Given that\[\vec{A}+\vec{B}+\vec{C}=0\]. Out of three vectors two are equal in magnitude and the magnitude of third vector is 2 times that of either of the two having equal magnitude. Then the angles between vectors are given by
A)
\[30{}^\circ ,\text{ }60{}^\circ ,\text{ }90{}^\circ \]
done
clear
B)
\[45{}^\circ ,\text{ }45{}^\circ ,\text{ }90{}^\circ \]
done
clear
C)
\[45{}^\circ ,\text{ }60{}^\circ ,\text{ }90{}^\circ \]
done
clear
D)
\[90{}^\circ ,\text{ }135{}^\circ ,\text{ }135{}^\circ \]
done
clear
View Answer play_arrow
E, m, I and G denote energy, mass, angular momentum and gravitational constant respectively, then the dimension of \[\frac{E{{l}^{2}}}{{{m}^{5}}{{G}^{2}}}\] are
A)
angle
done
clear
B)
length
done
clear
C)
mass
done
clear
D)
time
done
clear
View Answer play_arrow
A player kicks a football of mass 0.5 kg and the football begins to move with a velocity of 10 m/sec. If the contact between the leg and the football lasts for 1/50 sec, then the force acted on the football should be
A)
2500 N
done
clear
B)
1250 N
done
clear
C)
250 N
done
clear
D)
625 N
done
clear
View Answer play_arrow
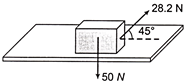
A body of weight 50 N placed on a horizontal surface is just moved by a force of 28.2 N. The frictional force and the normal reaction are
A)
10 N, 15 N
done
clear
B)
20 N, 30 N
done
clear
C)
2 N, 3 N
done
clear
D)
5 N, 6 N
done
clear
View Answer play_arrow
A bullet of mass m moving with velocity v strikes a suspended wooden block of mass M. If the block rises to a height h, the initial velocity of the block will be
A)
\[\sqrt{2gh}\]
done
clear
B)
\[\frac{M+m}{m}\,\sqrt{2gh}\]
done
clear
C)
\[\frac{m}{M+m}\,2gh\]
done
clear
D)
\[\frac{M+m}{M}\,\sqrt{2gh}\]
done
clear
View Answer play_arrow
If the value of g at the surface of the earth is \[9.8\text{ }m/se{{c}^{2}}\], then the value of g at a place 480 km above the surface of the earth will be (Radius of the earth is 6400 km)
A)
\[8.4\text{ }m/se{{c}^{2}}\]
done
clear
B)
\[9.8\text{ }m/se{{c}^{2}}\]
done
clear
C)
\[7.2\text{ }m/se{{c}^{2}}\]
done
clear
D)
\[4.2\text{ }m/se{{c}^{2}}\]
done
clear
View Answer play_arrow
If the work done in blowing a bubble of volume V is W, then the work done in blowing the bubble of volume 2 V from the same soap solution will be
A)
\[W/2\]
done
clear
B)
\[\sqrt{2}\,W\]
done
clear
C)
\[^{3}\sqrt{2}\,W\]
done
clear
D)
\[^{3}\sqrt{4}\,W\]
done
clear
View Answer play_arrow
A wooden block of volume \[1000\text{ }c{{m}^{3}}\] is suspended from a spring balance. It weighs 12 N in air. It is suspended in water such that half of the block is below the surface of water. The reading of the spring balance is
A)
10 N
done
clear
B)
9 N
done
clear
C)
8 N
done
clear
D)
7 N
done
clear
View Answer play_arrow
A certain mass of gas at 273 K is expanded to 81 times its volume under adiabatic condition. If \[\gamma \,\,=\,\,1.25\,\] for the gas, then its final temperature is
A)
\[-235{}^\circ \,C\]
done
clear
B)
\[-182{}^\circ \,C\]
done
clear
C)
\[-91{}^\circ \,C\]
done
clear
D)
\[0{}^\circ \,C\]
done
clear
View Answer play_arrow
An object of mass 0.2 kg executes simple; harmonic along the X-axis with frequency of\[25/\pi \,\,Hz\]. At the position \[x=\text{ }0.04\text{ }m\], the object has kinetic energy of 0.5 J and potential energy of 0.4 J amplitude of oscillation in meter is equal to
A)
0.05
done
clear
B)
0.06
done
clear
C)
0.01
done
clear
D)
none of these
done
clear
View Answer play_arrow
A stretched wire of length 110 cm is divided into three segments whose frequencies are in ratio\[1:2:3\]. Their lengths must be
A)
20 cm; 30 cm; 60 cm
done
clear
B)
60 cm; 30 cm; 20 cm
done
clear
C)
60 cm; 20 cm; 30 cm
done
clear
D)
30 cm; 60 cm; 20 cm
done
clear
View Answer play_arrow
Two point charges \[-\,q\,\,and\,\,+q/2\] are situated at the origin and at the point (a, 0, 0) respectively. The point along the X-axis where the electric field vanishes is
A)
\[x=\frac{a}{\sqrt{2}}\]
done
clear
B)
\[x=\sqrt{2}a\]
done
clear
C)
\[x=\,\,\frac{\sqrt{2}a}{\sqrt{2}-1}\]
done
clear
D)
\[x=\,\,\frac{\sqrt{2}a}{\sqrt{2}+1}\]
done
clear
View Answer play_arrow
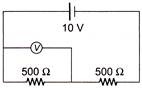
A voltmeter of resistance \[1000\,\,\Omega \] is connected across a resistance of \[500\,\,\Omega \] in the given circuit. What will be the reading of voltmeter
A)
1 V
done
clear
B)
2 V
done
clear
C)
6 V
done
clear
D)
4 V
done
clear
View Answer play_arrow
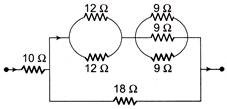
In the following circuit,\[18\,\,\Omega \] resistor develops, 2 J/sec due to current flowing through it. The power developed across \[10\,\,\Omega \], resistance is
A)
125 W
done
clear
B)
10 W
done
clear
C)
\[\frac{4}{5}\,\,W\]
done
clear
D)
25 W
done
clear
View Answer play_arrow
A moving coil galvanometer has 48 turns and area of coil is\[4\,\,\times \,\,{{10}^{-\,2}}{{m}^{2}}\]. If the magnetic field is 0.2 T, then to increase the current sensitivity by \[25\,%\] without changing area and field the number of turns should become
A)
24
done
clear
B)
36
done
clear
C)
60
done
clear
D)
54
done
clear
View Answer play_arrow
A magnet is parallel to a uniform magnetic field. If it is rotated by\[60{}^\circ \], the work done is 0.8 J. How much work is done in moving it \[30{}^\circ \] further
A)
\[0.8\times {{10}^{7}}\,ergs\]
done
clear
B)
0.4 J
done
clear
C)
8 J
done
clear
D)
0.8 ergs
done
clear
View Answer play_arrow
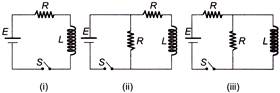
In which of the following circuit is the current maximum just after the switch S is closed?
A)
(i)
done
clear
B)
(ii)
done
clear
C)
(iii)
done
clear
D)
Both (ii) and (iii)
done
clear
View Answer play_arrow
The self-inductance of a choke coil is 10 mH. When it is connected with a 10 V dc source, then the loss of power is 20 watt. When it is connected with 10 volt ac source, loss of power is 10 watt. The frequency of ac source will be
A)
50 Hz
done
clear
B)
60 Hz
done
clear
C)
80 Hz
done
clear
D)
100 Hz
done
clear
View Answer play_arrow
If a voltage applied to an X-ray tube is increased to 1.5 times the minimum wavelength \[({{\lambda }_{min}})\] of an X-ray continuous spectrum shifts by\[\Delta \,\lambda = 26 pm\]. The initial voltage applied to the tube is
A)
\[\approx \,\,10\text{ }kV\]
done
clear
B)
\[\approx \,\,16\text{ }kV\]
done
clear
C)
\[\approx \,\,50\text{ }kV\]
done
clear
D)
\[\approx \,\,75\text{ }kV\]
done
clear
View Answer play_arrow
How much work must be done to pull apart the electron and the proton that make up the Hydrogen atom, if the atom is initially in the state with\[\operatorname{n}= 2\].
A)
\[13.6\times 1.6\times {{10}^{-19}}\,J\]
done
clear
B)
\[3.4\times 1.6\times {{10}^{-19}}\,J\]
done
clear
C)
\[1.51\times 1.6\times {{10}^{-19}}\,J\]
done
clear
D)
0
done
clear
View Answer play_arrow
In space charge limited region, the plate current in a diode is 10 mA for plate voltage 150 V. If the plate voltage is increased to 600 V, then the plate current will be
A)
10 mA
done
clear
B)
40 Ma
done
clear
C)
80 mA
done
clear
D)
160 mA
done
clear
View Answer play_arrow
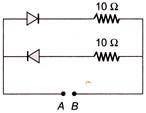
A 2 V battery is connected across the points A and B as shown in the figure given below. Assuming that the resistance of each diode is zero in forward bias and infinity in reverse bias, the current supplied by the battery when its positive terminal is connected to A is
A)
0.2 A
done
clear
B)
0.4 A
done
clear
C)
zero
done
clear
D)
0.1 A
done
clear
View Answer play_arrow
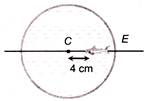
In a thin spherical fish bowl of radius 10 cm filled with water of refractive index \[4/3\] there is a small fish at a distance of 4 cm from the centre C as shown in figure. Where will the image of fish appears, if seen from E?
A)
5.2 cm
done
clear
B)
7.2 cm
done
clear
C)
4.2 cm
done
clear
D)
3.2 cm
done
clear
View Answer play_arrow
The intensity ratio of two coherent sources of light is p. They are interfering in some region and produce interference pattern. Then the fringe visibility is
A)
\[\frac{1+p}{2\sqrt{p}}\]
done
clear
B)
\[\frac{2\sqrt{p}}{1+p}\]
done
clear
C)
\[\frac{p}{1+p}\]
done
clear
D)
\[\frac{2p}{1+p}\]
done
clear
View Answer play_arrow
Given a uniform disc of mass M and radius R. A small disc of radius \[R/2\] is cut from this disc in such a way that the distance between the centres of the two discs is\[R/2\]. Find the moment of inertia of the remaining disc about a diameter of the original disc perpendicular to the line connecting the centres of the two discs
A)
\[\frac{3M{{R}^{2}}}{32}\]
done
clear
B)
\[\frac{5M{{R}^{2}}}{16}\]
done
clear
C)
\[\frac{11M{{R}^{2}}}{64}\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
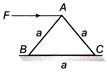
A horizontal force F is applied at the top of an equilateral triangular block having mass m. The minimum coefficient of friction required to topple the block before translation will be
A)
\[\frac{2}{\sqrt{3}}\]
done
clear
B)
\[\frac{1}{3}\]
done
clear
C)
\[\frac{1}{\sqrt{3}}\]
done
clear
D)
\[\frac{1}{2}\]
done
clear
View Answer play_arrow
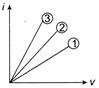
If three wires of same material but different dimension were used in place of unknown resistance, we get these I-V curve
Match the column according to correct curve:
Wire dimension Corresponding curve (p) \[\lambda =1\,\,m\] \[radius=1\,mm\] (i) Curve (1) (q) \[\lambda =1\,\,m\] \[radius=2\,mm\] (ii) Curve (2) (r) \[\lambda =\frac{1}{2}\,m\] \[radius=\frac{1}{2}\,mm\] (iii) Curve (3)
A)
(p)-(ii); (q)-(iii); (r)-(i)
done
clear
B)
(p)-(iii); (q)-(ii); (r)-(i)
done
clear
C)
(p)-(i); (q)-(ii); (r)-(iii)
done
clear
D)
(p)-(iii); (q)-(ii); (r)-(iii)
done
clear
View Answer play_arrow
A newly prepared radioactive nuclide has a decay constant\[\lambda \,of\,\,{{10}^{-}}^{6}\,{{s}^{-1}}\]. What is the approximate half-life of the nuclide?
A)
1 hour
done
clear
B)
1 day
done
clear
C)
1 week
done
clear
D)
1 month
done
clear
View Answer play_arrow
The speed of a motor launch with respect to water in a stream is 8 m/s, while water current?s speed is 3 m/s. When the launch began travelling upstream, a float was dropped from it. After travelling a distance of 4.8 km upstream, the launch turned back and caught up with the float. What is the total time which elapsed during the process?
A)
32 min
done
clear
B)
24 min
done
clear
C)
16 min
done
clear
D)
8 min
done
clear
View Answer play_arrow
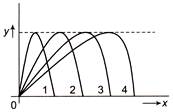
Figure shows four paths for a kicked football. Ignoring the effects of air on the flight, rank the paths according to initial horizontal velocity component, highest first
A)
1, 2, 3, 4
done
clear
B)
2, 3, 4, 1
done
clear
C)
3, 4, 1, 2
done
clear
D)
4, 3, 2, 1
done
clear
View Answer play_arrow
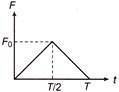
A particle of mass m moving with velocity u makes an elastic one dimensional collision with a stationary particle of mass m. They are in contact for a very short time T. Their force of interaction increases from zero to \[{{F}_{0}}\] linearly of time\[T/2\], and decreases linearly to zero in further time\[T/2\]. The magnitude of \[{{F}_{0}}\] is
A)
\[mu/T~\]
done
clear
B)
\[2mu/T\]
done
clear
C)
\[mu/T\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
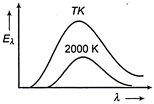
The adjoining diagram shows the spectral energy density distribution \[{{E}_{\lambda }}\] of a black body at two different temperatures. If the areas under the curves are in the ratio \[16 : 1\], the value of temperature T is
A)
32,000 K
done
clear
B)
16,000 K
done
clear
C)
8,000 K
done
clear
D)
4,000 K
done
clear
View Answer play_arrow
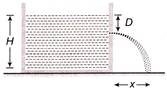
A tank is filled with water up to a height H. Water is allowed to come out of a hole P in one of the walls at a depth D below the surface of water. Express the horizontal distance x in terms of H and D
A)
\[x=\sqrt{D(H-D)}\]
done
clear
B)
\[x=\sqrt{\frac{D(H-D)}{2}}\]
done
clear
C)
\[x=2\,\sqrt{D(H-D)}\]
done
clear
D)
\[x=4\,\sqrt{D(H-D)}\]
done
clear
View Answer play_arrow
When a 4 kg mass is hung vertically on a light spring that obeys Hooke's law, the spring stretches by 2 cm. The work required to be done by an external agent in stretching this spring by 5 cm will be \[(g=9.8\,\,m/{{s}^{2}})\]
A)
4.900 joule
done
clear
B)
2.450 joule
done
clear
C)
0.495 joule
done
clear
D)
0.245 joule
done
clear
View Answer play_arrow
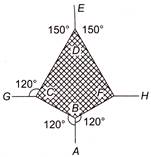
The adjacent figure is the part of a horizontally stretched net. section AB is stretched with a force of 10 N. The tensions in the sections BC and BF are
A)
10 N, 11 N
done
clear
B)
10 N, 6 N
done
clear
C)
10 N, 10 N
done
clear
D)
can't calculate due to insufficient data
done
clear
View Answer play_arrow
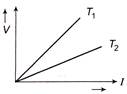
The voltage V and current I graph for a conductor at two different temperatures \[{{T}_{1}}\] and \[{{T}_{2}}\] are shown in the figure. The relation between\[{{T}_{1}}\,and\,{{T}_{2}}\] is
A)
\[{{T}_{1}}\,\,>\,\,{{T}_{2}}\]
done
clear
B)
\[{{T}_{1}}\,\,\approx \,\,{{T}_{2}}\]
done
clear
C)
\[{{T}_{1}}\,\,=\,\,{{T}_{2}}\]
done
clear
D)
\[{{T}_{1}}\,\,<\,\,{{T}_{2}}\]
done
clear
View Answer play_arrow
A long magnetic needle of length 2L, magnetic moment M and pole strength m units is broken into two pieces at the middle. The magnetic moment and pole strength of each piece will be
A)
\[\frac{M}{2},\,\,\frac{m}{2}\]
done
clear
B)
\[M,\,\,\frac{m}{2}\]
done
clear
C)
\[\frac{M}{2},\,\,m\]
done
clear
D)
M, m
done
clear
View Answer play_arrow
The energy that should be added to an electron, to reduce its de-Broglie wavelengths from\[{{10}^{-10}}\,m\,\,to\,\,0.5\times {{10}^{-\,10}}\,m\,\,\], will be
A)
four times the initial energy
done
clear
B)
thrice the initial energy
done
clear
C)
equal to the initial energy
done
clear
D)
twice the initial energy
done
clear
View Answer play_arrow
Energy released in the fission of a single \[_{92}{{U}^{235}}\]nucleus is 200 MeV. The fission rate of a \[_{92}{{U}^{235}}\]fuelled reactor operating at a power level of 5 W is
A)
\[1.56\times {{10}^{+10}}{{s}^{-1}}\]
done
clear
B)
\[1.56\times {{10}^{+11}}{{s}^{-1}}\]
done
clear
C)
\[1.56\times {{10}^{+16}}{{s}^{-1}}\]
done
clear
D)
\[1.56\times {{10}^{+17}}{{s}^{-1}}\]
done
clear
View Answer play_arrow
In a compound microscope, the focal lengths of two lenses are 1.5 cm and 6.25 cm. An object U is placed at 2 cm from objective and the final image is formed at 25 cm from eye lens. The distance between the two lenses is
A)
6.00 cm
done
clear
B)
7.75 cm
done
clear
C)
9.25 cm
done
clear
D)
11.00 cm
done
clear
View Answer play_arrow
Which two of the following five physical parameters have the same dimensions?
(1) energy density (2) refractive index (3) dielectric constant (4) Young?s modulus (5) magnetic field
A)
2 and 4
done
clear
B)
3 and 5
done
clear
C)
1 and 4
done
clear
D)
1 and 5
done
clear
View Answer play_arrow
If energy (E), velocity (V) and time (T) are A chosen as the fundamental quantities, the dimensional formula of surface tension will be
A)
\[[E{{V}^{-\,2}}\,{{T}^{-\,1}}]\]
done
clear
B)
\[[E{{V}^{-\,1}}\,{{T}^{-\,2}}]\]
done
clear
C)
\[[E{{V}^{-\,2}}\,{{T}^{-\,2}}]\]
done
clear
D)
\[[{{E}^{-\,2}}\,{{V}^{-\,1}}\,{{T}^{-\,3}}]\]
done
clear
View Answer play_arrow
The displacement x of a particle varies with time\[\operatorname{t}\, as\,\,x = a{{e}^{-at}}+ b{{e}^{\beta t}}\], where a, b, \[\alpha \,\,and\,\,\beta \] are positive constants. The velocity of the particle will
A)
go on decreasing with time
done
clear
B)
be independent of \[\alpha \,\,and\,\,\beta \]
done
clear
C)
drop to zero when \[\alpha \,\,=\,\,\beta \]
done
clear
D)
go on increasing with time
done
clear
View Answer play_arrow
A particle moves in a straight line with a constant acceleration. It changes its velocity from \[10 m{{s}^{-}}^{1}\,\,to\,\,20 m{{s}^{-}}^{1}\] while passing through a distance 135 m in t second. The value of t is
A)
10
done
clear
B)
1.8
done
clear
C)
12
done
clear
D)
9
done
clear
View Answer play_arrow
A stone tied to the end of a string of 1 m long is whirled in a horizontal circle with a constant v-4 speed. If the stone makes 22 revolutions in 44 s, what is the magnitude and direction of acceleration of the stone?
A)
\[\frac{{{\pi }^{2}}}{4}\,m{{s}^{-\,2}}\] and direction along the radius towards the centre
done
clear
B)
\[{{\pi }^{2}}\,m{{s}^{-}}^{2}\] and direction along the radius away from centre
done
clear
C)
\[{{\pi }^{2}}\,m{{s}^{-}}^{2}\] and direction along the radius towards the centre
done
clear
D)
\[{{\pi }^{2}}\,m{{s}^{-}}^{2}\] and direction along the tangent to the circle
done
clear
View Answer play_arrow
Penetration power of proton is
A)
more than electron
done
clear
B)
less than electron
done
clear
C)
more than neutron
done
clear
D)
none
done
clear
View Answer play_arrow
The nucleus of helium contains
A)
four protons
done
clear
B)
four neutrons
done
clear
C)
two neutrons and two protons
done
clear
D)
four protons and two electrons
done
clear
View Answer play_arrow
The unit \[J\text{ }P{{a}^{-}}^{1}\] is equivalent to
A)
\[{{m}^{3}}\]
done
clear
B)
\[c{{m}^{3}}\]
done
clear
C)
\[d{{m}^{3}}\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
The significant figures in 3400 are
A)
2
done
clear
B)
5
done
clear
C)
6
done
clear
D)
4
done
clear
View Answer play_arrow
5 ml of N HCl, 20 ml of N/2 \[{{H}_{2}}S{{O}_{4}}\] and 30 ml of N/3 \[HN{{O}_{3}}\] are mixed together and volume made to one litre. The normality of the resulting solution is
A)
\[\frac{N}{5}\]
done
clear
B)
\[\frac{N}{10}\]
done
clear
C)
\[\frac{N}{20}\]
done
clear
D)
\[\frac{N}{40}\]
done
clear
View Answer play_arrow
25 ml of a solution of barium hydroxide on titration with a 0.1 molar solution of hydrochloric acid gave a litre value of 35 ml. The molarity of barium hydroxide solution was
A)
0.07
done
clear
B)
0.14
done
clear
C)
0.28
done
clear
D)
0.35
done
clear
View Answer play_arrow
Formula of a metallic oxide is MO. The formula of its phosphate will be
A)
\[{{M}_{2}}{{(P{{O}_{4}})}_{2}}\]
done
clear
B)
\[M(P{{O}_{4}})\]
done
clear
C)
\[{{M}_{2}}P{{O}_{4}}\]
done
clear
D)
\[{{M}_{3}}{{(P{{O}_{4}})}_{2}}\]
done
clear
View Answer play_arrow
Which type of compounds show high melting and boiling points?
A)
Electrovalent compounds
done
clear
B)
Covalent compounds
done
clear
C)
Coordinate compounds
done
clear
D)
All the three types of compounds have equal melting and boiling points
done
clear
View Answer play_arrow
In any chemical reaction, equilibrium is supposed to be established when
A)
mutual opposite reactions undergo
done
clear
B)
concentration of reactants and products are equal
done
clear
C)
velocity of mutual reactions becomes equal
done
clear
D)
the temperature of mutual opposite reaction become equal
done
clear
View Answer play_arrow
Which of the following conditions represents an equilibrium?
A)
Freezing of ice in an open vessel, temperature of ice is constant
done
clear
B)
Few drops of water is present along with air in a balloon, temperature of balloon is constant
done
clear
C)
Water is boiling in an open vessel over stove, temperature of water is constant
done
clear
D)
All the statements [a], [b] and [c] are correct for the equilibrium
done
clear
View Answer play_arrow
The temperature of the system decreases in an
A)
adiabatic compression
done
clear
B)
isothermal compression
done
clear
C)
isothermal expansion
done
clear
D)
adiabatic expansion
done
clear
View Answer play_arrow
For the isothermal expansion of an ideal gas
A)
E and H increase
done
clear
B)
E increases but H decreases
done
clear
C)
H increases but E decreases
done
clear
D)
E and H are unaltered
done
clear
View Answer play_arrow
Which one of the following statements is wrong for gases?
A)
Gases do not have a definite shape and volume
done
clear
B)
Volume of the gas is equal to the volume of the container confining the gas
done
clear
C)
Confined gas exerts uniform pressure on the walls of its container in all directions
done
clear
D)
Mass of the gas cannot be determined by weighing a container in which it is enclosed
done
clear
View Answer play_arrow
\[{{N}_{2}}\] is found in a litre flask under 100 kpa pressure and \[{{O}_{2}}\] is found in another 3 litre flask under 320 kPa pressure. If the two flasks are connected, the resultant pressure is
A)
310 kPa
done
clear
B)
210 kPa
done
clear
C)
420 kPa
done
clear
D)
265 kPa
done
clear
View Answer play_arrow
\[_{6}{{C}^{14}}\] is formed from \[_{7}{{N}^{14}}\] in the upper atmosphere by the action of the fundamental particle
A)
Positron
done
clear
B)
Neutron
done
clear
C)
Electron
done
clear
D)
Proton
done
clear
View Answer play_arrow
Which of the following nuclides has the magic number of both protons and neutrons?
A)
\[_{50}S{{n}^{115}}\]
done
clear
B)
\[_{82}P{{b}^{206}}\]
done
clear
C)
\[_{82}P{{b}^{208}}\]
done
clear
D)
\[_{50}S{{n}^{118}}\]
done
clear
View Answer play_arrow
Under which category iodine crystals are placed among the following?
A)
Ionic crystal
done
clear
B)
Metallic crystal
done
clear
C)
Molecular crystal
done
clear
D)
Covalent crystal
done
clear
View Answer play_arrow
To get an w-type semiconductor, the impurity to be added to silicon should have which of the following number of valence electrons
A)
1
done
clear
B)
2
done
clear
C)
3
done
clear
D)
5
done
clear
View Answer play_arrow
The addition of a polar solvent to a solid electrolyte results in
A)
polarization
done
clear
B)
association
done
clear
C)
ionization
done
clear
D)
non-liberation of heat
done
clear
View Answer play_arrow
During electrolysis of NaCl solution, part of the reaction is\[N{{a}^{+}}\,+\,\,{{e}^{-}}\,\,\to \,\,Na\]. This is termed as
A)
oxidation
done
clear
B)
reduction
done
clear
C)
deposition
done
clear
D)
cathode reaction
done
clear
View Answer play_arrow
Which of the following ores does not represent the ore of iron?
A)
Haematite
done
clear
B)
Magnetite
done
clear
C)
Cassiterite
done
clear
D)
Limonite
done
clear
View Answer play_arrow
Copper can be extracted from
A)
Kupfemickel
done
clear
B)
Dolomite
done
clear
C)
Galena
done
clear
D)
Malachite
done
clear
View Answer play_arrow
The metal which displaces hydrogen from a boiling caustic soda solution is
A)
As
done
clear
B)
Zn
done
clear
C)
Mg
done
clear
D)
Fe
done
clear
View Answer play_arrow
The sum of protons, electrons and neutrons in the heaviest isotope of hydrogen is
A)
6
done
clear
B)
5
done
clear
C)
4
done
clear
D)
3
done
clear
View Answer play_arrow
Which of the following compounds is used in antiknock compositions to prevent the deposition of oxides of lead on spark plug, combustion chamber and exhaust pipe?
A)
Glycerol
done
clear
B)
Glycol
done
clear
C)
1, 2-dibromoethane
done
clear
D)
Benzene
done
clear
View Answer play_arrow
Which of the following is oxidised by\[KMn{{O}_{4}}\]?
A)
Methane
done
clear
B)
Pentane
done
clear
C)
Isobutane
done
clear
D)
Neopentane
done
clear
View Answer play_arrow
In a reaction the ferrous \[(F{{e}^{2+}})\] iron is oxidised to ferric \[(F{{e}^{3+}})\] ion. The equivalent weight of the ion in the above reaction is equal to
A)
half of the atomic weight
done
clear
B)
1/5 of the atomic weight
done
clear
C)
the atomic weight
done
clear
D)
twice the atomic weight
done
clear
View Answer play_arrow
Which forms coloured salts?
A)
Metals
done
clear
B)
Non-metals
done
clear
C)
p-block elements
done
clear
D)
Transitional elements
done
clear
View Answer play_arrow
Which of the following is not true for ligand-metal complex?
A)
Larger the ligand, the more stable is the metal-ligand bond
done
clear
B)
Highly charged ligand forms strong bond
done
clear
C)
Larger the permanent dipole moment of ligand, the more stable is the bond
done
clear
D)
Greater the ionization potential of central metal, the stronger is the bond
done
clear
View Answer play_arrow
What is the coordination number of the metal in\[{{\left[ Co{{(en)}_{2}}C{{l}_{2}} \right]}^{+}}\]?
A)
4
done
clear
B)
5
done
clear
C)
6
done
clear
D)
3
done
clear
View Answer play_arrow
When copper turnings are added to silver nitrate solution, a blue coloured solution is formed after some time. It is because, copper
A)
displaces silver from the solution
done
clear
B)
forms a blue coloured complex with \[AgN{{O}_{3}}\]
done
clear
C)
is oxidised to \[C{{u}^{2+}}\]
done
clear
D)
is reduced to \[C{{u}^{2+}}\]
done
clear
View Answer play_arrow
When \[S{{n}^{2+}}\] changes to \[S{{n}^{4+}}\] in a reaction
A)
it loses two electrons
done
clear
B)
it gains two electrons
done
clear
C)
it loses two protons
done
clear
D)
it gains two protons
done
clear
View Answer play_arrow
In the periodic table, the element with atomic number 16 will be placed in the group
A)
third
done
clear
B)
fourth
done
clear
C)
fifth
done
clear
D)
sixth
done
clear
View Answer play_arrow
The d-block elements consist mostly of
A)
mono valent metals
done
clear
B)
all non-metals
done
clear
C)
elements which generally form stoichiometric metal oxide
done
clear
D)
many metals with catalytic properties
done
clear
View Answer play_arrow
The equivalent conductance at infinite dilution of a weak acid such as HF
A)
can be determined by measurement of very dilute HF solution
done
clear
B)
can be determined by extrapolation of measurements on dilute solutions of HCl, Br and HI
done
clear
C)
can best be determined from measurements on dilute solutions of NaF, NaCl and HCl.
done
clear
D)
is an undefined quantity
done
clear
View Answer play_arrow
If \[\alpha \] is the degree of ionization, C the concentration of a weak electrolyte and \[{{K}_{a}}\] the acid ionization constant, then the correct relationship between \[\alpha \], C and \[{{K}_{a}}\] is
A)
\[{{\alpha }^{2}}=\,\sqrt{\frac{{{K}_{a}}}{C}}\]
done
clear
B)
\[{{\alpha }^{2}}=\,\sqrt{\frac{{{C}_{a}}}{K}}\]
done
clear
C)
\[\alpha =\,\sqrt{\frac{{{K}_{a}}}{C}}\]
done
clear
D)
\[\alpha =\,\sqrt{\frac{{{C}_{a}}}{K}}\]
done
clear
View Answer play_arrow
The adsorption of a gas on a solid surface varies with pressure of the gas in which of the following manner?
A)
Fast \[\to \] slow \[\to \] independent of the pressure
done
clear
B)
Slow \[\to \] fast \[\to \] independent of the pressure
done
clear
C)
Independent of the pressure \[\to \] fast \[\to \] slow
done
clear
D)
Independent of the pressure \[\to \] slow \[\to \] fast
done
clear
View Answer play_arrow
In Freundlich adsorption, isotherm adsorption is proportional to pressure P as
A)
\[{{P}^{0}}\]
done
clear
B)
P
done
clear
C)
\[{{P}^{n}}\]
done
clear
D)
\[{{P}^{1/n}}\]
done
clear
View Answer play_arrow
For the reaction \[{{\operatorname{N}}_{3}}\left( g \right) + 3{{H}_{2}}\left( g \right)\,\,\to \,\,2N{{H}_{3}}\left( g \right)\] under certain conditions of temperature and partial pressure of the reactants, the rate of formation of \[N{{H}_{3}}\] is\[0.001\text{ }kg\,\,{{h}^{-1}}\]. The rate of conversion of \[{{H}_{2}}\], under the same conditions is
A)
\[1.82\times {{10}^{-4}}\,kg/hr\]
done
clear
B)
0.0015 kg/hr
done
clear
C)
\[1.52\times {{10}^{4}}\,kg/hr\]
done
clear
D)
\[1.82\times {{10}^{-14}}\,kg/hr\]
done
clear
View Answer play_arrow
For the reaction \[{{N}_{2}}+3{{H}_{2}}\to 2N{{H}_{3}}\] If \[\frac{\Delta \left[ N{{H}_{3}} \right]}{\Delta t}\,=\,\,2\times {{10}^{-\,4}}\,mol\,{{1}^{-1}}\,{{s}^{-1}}\], the value of \[\frac{-\Delta \left[ {{H}_{2}} \right]}{\Delta t}\] would be
A)
\[1\times {{10}^{-4}}\,mol\,\,{{1}^{-1}}\,{{s}^{-1}}\]
done
clear
B)
\[3\times {{10}^{-4}}\,mol\,\,{{1}^{-1}}\,{{s}^{-1}}\]
done
clear
C)
\[4\times {{10}^{-4}}\,mol\,\,{{1}^{-1}}\,{{s}^{-1}}\]
done
clear
D)
\[6\times {{10}^{-4}}\,mol\,\,{{1}^{-1}}\,{{s}^{-1}}\]
done
clear
View Answer play_arrow
The ionic strength of \[N{{a}^{+}}\] on mixing 100 mL 0.1 M Nacl and 100 mL 0.1 M \[N{{a}_{2}}S{{O}_{4}}\] is:
A)
0.2
done
clear
B)
0.1
done
clear
C)
0.3
done
clear
D)
0.075
done
clear
View Answer play_arrow
An organic compound contains carbon hydrogen and oxygen. Its elemental analysis gave C,\[38.71\,%,\text{ }and\text{ }H,\text{ }9.67\,%\]. The empirical formula of the compound would be:
A)
\[C{{H}_{3}}O\]
done
clear
B)
\[C{{H}_{2}}O\]
done
clear
C)
CHO
done
clear
D)
\[C{{H}_{4}}O\]
done
clear
View Answer play_arrow
The frequency of radiations emitted when electron falls from \[\operatorname{n}= 4 \,to\,\,n =1\] in H-atom would be (Given \[{{G}_{1}}\,for\,\,H=2.18\,\,\times \,\,1{{0}^{-\,18}}\,J\,\,ato{{m}^{-\,1}}\] and \[h=6.625\times {{10}^{-34}}\,Js.\]
A)
\[1.54\times 1015\,{{s}^{-}}^{1}\]
done
clear
B)
\[1.03\times 1015\,{{s}^{-}}^{1}\]
done
clear
C)
\[3.08\times 1015\,{{s}^{-}}^{1}\]
done
clear
D)
\[2.0\times 1015\,{{s}^{-}}^{1}\]
done
clear
View Answer play_arrow
The energy absorbed by each molecule \[({{A}_{2}})\] of a substance is \[4.4 \times 1{{0}^{19}}J\] and bond energy per molecule is \[4.0 \times 1{{0}^{19}}J\]. The kinetic energy of the molecule per atom will be
A)
\[2.0\times {{10}^{-}}^{20}\,J\]
done
clear
B)
\[2.2\times {{10}^{-}}^{19}\,J\]
done
clear
C)
\[2.0\times {{10}^{-}}^{19}\,J\]
done
clear
D)
\[4.0\times {{10}^{-}}^{20}\,J\]
done
clear
View Answer play_arrow
Lanthanoids are:
A)
14 elements in the sixth period (at. no. 90 XJ to 103) that have 4f-subshell
done
clear
B)
14 elements in the seventh period (at. no. 90 to 103) that have 5f-subshell
done
clear
C)
14 elements in the sixth period (at. no. 58 to 71) that have 4f-subshell
done
clear
D)
14 elements in the seventh period (at. no. 58 to 71) that have 4f-subshell
done
clear
View Answer play_arrow
The amphids arc cuticular elevations on the ventro-lateral lips of Ascaris. These are
A)
tactoreceptors
done
clear
B)
chemoreceptors
done
clear
C)
olfactoreceptors
done
clear
D)
tangoreceptors
done
clear
View Answer play_arrow
Hydra recognises its prey by
A)
chemical stimulus of prey
done
clear
B)
nematocyst
done
clear
C)
mechanical stimulus of prey
done
clear
D)
some special organs
done
clear
View Answer play_arrow
The major constituent of vertebrate bone is
A)
potassium hydroxide
done
clear
B)
sodium chloride
done
clear
C)
calcium carbonate
done
clear
D)
calcium phosphate
done
clear
View Answer play_arrow
The function of ductus choledochus is to carry
A)
urine
done
clear
B)
ova
done
clear
C)
sperms
done
clear
D)
bile
done
clear
View Answer play_arrow
Which of the following gland controls egg production and moulting in insects?
A)
corpus callosum
done
clear
B)
corpora allata
done
clear
C)
corpora cardiac
done
clear
D)
all of these
done
clear
View Answer play_arrow
Chordae tendinae are found in
A)
ventricles of brain
done
clear
B)
joints of legs
done
clear
C)
ventricles of heart
done
clear
D)
atria of heart
done
clear
View Answer play_arrow
In mammals, the digestion of starch starts from
A)
oesophagus
done
clear
B)
mouth
done
clear
C)
duodenum
done
clear
D)
stomach
done
clear
View Answer play_arrow
Which of the following is flightless bird?
A)
ostrich
done
clear
B)
emu
done
clear
C)
kiwi
done
clear
D)
all of these
done
clear
View Answer play_arrow
The camel?s hump is composed of a tissue, which provides water when oxidised is, called
A)
areolar
done
clear
B)
skeletal
done
clear
C)
adipose
done
clear
D)
muscular
done
clear
View Answer play_arrow
Wharton's duct is the duct of
A)
submandioular salivary gland
done
clear
B)
parotid gland
done
clear
C)
submaxillary gland
done
clear
D)
all of these
done
clear
View Answer play_arrow
The horns of Rhinoceros are composed of
A)
chitin
done
clear
B)
bone
done
clear
C)
keratin
done
clear
D)
cartilage
done
clear
View Answer play_arrow
Sharpey's perforating fibres are related with
A)
muscle contraction
done
clear
B)
fixing of teeth
done
clear
C)
heart contraction
done
clear
D)
all of these
done
clear
View Answer play_arrow
Knock knee disease is due to
A)
deficiency in tyrosine amino acid
done
clear
B)
hormonal imbalance
done
clear
C)
excess fluoride concentration in waterbody
done
clear
D)
genetical abnormality in males
done
clear
View Answer play_arrow
Sweat glands, in human skin, are located in the
A)
malpighian layer of epidermis
done
clear
B)
dermis of skin
done
clear
C)
subdermal layer of fat cells
done
clear
D)
glandular layer of epidermis
done
clear
View Answer play_arrow
Where does the conversion of harmful prussic acid into potassium sulphocyanide take place?
A)
bone marrow
done
clear
B)
spleen
done
clear
C)
lymph glands
done
clear
D)
liver
done
clear
View Answer play_arrow
Which of the following is not applicable to coelenterates?
A)
choanocytes
done
clear
B)
coelenteron
done
clear
C)
radial symmetry
done
clear
D)
nematoblasts
done
clear
View Answer play_arrow
Function of contractile vacuole in protozoa is
A)
osmoregulation
done
clear
B)
locomotion
done
clear
C)
respiration
done
clear
D)
digestion of food
done
clear
View Answer play_arrow
Animals belonging to order rodentia have
A)
long canines
done
clear
B)
long incisors
done
clear
C)
long molars
done
clear
D)
short incisors
done
clear
View Answer play_arrow
How many teeth grow only once in the life of a man?
A)
20
done
clear
B)
8
done
clear
C)
32
done
clear
D)
12
done
clear
View Answer play_arrow
Glycosidic link is broken in digestion of
A)
starch
done
clear
B)
protein
done
clear
C)
lipid
done
clear
D)
all of these
done
clear
View Answer play_arrow
Which of the following is most convincing reasons for increasing population growth in a country?
A)
low population of old people
done
clear
B)
high birth rate
done
clear
C)
high population of young children
done
clear
D)
low mortality rate
done
clear
View Answer play_arrow
Radial symmetry is usually exhibited in animals, which
A)
are aquatic
done
clear
B)
have ciliary mode of feeding
done
clear
C)
are attached to some substratum (sedentary)
done
clear
D)
none of these
done
clear
View Answer play_arrow
Which of the following has been declared a killer disease under Factory Act?
A)
tuberculosis
done
clear
B)
asbestosis
done
clear
C)
shigellosis
done
clear
D)
silicosis
done
clear
View Answer play_arrow
In Gloriosa, the tendrillar part is formed by B
A)
leaf petiole
done
clear
B)
stipule
done
clear
C)
axillary bud
done
clear
D)
leaf apex
done
clear
View Answer play_arrow
If PH of stomach is 1.6, then which enzyme will digest protein?
A)
amylase
done
clear
B)
trypsin
done
clear
C)
erypsin
done
clear
D)
pepsin
done
clear
View Answer play_arrow
When ovules at two points are develop from the inner wall of the unilocular ovary, the placentation is called
A)
marginal
done
clear
B)
parietal
done
clear
C)
superficial
done
clear
D)
basal
done
clear
View Answer play_arrow
Yellow bone marrow is found especially in the medullary cavity of
A)
short bones
done
clear
B)
spongy bones
done
clear
C)
long bones
done
clear
D)
all of these
done
clear
View Answer play_arrow
Carbon dioxide is necessary for photosynthesis. The chemical used to remove this gas most effectively from entering a control apparatus is
A)
calcium oxide
done
clear
B)
distilled water
done
clear
C)
potassium hydroxide solution
done
clear
D)
sodium carbonate
done
clear
View Answer play_arrow
'Sudan Black B' stain is used to stain
A)
nucleic acid
done
clear
B)
protein
done
clear
C)
polysaccharide
done
clear
D)
lipid
done
clear
View Answer play_arrow
Life in biosphere is abundant between
A)
200 m below sea level to 6000 m above sea level
done
clear
B)
6000 m below sea level to 6000 m above sea level
done
clear
C)
11000 m below sea level to 9000 m above sea level
done
clear
D)
6000 m below sea level to 2000 m above sea level.
done
clear
View Answer play_arrow
Which of the following correctly represents the flow of genetic information?
A)
DNA \[\to \] RNA \[\to \] protein
done
clear
B)
RNA \[\to \] DNA \[\to \] protein
done
clear
C)
RNA \[\to \] protein \[\to \] DNA
done
clear
D)
protein \[\to \] RNA \[\to \] DNA
done
clear
View Answer play_arrow
Starch and cellulose are the compounds of many units of
A)
amino acids
done
clear
B)
glycerol
done
clear
C)
simple sugars
done
clear
D)
fatty acids
done
clear
View Answer play_arrow
An ecological pyramid of biomass is the representation of the ecosystems
A)
energy flow through each trophic level
done
clear
B)
population in each food web
done
clear
C)
tissue organisation at each trophic level
done
clear
D)
all of these
done
clear
View Answer play_arrow
Which part of the world has a high density of organisms?
A)
deciduous forests
done
clear
B)
grasslands
done
clear
C)
tropical rain forests
done
clear
D)
savannahs
done
clear
View Answer play_arrow
In a lake, the upper warmer oxygen-rich circulating water layer zone is referred to
A)
limnetic zone
done
clear
B)
epilimnion
done
clear
C)
promndal zone
done
clear
D)
hypolimnion
done
clear
View Answer play_arrow
Red Data Book deals with
A)
plants on the verge of extinction
done
clear
B)
endemic plant
done
clear
C)
plants showing photoperiodism
done
clear
D)
plants that are extinct
done
clear
View Answer play_arrow
Which of the following is not a correct match?
A)
Aflatoxin - Aspergillus parasiticus
done
clear
B)
Bengal famine - Helminthosporiurn
done
clear
C)
Redrust of tea - Melampsora
done
clear
D)
Tikka disease of groundnut - Cercospora
done
clear
View Answer play_arrow
How many cytochromes are involved in the electron transport chain?
A)
five
done
clear
B)
two
done
clear
C)
six
done
clear
D)
four
done
clear
View Answer play_arrow
In gymnosperm, the endosperm is formed by
A)
fusion of two polar nuclei
done
clear
B)
fertilised egg
done
clear
C)
fusion of one polar nucleus
done
clear
D)
germination of one megaspore
done
clear
View Answer play_arrow
Krantz anatomy is seen in
A)
Euphorbia hirta
done
clear
B)
Citrus indica
done
clear
C)
Mangifera indica
done
clear
D)
Zeamays
done
clear
View Answer play_arrow
During photosynthesis, oxygen in glucose comes from
A)
oxygen in air
done
clear
B)
water
done
clear
C)
carbon dioxide
done
clear
D)
both [b] and [c]
done
clear
View Answer play_arrow
The niche of a population is the
A)
set of conditions that it interacts
done
clear
B)
place where it lives
done
clear
C)
set of conditions and resources it uses
done
clear
D)
geographical area that it covers
done
clear
View Answer play_arrow
The best source of vitamin C is
A)
Citrus indica
done
clear
B)
Glycine max
done
clear
C)
Emblica officinalis
done
clear
D)
Arachis hypogea
done
clear
View Answer play_arrow
The molar ratio of chlorophyll and xanthophyll is
A)
\[4:1\]
done
clear
B)
\[3:1\]
done
clear
C)
\[1:1\]
done
clear
D)
\[2:1\]
done
clear
View Answer play_arrow
Shifting cultivation requires
A)
alternate crop pattern on a particular area
done
clear
B)
huge amount of commercial fertiliser
done
clear
C)
excessive soil erosion for crop production
done
clear
D)
long time for the regeneration of soil
done
clear
View Answer play_arrow
Major source of sugar in the world is
A)
Citrullus vulgaris
done
clear
B)
Beta vulgaris
done
clear
C)
Saccharum officinarum
done
clear
D)
Annona squamosa
done
clear
View Answer play_arrow
Penicillium does not allow the growth of bacterium Staphylococcus. This sort of relationship is called
A)
commensalism
done
clear
B)
antagonism
done
clear
C)
ammensalism
done
clear
D)
mutualism
done
clear
View Answer play_arrow
The reflectivity percentage of incident light on the earth is meteorologically called as
A)
reradiation
done
clear
B)
tornado
done
clear
C)
irrefraction
done
clear
D)
albedo
done
clear
View Answer play_arrow
Plants which can withstand wide range of temperature tolerance are called
A)
monothermic
done
clear
B)
stenothermic
done
clear
C)
mesothermic
done
clear
D)
eurythermic
done
clear
View Answer play_arrow
Which of the following is not a correct match?
A)
chord moss - Funaria
done
clear
B)
maiden hair fern - Ginkgo
done
clear
C)
walking fern - Camptosorus
done
clear
D)
bog moss - Sphagnum
done
clear
View Answer play_arrow
All of the following statements concerning the Actinomycetes filamentous soil bacterium Frankia are correct EXCEPT that Frankia
A)
Forms specialized vesicles in which the nitrogenase is protected from oxygen by a chemical barrier involving triterpene hopanoids
done
clear
B)
Can induce root nodules on many plant species
done
clear
C)
Like Rhizobium, it usually infects its host plant through root hair deformation and stimulates cell proliferation in the host?s cortex
done
clear
D)
Cannot fix nitrogen in the free-living stale
done
clear
View Answer play_arrow
In eubacteria, a cellular component that resembles eukaryotic cell is:
A)
Cell wall
done
clear
B)
Plasma membrane
done
clear
C)
Nucleus
done
clear
D)
Ribosomes
done
clear
View Answer play_arrow
Which one of the following pairs of plants are not seed producers?
A)
Fern and Funaria
done
clear
B)
Funaria and Ficus
done
clear
C)
Ficus and Chlamydomonas
done
clear
D)
Punica and Pinus
done
clear
View Answer play_arrow
Spore dissemination in some liverworts is aided by:
A)
Peristome teeth
done
clear
B)
Elaters
done
clear
C)
Indusium
done
clear
D)
Calyptra
done
clear
View Answer play_arrow
Two common characters found in centipede cockroach, and crab are:
A)
Compound eyes and anal cerci
done
clear
B)
Jointed legs and chitinous exoskeletoa
done
clear
C)
Green gland and tracheae
done
clear
D)
Book lungs and antennae
done
clear
View Answer play_arrow
Which one of the following phyla is correctly matched with its two general characteristic?
A)
Echinodermata - Pentamerous radical symmetry and mostly internal fertilization
done
clear
B)
Mollusca - Normally oviparous and development through a trochophore or veliger larva
done
clear
C)
Arthropoda - Body divided into head, thorax and abdomen and respiration by tracheae
done
clear
D)
Chordata - Notochord at some stage and separate anal and urinary openings to the outside
done
clear
View Answer play_arrow
The Replum is present in the ovary of flower of:
A)
sunflower
done
clear
B)
pea
done
clear
C)
lemon
done
clear
D)
mustard
done
clear
View Answer play_arrow
Placenta and pericarp are both edible portions in:
A)
Apple
done
clear
B)
Banana
done
clear
C)
Tomato
done
clear
D)
Potato
done
clear
View Answer play_arrow
Chlorenchyma found in:
A)
Cytoplasm of Chlorella
done
clear
B)
Mycelium of a green mould such as Aspergillus
done
clear
C)
Spore capsule of a moss
done
clear
D)
Pollen tube of Pinus
done
clear
View Answer play_arrow
Lenticels are involved in
A)
Transpiration
done
clear
B)
Gaseous exchange
done
clear
C)
Food transport
done
clear
D)
Photosynthesis
done
clear
View Answer play_arrow
Which of the following is correctly stated as it happens in the common cockroach?
A)
The food is ground by mandibles and gizzard
done
clear
B)
Malpighian tubules are excretory organs projecting out from the colon
done
clear
C)
Oxygen is transported by haemoglobin in blood
done
clear
D)
Nitrogenous excretory product is urea
done
clear
View Answer play_arrow
What external changes are visible after the last moult of a cockroach nymph?
A)
Mandibles become harder
done
clear
B)
Anal cerci develop
done
clear
C)
Both fore wings and hind wings develop
done
clear
D)
Labium develops
done
clear
View Answer play_arrow
Stroma in the chloroplast of higher plant contains:
A)
Chlorophyll
done
clear
B)
Light-independent reaction enzymes
done
clear
C)
Light-dependent reaction enzymes
done
clear
D)
Ribosomes
done
clear
View Answer play_arrow
Important site for the formation of glycol proteins and glycolipids is:
A)
Lysosome
done
clear
B)
Vacuole
done
clear
C)
Golgi apparatus
done
clear
D)
Plastid
done
clear
View Answer play_arrow
The osmotic expansion of a cell kept in water is chiefly regulated by:
A)
Mitochondria
done
clear
B)
Vacuoles
done
clear
C)
Plastids
done
clear
D)
Ribosomes
done
clear
View Answer play_arrow
About 98 percent of the mass of every living organism is composed of just six elements including carbon, hydrogen, nitrogen, oxygen and:
A)
Calcium and phosphorus
done
clear
B)
Phosphorus and sulphur
done
clear
C)
Sulphur and magnesium
done
clear
D)
Magnesium and sodium
done
clear
View Answer play_arrow
Transition state structure of the substrate formed during an enzymatic reaction is:
A)
Transient but stable
done
clear
B)
Permanent but unstable
done
clear
C)
Transient and unstable
done
clear
D)
Permanent and stable
done
clear
View Answer play_arrow
The complex formed by a pair of synapsed homologous chromosomes is called:
A)
Equatorial plate
done
clear
B)
Kinetochore
done
clear
C)
Bivalent
done
clear
D)
Axoneme
done
clear
View Answer play_arrow
One of the free-living anaerobic nitrogen-fixer is
A)
Beijerinckia
done
clear
B)
Rhodospirillum
done
clear
C)
Rhizobium
done
clear
D)
Azotobacter
done
clear
View Answer play_arrow
Which one of the following is correctly matched?
A)
Potassium - Readily immobilization
done
clear
B)
Bakane of rice seedlings - F skoog
done
clear
C)
Passive transport of nutrients - ATP
done
clear
D)
Apoplast - Plasmodesmata
done
clear
View Answer play_arrow
Electrons from excited chlorophyll molecule of photosystem II are accepted first by:
A)
Quinone
done
clear
B)
Ferredoxin
done
clear
C)
Cytochrome-b
done
clear
D)
Cytochrome-f
done
clear
View Answer play_arrow
CAM helps the plants in:
A)
Reproduction
done
clear
B)
Conserving water
done
clear
C)
Secondary growth
done
clear
D)
Disease resistance
done
clear
View Answer play_arrow
A process that makes important difference between \[{{C}_{3}}\,\,and\,\,{{C}_{4}}\] plants is
A)
Photosynthesis
done
clear
B)
Photorespiration
done
clear
C)
Transpiration
done
clear
D)
Glycolysis
done
clear
View Answer play_arrow
One of the synthetic auxin is:
A)
IBA
done
clear
B)
NAA
done
clear
C)
IAA
done
clear
D)
GA
done
clear
View Answer play_arrow
Which one of the following growth regulators is known as stress hormone?
A)
Abscisic acid
done
clear
B)
Ethylene
done
clear
C)
\[G{{A}_{3}}\]
done
clear
D)
Indole acetic acid
done
clear
View Answer play_arrow
Which one of the following statement is true regarding digestion and absorption of food in humans?
A)
About \[60%\] of starch is hydrolysed by salivary amylase in our mouth
done
clear
B)
Oxyntic cells in our stomach secrete the proenzyme pepsinogen
done
clear
C)
Fructose and amino acids are absorbed through intestinal mucosa with the help of carrier ions like \[N{{a}^{+}}\]
done
clear
D)
Chylomicrons are small lipoprotein particles that are transported from intestine into blood capillaries
done
clear
View Answer play_arrow
Two friends are eating together on a dining table. One of them suddenly starts coughing while swallowing some food. This coughing would have been due to improper movement of:
A)
Tongue
done
clear
B)
Epiglottis
done
clear
C)
Diaphragm
done
clear
D)
Neck
done
clear
View Answer play_arrow
The primary dentition in human differs from permanent dentition in not having one of the following type of teeth:
A)
Incisors
done
clear
B)
Canine
done
clear
C)
Premolars
done
clear
D)
Molars
done
clear
View Answer play_arrow
Arteries are best defined as the vessel which:
A)
carry blood from one visceral organ to another visceral organ
done
clear
B)
supply oxygenated blood to the different organs
done
clear
C)
carry blood away from the heart to different organs
done
clear
D)
break up into capillaries which reunite to form a vein
done
clear
View Answer play_arrow
Erythropoiesis starts in:
A)
Red bone marrow
done
clear
B)
Kidney
done
clear
C)
Liver
done
clear
D)
Spleen
done
clear
View Answer play_arrow
Alzheimer disease in humans is associated with the deficiency of:
A)
Gamma aminobutyric acid (GABA)
done
clear
B)
Dopamine
done
clear
C)
Glutamic acid
done
clear
D)
Acetylcholine
done
clear
View Answer play_arrow
Injury localized to the hypothalamus would most likely disrupt:
A)
short-term memory
done
clear
B)
coordination during locomotion
done
clear
C)
executive functions, such as decision making.
done
clear
D)
regulation of body temperature
done
clear
View Answer play_arrow
The blood calcium level is lowered by the deficiency of:
A)
both calcitonin and parathormone
done
clear
B)
calcitonin
done
clear
C)
parathormone
done
clear
D)
thyroxine
done
clear
View Answer play_arrow
What is correct to say about the hormone action in humans?
A)
In females, FSH first binds with specific receptors on ovarian cell membrane
done
clear
B)
FSH stimulates the secretion of estrogen and progesterone
done
clear
C)
Glucagon is secreted by \[\beta -cells\] of islets of Langerhans and stimulates glycogenolysis
done
clear
D)
Secretion of thymosins is stimulated with aging
done
clear
View Answer play_arrow
Identify the hormone with its correct matching of source and function:
A)
Oxytocin - posterior pituitary, growth and maintenance of mammary glands.
done
clear
B)
Melatonin - pineal gland, regulates the, normal rhythm of sleepwake cycle.
done
clear
C)
Progesterone - corpus-luteum, stimulation of growth and activities of female secondary sex organs.
done
clear
D)
atrial natriuretic factor - ventricular wall increases the blood pressure.
done
clear
View Answer play_arrow
A fruit developed from hypanthodium inflorescence is called:
A)
Caryopsis
done
clear
B)
Hesperidium
done
clear
C)
Sorosis
done
clear
D)
Syconus
done
clear
View Answer play_arrow
Seed coat is not thin membranous in:
A)
Maize
done
clear
B)
Coconut
done
clear
C)
Groundnut
done
clear
D)
Gram
done
clear
View Answer play_arrow
Male gametophyte in angiosperms produces:
A)
Three sperms
done
clear
B)
Two sperms and a vegetative cell
done
clear
C)
Single sperm and a vegetative cell
done
clear
D)
Single sperm and two vegetative cells
done
clear
View Answer play_arrow
Sertoli cells are found in:
A)
ovaries and secrete progesterone
done
clear
B)
adrenal cortex and secrete adrenaline
done
clear
C)
seminiferous tubules and provide nutrition to germ cells
done
clear
D)
pancreas and secrete cholecystokinin
done
clear
View Answer play_arrow
Which one of the following statements is false in respect of viability of mammalian sperm?
A)
Viability of sperm is determined by its motility
done
clear
B)
Sperms must be concentrated in a thick suspension
done
clear
C)
Sperm is viable for only up to 24 hours.
done
clear
D)
Survival of sperm depends on the pH of the medium and is more active in alkaline medium.
done
clear
View Answer play_arrow
 Match the column according to correct curve:
Match the column according to correct curve: