Carbonic anhydrase, B-\[{{H}_{2}}C{{O}_{3}}\], C- Carbonic hydratase
done
clear
View Answer play_arrow
Match the following columns.
Column I Column II A. Eardrum 1. Ciliated epithelium B. Vagina 2. Straited squamous C. Urinary bladder 3. Simple squamous D. Trachea 4. Transitional epithelium
A)
A-1, B-3, C-2, D-4
done
clear
B)
A-2, B-1, C-3, D-4
done
clear
C)
A-3, B-2, C-4, D-1
done
clear
D)
A-4, B-1, C-2, D-3
done
clear
View Answer play_arrow
Epiphysis cerebri is also called as... A .... TCT (Thyrocalcitonin) ... .. the calcium level choose the correct option for A and B in blood from the given option.
A)
A- Pituitary gland, B- Increases
done
clear
B)
A- Adrenal gland, B- Increases
done
clear
C)
A- Pineal gland, B- Decreases
done
clear
D)
A- Parathyroid gland, B- Decreases
done
clear
View Answer play_arrow
Phenotypic plasticity is the phenomenon in which
A)
genotypic changes in response to habitat
done
clear
B)
migration temporarily
done
clear
C)
phenotypic changes in response to variations in environment
done
clear
D)
hibernation
done
clear
View Answer play_arrow
A non-sense mutation is done by I. UAA II. UAG III. UGA
A)
I and II
done
clear
B)
II and III
done
clear
C)
I and III
done
clear
D)
I, II and III
done
clear
View Answer play_arrow
Recently RNA interference method has been used in the development of new varieties of plants which is resistant to
A)
viruses
done
clear
B)
bollworms
done
clear
C)
nematodes
done
clear
D)
bacterial blights
done
clear
View Answer play_arrow
Match the following columns.
Column I Column II A. Tornaria 1. Star fish B. Tadpole 2. Jelly fish C. Trochophore 3. Balanoglossus D. Brachiolaria 4. Herdmania 5. Nereis
A)
A-5, B-1, C-4, D-2
done
clear
B)
A-3, B-4, C-5, D-1
done
clear
C)
A-1, B-5, C-4, D-2
done
clear
D)
A-4, B-2, C-1, D-3
done
clear
View Answer play_arrow
Necrosis in crops is due to the deficiency of which ions/elements?
A)
calcium, phosphorus, potassium, zinc
done
clear
B)
sulphur, calcium, potassium, molybdenum
done
clear
C)
nitrogen, potassium, sulphur, molybdenum
done
clear
D)
manganese, sulphur, nitrogen, calcium
done
clear
View Answer play_arrow
Decline in forest is also termed as ... A ... effect. Maintaining a constant internal temperature is termed as ... .... Select the correct option for A and B.
A)
A-Cattle effect, B-Ectoderms
done
clear
B)
A-Waldsterben effect, B-Homeostasis
done
clear
C)
A-Plagiosere, B-Homeostasis
done
clear
D)
A-Jhum, B-Ectoderms
done
clear
View Answer play_arrow
Two species of water lilies in the same pond do not interbreed because one bloom at night and other during day. The reproductive barrier between them is the concept of
A)
biological species
done
clear
B)
morphological species
done
clear
C)
polytypic species
done
clear
D)
evolutionary species
done
clear
View Answer play_arrow
Natural hallucinogen is I. Mescaline II. Psilocybin III. Hemp products Choose the correct option.
A)
I and II
done
clear
B)
II and III
done
clear
C)
I and III
done
clear
D)
I, II and III
done
clear
View Answer play_arrow
Tendency of one cross over to affect other is called
A)
coefficient of coincidence
done
clear
B)
position effect
done
clear
C)
interference
done
clear
D)
cis-trans effect
done
clear
View Answer play_arrow
Which one of the following is true?
A)
Cockroach - 10 pairs of spiracles
done
clear
B)
Earthworm - Setae are present on first clitellum
done
clear
C)
Rat - Right kidney is slightly lower in position than left one
done
clear
D)
Frog - body divisible into neck and trunk
done
clear
View Answer play_arrow
Which of the given option is the protein synthesis inhibitor which acts as analog of tRNA is?
A)
Puromycin
done
clear
B)
Nifampin
done
clear
C)
Streptomycin
done
clear
D)
Tetracycline
done
clear
View Answer play_arrow
Stirred-tank bioreactors (most common type of aerobic bioreactor) have been designed for
A)
availability of oxygen throughout the process
done
clear
B)
ensuring anaerobic conditions in the culture vessel
done
clear
C)
purification of the product
done
clear
D)
addition of preservatives to the product
done
clear
View Answer play_arrow
Assertion [A] Fixed macrophages occur only in the connective tissue. Reason [R] Role of neutrophils is different from that of macrophages.
A)
Both A and R are true and R is the correct explanation of
done
clear
B)
Both A and R are true, but R is not the correct explanation of A
done
clear
C)
A is true, but R is false
done
clear
D)
Both A and R are false
done
clear
View Answer play_arrow
Ambulacral system is also called ... A.... Which is the characteristic feature of phylum ... B ....
A)
blood vascular system Poriefera
done
clear
B)
blood vascular system Arthoploda
done
clear
C)
Water vascular system Pocrfera
done
clear
D)
Water vascular system Echinodermata
done
clear
View Answer play_arrow
The motion colloidal particles is always under the affect of which movement given below.
A)
Tyndall effect
done
clear
B)
cyclosis
done
clear
C)
Brownian movement
done
clear
D)
contractile movement
done
clear
View Answer play_arrow
IUD is stand for Intrauterine devices. They may be following types. I. Medicated II. Fe releasing III. Hormone releasing Choose the correct option.
A)
I and II
done
clear
B)
I and III
done
clear
C)
II and III
done
clear
D)
None of these
done
clear
View Answer play_arrow
Consider the following statements. I. Calvin cycle is known as \[{{\operatorname{C}}_{3}}-cycle\]. II. \[{{\operatorname{C}}_{3}}-plants\] have Kranz type of leaf anatomy. III. \[C{{O}_{2}}\] fixation in \[{{\operatorname{C}}_{3}}-plants\] is done through 3PGA. IV. \[{{C}_{4}}-\,plants\] show more photorespiration as compared to \[{{\operatorname{C}}_{3}}-plants\], resulting in higher production of dry matter. Which of the statements given above are correct?
A)
I, II and III
done
clear
B)
I, III and IV
done
clear
C)
I and III
done
clear
D)
II and IV
done
clear
View Answer play_arrow
Eggs of humans are (on the basis of amount of yolk present)
A)
alecithal
done
clear
B)
mesolecithal
done
clear
C)
microlecithal
done
clear
D)
macrdecithal
done
clear
View Answer play_arrow
RQ (Respiratory Quotient) is the ratio of which gases during the respiration.
A)
\[RQ=\frac{volume\,\,of\,\,C{{O}_{2}}\,\,absorbed}{volume\,\,of\,\,{{O}_{2}}\,\,absorbed}\]
done
clear
B)
\[RQ=\frac{volume\,\,of\,\,C{{O}_{2}}\,\,evolved}{volume\,\,of\,\,{{O}_{2}}\,\,evolved}\]
done
clear
C)
\[RQ=\frac{volume\,\,of\,\,C{{O}_{2}}\,\,absorbed}{volume\,\,of\,\,{{O}_{2}}\,\,evolved}\]
done
clear
D)
\[RQ=\frac{volume\,\,of\,\,C{{O}_{2}}\,\,evolved}{volume\,\,of\,\,{{O}_{2}}\,\,absorbed}\]
done
clear
View Answer play_arrow
Sphagnum is used as a packing material for trans-shipment of living material because
A)
it serves as a disinfectant
done
clear
B)
it reduces transpiration
done
clear
C)
it is easily available
done
clear
D)
it is hygroscopic
done
clear
View Answer play_arrow
The base composition of a bacterial virus is \[A=22\,%\] \[T=22\,%\] \[G=28\,%\] \[C\,\,=\,\,28\,%\]
Its genetic material will be
A)
single-stranded DNA
done
clear
B)
double-stranded RNA
done
clear
C)
double-stranded DNA
done
clear
D)
single-stranded RNA
done
clear
View Answer play_arrow
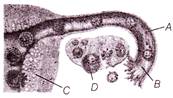
In the given figure identify.
I. Site of fertilisation II. Site of implantation
A)
I - A, II - B
done
clear
B)
I - D, II - C
done
clear
C)
I - A, II - C
done
clear
D)
I - C, II - D
done
clear
View Answer play_arrow
Consider the following statements.
I. ATP was discovered by Lohmann and ATP cycle by Lipmann, who won Nobel Prize in 1953.
II. Acetyle Co-A is a initial compound of Krebs' cycle
III. Krebs? cycle takes in the matrix and ATP formation takes place in cytoplasm.
Which of the statements given above are correct?
A)
I and II
done
clear
B)
I and III
done
clear
C)
II and III
done
clear
D)
I, II and III
done
clear
View Answer play_arrow
Development of land plants, wingless insects and scorpions is an example of which type of evolution from the given below options.
A)
coevolution
done
clear
B)
mimicry
done
clear
C)
quantum evolution
done
clear
D)
salt a tory evolution
done
clear
View Answer play_arrow
Consider the following statements. I. Micro propagation is a method which includes propagation of plants by culturing the cells tissues or organs called tissue culture. II. The culturing of cells or tissues results in the formation of an undifferentiated mass of cells, called callus. Which of the statements given above is/are correct?
A)
Only I
done
clear
B)
Only II
done
clear
C)
I and II
done
clear
D)
None of these
done
clear
View Answer play_arrow
Foramen of magendie is the median aperture of medulla oblongata which in turn is the component of
A)
Diencephalon
done
clear
B)
Mesencephalon
done
clear
C)
Rhomb encephalon
done
clear
D)
Pros encephalon
done
clear
View Answer play_arrow
Name the leucocytes which is the largest in all types of leucocytes and constitute \[2-10%\] of blood leucocytes.
A)
Neutrophyls
done
clear
B)
N-K cells
done
clear
C)
Monocytes
done
clear
D)
Both [a] and [b]
done
clear
View Answer play_arrow
The bond which is responsible for DNA (Deoxyribonucleic acid) strand to be antiparallel is
A)
phosphodiester bonds
done
clear
B)
H-bonds
done
clear
C)
peptide bonds
done
clear
D)
disulphide bonds
done
clear
View Answer play_arrow
Arrange in correct order of biotic succession in pond/lake. I. Woodland Stage II. Plankton Stage III. Climax Forest IV. Floating Stage V. Marsh Meadow Stage Choose the correct option.
A)
\[\operatorname{I}\to II \to III\to IV\to V\]
done
clear
B)
\[I\operatorname{I}\to IV \to V\to I\to III\]
done
clear
C)
\[\operatorname{I}\to V \to IV\to II\to III\]
done
clear
D)
\[II\operatorname{I}\to I \to II\to V\to IV\]
done
clear
View Answer play_arrow
Water is lost in a liquid state in some plants from margins of leaves through hydathodes. These hydathodes
A)
remain open at night and closed during day
done
clear
B)
remain open during day and closed at night
done
clear
C)
remain always open
done
clear
D)
do not show any specificity in opening and closing
done
clear
View Answer play_arrow
Match the following columns.
Column I Column II A. Java ape man 1. \[165{}^\circ \,C\] B. Peking man 2. \[850-1100{}^\circ \,C\] C. Cro-magnon man 3. \[800-100{}^\circ \,C\]
A)
A-1, B-2, C-3
done
clear
B)
A-3, B-2, C-1
done
clear
C)
A-3, B-1, C-2
done
clear
D)
A-1, B-3, C-2
done
clear
View Answer play_arrow
Choose the correct option for A, B, C from the given option. I. Decrease in number of RBC is called ... A .... II. Rise in number of RBC is called ... B .... III. Erythropoietin is released by ... C ....
A)
A-Polycythemia B-Erythrocytopenia C-Erythropoietin
done
clear
B)
A-Erythropoietin B-Erythrocytopenia C-Polycythemia
done
clear
C)
A-Erythropoietin B-Polycythemia C-Erythropoietin
done
clear
D)
A-Erythrocytopenia B-Polycythemia C-Erythropoietin
done
clear
View Answer play_arrow
Formation of erythrocytes is called erythropoiesis. In early few weeks of embryonic life, primitive nucleated RBCs are produced in the
A)
Liver
done
clear
B)
Yolk sac
done
clear
C)
bone marrow
done
clear
D)
Both [b] and [c]
done
clear
View Answer play_arrow
Identify the major group of psychotropic drug I. Tranquillisers II. Stimulants III. LSD IV. Bhang V. Opiate
A)
I, II and III
done
clear
B)
II, III and IV
done
clear
C)
III, IV and V
done
clear
D)
I, II and V
done
clear
View Answer play_arrow
Choose the correct option for A and B
A)
A-Triticum turgidum, B-Secale cereale
done
clear
B)
A-Tetraploid wheat, B-Rye
done
clear
C)
[a] or [b]
done
clear
D)
A-Secale cereal, B-Triticum turgidum
done
clear
View Answer play_arrow
Which of the following is the direct ancestor of Homo sapiens?
A)
Ramapithecus
done
clear
B)
Cro-magnon man
done
clear
C)
Neanderthal man
done
clear
D)
Homo erectus
done
clear
View Answer play_arrow
Which of the following is the longest part in alimentary canal? I. Stomach II. Small intestine III. Large intestine IV. Oesophagus V. Pharynx
A)
I and V
done
clear
B)
Only II
done
clear
C)
III and IV
done
clear
D)
Only III
done
clear
View Answer play_arrow
ZO-ZZ type of sex determination is found in
A)
butterflies
done
clear
B)
reptiles
done
clear
C)
roundworms
done
clear
D)
birds
done
clear
View Answer play_arrow
In-Lac-operon (inducible operon system) the represser protein birds with which part of DNA
A)
Regulator gene
done
clear
B)
Operator gene
done
clear
C)
Promoter gene
done
clear
D)
Structural gene
done
clear
View Answer play_arrow
Which one is correct statement for supplementary genes?
A)
Genes with multiple phenotypic effect
done
clear
B)
Pair of non-allelic dominant genes, which produce a new trait when present in dominant form
done
clear
C)
Pair of non-allelic gene, dominant allel of one of gene is without any independent effect
done
clear
D)
Genes with no effect on appearance of the organism
done
clear
View Answer play_arrow
DNA ligase is used to seal the two strands of DNA. This unique enzyme was firstly discovered from which organism.
A)
E. coli
done
clear
B)
\[\phi \times 176\]
done
clear
C)
A. tumefaceins
done
clear
D)
H. in fuenza
done
clear
View Answer play_arrow
Which of the following phenomenon do not supports Darwin's concept of natural selection?
I. Production of 'Dolly' the sheep by cloning
II. Development of transgenic animals
III. Prevalence of pesticide resistant insects
Choose the correct option.
A)
I and II
done
clear
B)
II and III
done
clear
C)
I, II and III
done
clear
D)
Only III
done
clear
View Answer play_arrow
The first milk cooperative in India was Amul. It was set-up by which scientist
A)
Dr. Panchanan Maheshwari
done
clear
B)
Dr. Ram Dev Mishra
done
clear
C)
Dr. Birbal Sahani
done
clear
D)
Dr. Varghese Kurian
done
clear
View Answer play_arrow
Match the following columns.
Column I Column II A. Paleolithic 1. Bronze age B. Mesolithic 2. Use of clothes and utensils C. Neolithic 3. Iron age D. Present 4. Age of reading writing husbandry 5. Age of tools stone and bone
A)
A-5, B-1, C-2, D-4
done
clear
B)
A-1, B-2, C-4, D-3
done
clear
C)
A-5, B-4, C-1, D-2
done
clear
D)
A-5, B-2, C-4, D-3
done
clear
View Answer play_arrow
Carotene is the hydrocarbons. They imparts colours to plant parts like chillies, tomato. Find out the name of carotene which is found in the chillies and tomato from the given options
A)
\[\beta -carotene\]
done
clear
B)
Lycopene
done
clear
C)
\[\alpha -carotene\]
done
clear
D)
\[\lambda -carotene\]
done
clear
View Answer play_arrow
Match the following columns.
Column I Column II A. 21st March 1. World forest day B. 22nd March 2. Word earth day C. 22nd April 3. World water day D. 5th June 4. World environment day
A)
A-1, B-2, C-3, D-4
done
clear
B)
A-1, B-3, C-2, D-4
done
clear
C)
A-4, B-3, C-2, D-1
done
clear
D)
A-4, B-2, C-3, D-1
done
clear
View Answer play_arrow
Which of the following show convergent evolution?
A)
Rabbit and dog
done
clear
B)
Star fish and jelly fish
done
clear
C)
Fish and whale
done
clear
D)
Bacteria and Amoeba
done
clear
View Answer play_arrow
Separate these according to ex situ and in situ conservation of biodiversity. I. Hot spot II. Biosphere reserve III. National parks IV. Gardens V. Seed banks VI. Aquaria
A)
In situ-IV, V, VI, Ex situ- I, II, III
done
clear
B)
In situ-I, II, III, Ex situ- IV, V, VI
done
clear
C)
In situ-I, IV, V, Ex situ- II, III, V
done
clear
D)
In situ-II, III, VI, Ex situ- I, IV, V
done
clear
View Answer play_arrow
Identify a Mendelian disorder from the following.
A)
Down's syndrome
done
clear
B)
Turner's syndrome
done
clear
C)
Phenylketonuria
done
clear
D)
Klinefelter's syndrome
done
clear
View Answer play_arrow
Pentadiplondra brazzeona, plant from west Africa produces
A)
phosphatases
done
clear
B)
brazzein
done
clear
C)
ligases
done
clear
D)
ribonucleases
done
clear
View Answer play_arrow
Which of the following is called lung of the planet?
A)
Tropical deciduous forest
done
clear
B)
Amazon rainforest
done
clear
C)
Pampas
done
clear
D)
Downs
done
clear
View Answer play_arrow
Golden rice is a transgenic variety of rice (Oryza sativa). Which contain the goods quantity of
A)
vitamin-A
done
clear
B)
\[\beta -carotene\]
done
clear
C)
Both [a] and [b]
done
clear
D)
vitamin-A and D
done
clear
View Answer play_arrow
Nephron is the functional unit of kidney. Podocyte is present in which of nephron
A)
outer wall of Bowman's capsule
done
clear
B)
wall of glomerulus
done
clear
C)
inner wall of Bowman's capsule
done
clear
D)
neck of nepron
done
clear
View Answer play_arrow
Transformation of certain plants (i.e., cotton) contains genes encoding on insecticidal protein. That insecticidal called as protein is obtained from which of the bacteria given below.
A)
B. thuringiensis
done
clear
B)
Anabaena
done
clear
C)
P. fluorescence
done
clear
D)
Nostoc
done
clear
View Answer play_arrow
A hereditary disease, which is never passed of from father to son is
A)
autosomal linked disease
done
clear
B)
X-chromosomal linked disease
done
clear
C)
Y-chromosomal linked disease
done
clear
D)
None of these
done
clear
View Answer play_arrow
In parallel evolution generally convergent evolution is seen. Which of the following is an example of parallel evolution?
A)
Insects wings and wings of bird
done
clear
B)
Darwin's finches
done
clear
C)
Pouched mammals
done
clear
D)
Tasmanian wolf
done
clear
View Answer play_arrow
Which of the following is a root less aquatic plant in which a portion of the leaf form a sac like structure for trapping insects?
A)
Nepenthes
done
clear
B)
Utricularia
done
clear
C)
Cephaleuros
done
clear
D)
Drosophyllum
done
clear
View Answer play_arrow
Milk is converted to the casein by the action of which hormone given below.
A)
rennin
done
clear
B)
amylase
done
clear
C)
intestinal bacteria
done
clear
D)
inverses
done
clear
View Answer play_arrow
The main aim of Earth Summit of Rio de Janeiro (1992) was to
A)
compilation of red list
done
clear
B)
establishment of biosphere reserve
done
clear
C)
conservation on biodiversity
done
clear
D)
conservation of tiger and stags
done
clear
View Answer play_arrow
Which of the fungi is recently being sequenced for making of ligin, so that can be used in paper industry is
A)
E. coli
done
clear
B)
K. plontiola
done
clear
C)
P. chrysoporium
done
clear
D)
CAMV 195
done
clear
View Answer play_arrow
Match the following columns.
Column I Column II A. Eurythermal organisms 1. Body temperature constant B. Stenothermal organisms 2. Wide range of temperature change C. Endothermic organisms 3. Body temperature varies D. Poikilothermal organisms 4. Narrow temperature change
A)
A-2, B-4, C-1, D-3
done
clear
B)
A-1, B-2, C-3, D-4
done
clear
C)
A-4, B-3, C-2, D-1
done
clear
D)
A-2, B-1, C-4, D-3
done
clear
View Answer play_arrow
Which one of the statements gives below is correct for nitrogen-fixing and legumes?
A)
Legumes are incapable of fixing nitrogen
done
clear
B)
Legumes fix-nitrogen only by bacteria present in root nodules
done
clear
C)
Legumes fix-nitrogen only by bacteria present in soil
done
clear
D)
Legumes fix-nitrogen through bacteria living in fruits
done
clear
View Answer play_arrow
Which of the following plants material is widely used in the preparation of culture medium?
A)
Pinus seeds
done
clear
B)
Coconut fruit milk
done
clear
C)
Borassus leaves
done
clear
D)
Cycas starch
done
clear
View Answer play_arrow
In short day plants the flowering is induced by which of the condition given below.
A)
short day and interrupted long night
done
clear
B)
short night
done
clear
C)
long day and interrupted night
done
clear
D)
None of these
done
clear
View Answer play_arrow
Choose the correct statements. I. Ichthyophis is a member of Amphibia. II. Limbless amphibians belong to the order-Anura. III. Amniotic eggs is characteristic feature of amphibians. IV. Amphibians do not have epidermal scales.
A)
I, II and III
done
clear
B)
II, III and IV
done
clear
C)
I, III and IV
done
clear
D)
All of these
done
clear
View Answer play_arrow
Which among the following strand gives palindromic complementary strand?
A)
\[5'\,AGLTTG\,\,3'\]
done
clear
B)
\[5'\,GAATTC\,\,3'\]
done
clear
C)
\[5'\,CCGAAT\,\,3'\]
done
clear
D)
\[5'\,ATATTC\,\,3'\]
done
clear
View Answer play_arrow
Polydactyl in man is a condition in which more than 10 fingers are present is caused due to which type of gene?
A)
Autosomal dominant gene
done
clear
B)
Autosomal recessive gene
done
clear
C)
Sex-linked dominant gene
done
clear
D)
Sex-linked recessive gene
done
clear
View Answer play_arrow
Which of the following statements is not correct?
A)
Cladode is develops by the modification of stem branches of limited growth and is photosynthetic
done
clear
B)
The modified stem of Opuntia is phylloclade
done
clear
C)
Corm is a modification of stem because it bears node and internodes
done
clear
D)
Sucker is an underground modification of stem
done
clear
View Answer play_arrow
Scales, found on the skin of cartilaginous fishes are
A)
glenoid
done
clear
B)
cycloid
done
clear
C)
plocoid
done
clear
D)
ctenoid
done
clear
View Answer play_arrow
HMP (Hexose monophosphate) shunt is an alternative to which of the anaerobic pathway given below
A)
Kreb's cycle
done
clear
B)
Calvin cycle
done
clear
C)
Glycolysis
done
clear
D)
\[{{\operatorname{C}}_{4}}-pathway\]
done
clear
View Answer play_arrow
A person met with an accident and there is no time to check his blood group, which of the following must be given to him?
A)
\[O,\,\,R{{h}^{+}}\]
done
clear
B)
\[O,\,\,R{{h}^{-}}\]
done
clear
C)
\[\operatorname{AB}, R{{h}^{-}}\]
done
clear
D)
\[\operatorname{AB}, R{{h}^{-}}\]
done
clear
View Answer play_arrow
Which of the following is caused by a virus? I. Dengue II. Rabies III. Chikungunya IV. Malaria Choose the correct option
A)
I, II, IV
done
clear
B)
I, III. IV
done
clear
C)
II, III, IV
done
clear
D)
I, IV, III
done
clear
View Answer play_arrow
Pentose phosphate pathway, an alternative pathway of respiration was elucidated by
A)
Hooker and Dickens
done
clear
B)
Warburg and Dickens
done
clear
C)
Blackman and Warburg
done
clear
D)
Kelvin and Blackman
done
clear
View Answer play_arrow
\[\operatorname{Vitamin}-{{D}_{3}}\] is essential for the \[C{{a}^{2+}}\] absorption. Active form of \[vitamin-{{D}_{3}}\] is also called as
A)
calciferol
done
clear
B)
cholecalciferol
done
clear
C)
calcitriol
done
clear
D)
calcidiol
done
clear
View Answer play_arrow
Respiratory centre is the centre which controls the rhythm of respiration located in which part of brain.
A)
pons violin
done
clear
B)
medulla oblongata
done
clear
C)
cerebellum
done
clear
D)
corpora quadrigemina
done
clear
View Answer play_arrow
Which of the following are examples of anabolic reactions? I. Formation of starch from glucose II. Release of \[C{{O}_{2}}\] from body III. Formation of lipids IV. Synthesis of organic compounds
A)
I, II, III
done
clear
B)
I, III, IV
done
clear
C)
II, III, IV
done
clear
D)
II, I, IV
done
clear
View Answer play_arrow
Hypodermis in the stem of Cucurbita is made up of angular collenchyma. The vascular bundles are conjoint open and bi collateral, that means
A)
phloem is present towards outer side of xylem
done
clear
B)
phloem is present towards inner side of xylem
done
clear
C)
phloem is present-on both side of xylem
done
clear
D)
phloem is present in small amount
done
clear
View Answer play_arrow
Wings of bat mosquito and pigeon exhibit, which type of evolution?
A)
Atavism
done
clear
B)
Vestigial
done
clear
C)
Divergent
done
clear
D)
Convergent
done
clear
View Answer play_arrow
Calcium and phosphate are released from the bone into blood under the influence of hormones.
A)
Thyronine
done
clear
B)
Parathormone
done
clear
C)
Calcitonin
done
clear
D)
Both [a] and [c]
done
clear
View Answer play_arrow
Which of the following statements (s) is/are correct? I. In animals, cutaneous gland secrete wax lanolin II. Cerumen or ear wax is secreted by cutaneous grands. III. Bees build their hives from wax secreted by cutaneous glands. IV. Paraffin wax is obtained from petroleum.
A)
I and II
done
clear
B)
III and IV
done
clear
C)
All except III
done
clear
D)
None of these
done
clear
View Answer play_arrow
Assertion [A] In collateral vascular bundle phloem is situated towards outer side and xylem towards inner side. Reason [R] In monocot stem cambium is present.
A)
Both A and R are true and R is the correct explanation of A
done
clear
B)
Both A and R are true, but R is not the correct explanation of A
done
clear
C)
A is true, but R is false
done
clear
D)
A is false, but R is true
done
clear
View Answer play_arrow
Immediate product of transcription in eukaryotes is
A)
hnRNA
done
clear
B)
mRNA
done
clear
C)
SnRNA
done
clear
D)
cDNA
done
clear
View Answer play_arrow
Which one of the following is not correct?
A)
Cobalt chloride paper is used to compare rate of transpiration on two surface of leaf
done
clear
B)
Poro meter is used for measuring the size of stomata
done
clear
C)
Meaningful girdling experiments cannot be done on sugarcane because vascular bundles are scattered
done
clear
D)
Sunken stomata and hairy surface increase transpiration
done
clear
View Answer play_arrow
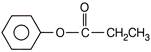
 I. Site of fertilisation II. Site of implantation
I. Site of fertilisation II. Site of implantation 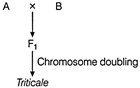 Choose the correct option for A and B
Choose the correct option for A and B 