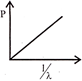
If P represents radiation pressure, C represents speed of light and Q represents radiation striking unit area second, then non zero integers x, y and z, such that \[{{P}^{x}},{{Q}^{y}},{{C}^{z}}\] is dimensionless are:
A)
x = 1, y = 1, z = -1
done
clear
B)
x = 1, y = -1, z = 1
done
clear
C)
x = -1, y = 1, z = 1
done
clear
D)
x = I, y = I, z = 1
done
clear
View Answer play_arrow
The time period of second's pendulum is 2 sec the spherical bob which is empty from inside has a mass of 50g. This is now replaced by another solid bob of same radius but having a different mass of 100g. The new time period will be:
A)
4 sec
done
clear
B)
1 sec
done
clear
C)
2 sec
done
clear
D)
8 sec
done
clear
View Answer play_arrow
If R is the horizontal range for an inclination and h is the maximum height reached by the projectile, then the maximum range is given by:
A)
\[\frac{{{R}^{2}}}{8h}-2h\]
done
clear
B)
\[\frac{{{R}^{2}}}{8h}-2gh\]
done
clear
C)
\[\frac{{{R}^{2}}}{8h}+2h\]
done
clear
D)
\[\frac{{{R}^{2}}}{8h}\]
done
clear
View Answer play_arrow
An Indian rubber cord 'L' metres long and area of cross section \[A({{m}^{2}})\]is suspended vertically. Density of rubber is \[\rho \,\,kg/{{m}^{3}}\]and young's modulus of rubber is\[Y\,N/{{m}^{2}}\]. If the rod extends \[\ell \] metres under its own weight, then extension \[\Delta \ell \] is:
A)
\[\frac{{{L}^{2}}\rho g}{Y}\]
done
clear
B)
\[\frac{{{L}^{2}}\rho g}{2Y}\]
done
clear
C)
\[\frac{{{L}^{2}}\rho g}{4Y}\]
done
clear
D)
\[\frac{Y}{{{L}^{2}}\rho g}\]
done
clear
View Answer play_arrow
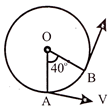
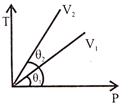
A particle is moving in a circle of radius r centered at o with constant speed v. The change in velocity in moving from A to B:
A)
2v cos \[{{40}^{{}^\circ }}\]
done
clear
B)
2v sm \[{{40}^{{}^\circ }}\]
done
clear
C)
2v cos \[{{20}^{{}^\circ }}\]
done
clear
D)
2v sin \[{{20}^{{}^\circ }}\]
done
clear
View Answer play_arrow
Vector perpendicular to: a\[Cos\,\theta \,\hat{i}+b\text{ }Sin\,\theta \hat{j}\]
A)
\[b\,\sin \,\theta \,\hat{i}+a\,\cos \,\theta \hat{j}\]
done
clear
B)
\[\frac{1}{a}\,\sin \,\theta \,\hat{i}+\frac{1}{b}\,\cos \,\theta \hat{j}\]
done
clear
C)
\[5\hat{k}\]
done
clear
D)
\[\frac{3\,\hat{i}}{a}+\,\cos \,\theta \hat{j}\]
done
clear
View Answer play_arrow
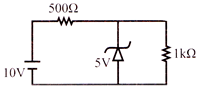
In the following circuit, the current flowing through \[1k\Omega \]resistor is:
A)
0mA
done
clear
B)
5mA
done
clear
C)
10mA
done
clear
D)
15mA
done
clear
View Answer play_arrow
The cylindrical tube of a spray pump has radius R, one end of which has n fine holes, each of radius r. If the speed of the liquid in the tube is V, the speed of the ejection of the liquid through the holes is:
A)
\[\frac{V{{R}^{2}}}{{{n}^{3}}{{r}^{2}}}\]
done
clear
B)
\[\frac{{{V}^{2}}R}{nr}\]
done
clear
C)
\[\frac{V{{R}^{2}}}{{{n}^{2}}{{r}^{2}}}\]
done
clear
D)
\[\frac{V{{R}^{2}}}{n{{r}^{2}}}\]
done
clear
View Answer play_arrow
A proton accelerated through a potential V has de-broglie wave length\[\lambda \]. Then, broglie wavelength of an \[\alpha \]-particle accelerated through the same potent V:
A)
\[\frac{\lambda }{2}\]
done
clear
B)
\[\frac{\lambda }{\sqrt{2}}\]
done
clear
C)
\[\frac{\lambda }{2\sqrt{2}}\]
done
clear
D)
\[\frac{\lambda }{4}\]
done
clear
View Answer play_arrow
The effect due to uniform magnetic field on a freely suspended magnetic needle is as follows:
A)
Both torque and net force are present
done
clear
B)
Torque is present but no net force
done
clear
C)
Both torque and net force are absent
done
clear
D)
Net force is present but not torque
done
clear
View Answer play_arrow
When an electron jumps from the orbit n = 2 to n = 4, then wavelength of the radiations absorbed will be: (R is Rydberg constant)
A)
\[\frac{16}{3R}\]
done
clear
B)
\[\frac{16}{5R}\]
done
clear
C)
\[\frac{5R}{16}\]
done
clear
D)
\[\frac{3R}{16}\]
done
clear
View Answer play_arrow
Two long straight wires are set parallel to each other at separation r and each carries a current I in the same direction. The strength of the agnetic field at any point midway between the two wires is:
A)
\[\frac{{{\mu }_{{}^\circ }}I}{\pi r}\]
done
clear
B)
\[\frac{2{{\mu }_{{}^\circ }}I}{\pi r}\]
done
clear
C)
\[\frac{{{\mu }_{{}^\circ }}I}{2\pi r}\]
done
clear
D)
\[Zero\]
done
clear
View Answer play_arrow
A particle accelerating uniformly has velocity v at time\[{{t}_{1}}\]. What is work done in time t?
A)
\[\frac{1}{2}\left[ \frac{m{{v}^{2}}}{t_{1}^{2}} \right]{{t}^{2}}\]
done
clear
B)
\[\frac{1}{2}\left[ \frac{mv}{{{t}_{1}}} \right]{{t}^{2}}\]
done
clear
C)
\[\left[ \frac{m{{v}^{2}}}{t_{1}^{2}} \right]{{t}^{2}}\]
done
clear
D)
\[\left[ \frac{2\,m{{v}^{2}}}{t_{1}^{2}} \right]{{t}^{2}}\]
done
clear
View Answer play_arrow
Two identical photo cathodes receive light of frequencies \[{{f}_{1}}\] and \[{{f}_{2}}\] If the velocities of the photoelectrons (of mass m) coming out are respectively \[{{V}_{1}}\] and\[{{V}_{2}}\], then:
A)
\[V_{1}^{2}-V_{2}^{2}=\frac{2h}{m}({{f}_{1}}-{{f}_{2}})\]
done
clear
B)
\[{{V}_{1}}-{{V}_{2}}=\sqrt{\frac{2h}{m}({{f}_{1}}-{{f}_{2}})}\]
done
clear
C)
\[V_{1}^{2}+V_{2}^{2}=\frac{2h}{m}({{f}_{1}}-{{f}_{2}})\]
done
clear
D)
\[{{V}_{1}}={{V}_{2}}\]
done
clear
View Answer play_arrow
Bernoulli's equation for steady, non-viscous, incompressible flow expresses the conservation of:
A)
Angular momentum
done
clear
B)
Density
done
clear
C)
Momentum
done
clear
D)
Energy
done
clear
View Answer play_arrow
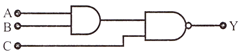
The output Y, when all the three inputs are first high and then low will respectively be:
A)
1, 0
done
clear
B)
1, 1
done
clear
C)
0, 0
done
clear
D)
0, 1
done
clear
View Answer play_arrow
The depth at which the value of acceleration due to gravity becomes \[\frac{1}{n}\] time the value at the surface is:
A)
\[\frac{R}{n}\]
done
clear
B)
\[\frac{R(n-1)}{n}\]
done
clear
C)
\[\frac{Rn}{(n-1)}\]
done
clear
D)
\[\frac{R}{{{n}^{2}}}\]
done
clear
View Answer play_arrow
The current in a self-inductance L = 40mH is to be increased uniformly from 1A to 11A in 4ms. The emf induced in the inductor during the process is:
A)
100V
done
clear
B)
0.4V
done
clear
C)
40V
done
clear
D)
440V
done
clear
View Answer play_arrow
If the equation of a progressive wave is given as \[y=a\,Sin\,\pi \,\left[ \frac{t}{2}-\frac{x}{4} \right]\] where x is in metres and t is in seconds, then the distance through which the waves moves in 8 sec is:
A)
2m
done
clear
B)
16m
done
clear
C)
4m
done
clear
D)
8m
done
clear
View Answer play_arrow
A filament bulb (500W, 100V) is to be used in a 230V main supply. When a resistance R is connected in series, it works perfectly and the bulb consumes 500W. The value of R is:
A)
13Q
done
clear
B)
230Q
done
clear
C)
46Q
done
clear
D)
26Q
done
clear
View Answer play_arrow
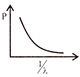
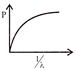
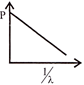
Which of the following figures represent the variation of particle momentum and the associated de-broglie wavelength:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
An ideal gas goes from state A to state B via three different processes as indicated in the P-V diagram. If \[{{Q}_{1}},{{Q}_{2}}\] and \[{{Q}_{3}}\] indicate the heat absorbed by the three processes and \[\Delta {{U}_{1}},\Delta {{U}_{2}}\]and\[\Delta {{U}_{3}}\] indicate the change in internal energy along the three processes respectively, then:
A)
\[{{Q}_{1}}>{{Q}_{2}}>{{Q}_{3}}\] and\[\Delta {{U}_{1}}=\Delta {{U}_{2}}=\Delta {{U}_{3}}\]
done
clear
B)
\[{{Q}_{3}}>{{Q}_{2}}>{{Q}_{1}}\] and \[\Delta {{U}_{1}}=\Delta {{U}_{2}}=\Delta {{U}_{3}}\]
done
clear
C)
\[{{Q}_{1}}={{Q}_{2}}={{Q}_{3}}\] and \[\Delta {{U}_{1}}>\Delta {{U}_{2}}>\Delta {{U}_{3}}\]
done
clear
D)
\[{{Q}_{3}}>{{Q}_{2}}>{{Q}_{1}}\] and \[\Delta {{U}_{1}}>\Delta {{U}_{2}}>\Delta {{U}_{3}}\]
done
clear
View Answer play_arrow
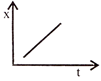
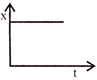
Position-time graph for motion with zero acceleration is:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Ratio of escape velocity at earth \[({{v}_{e}})\]to the escape velocity at a planet \[({{v}_{p}})\] whose radius and mean density are twice as that of earth is:
A)
\[1:2~\]
done
clear
B)
\[1:2~\sqrt{2}\]
done
clear
C)
\[1:4\]
done
clear
D)
\[1:\sqrt{2}\]
done
clear
View Answer play_arrow
A particle of mass \[{{m}_{1}}\] is moving with a velocity, \[{{V}_{1}}\] and another particle of mass, \[{{m}_{2}}\] is moving with a velocity,\[{{V}_{2}}\]. Both of them have the same momentum but their different kinetic energies are \[{{E}_{1}}\]and \[{{E}_{2}}\] respectively. If \[{{m}_{1}}>{{m}_{2}}\] then:
A)
\[{{E}_{1}}<{{E}_{2}}\]
done
clear
B)
\[{{E}_{1}}{{m}_{2}}={{m}_{1}}{{E}_{2}}\]
done
clear
C)
\[E=\,\,>\,{{E}_{2}}\]
done
clear
D)
\[{{E}_{1}}={{E}_{2}}\]
done
clear
View Answer play_arrow
The number of possible natural oscillations of air column in a pipe closed at one end of length 85 cm whose frequencies lie below 1250 Hz are: (velocity of sound = 340 m/s)
A)
4
done
clear
B)
5
done
clear
C)
7
done
clear
D)
6
done
clear
View Answer play_arrow
A 10m long wire of resistance\[20\Omega \] is connected in series with a battery of emf 3V a resistance of\[10\Omega \]. The potential gradient along the wire (in V/m) is:
A)
0.02
done
clear
B)
0.1
done
clear
C)
0.2
done
clear
D)
1.2
done
clear
View Answer play_arrow
The coordinates of a moving particle at any time t are given by \[x=\alpha {{t}^{3}}\]and\[y=\beta {{t}^{3}}\]. The speed of the particle at time t is:
A)
\[3t\sqrt{{{\alpha }^{2}}+{{\beta }^{2}}}\]
done
clear
B)
\[3{{t}^{2}}\sqrt{{{\alpha }^{2}}+{{\beta }^{2}}}\]
done
clear
C)
\[{{t}^{2}}\sqrt{{{\alpha }^{2}}+{{\beta }^{2}}}\]
done
clear
D)
\[\sqrt{{{\alpha }^{2}}+{{\beta }^{2}}}\]
done
clear
View Answer play_arrow
In an adiabatic change, the pressure and temperature of a monoatomic gas are related with relation \[P\propto {{T}^{C}}\] where, C is equal to:
A)
\[\frac{5}{4}\]
done
clear
B)
\[\frac{5}{3}\]
done
clear
C)
\[\frac{5}{2}\]
done
clear
D)
\[\frac{3}{5}\]
done
clear
View Answer play_arrow
Two coherent monochromatic beams of intensities I and 41 respectively are superposed. Then, the maximum and minimum intensities in the resulting pattern are:
A)
5I and 3I
done
clear
B)
9I and 3I
done
clear
C)
4I and I
done
clear
D)
9I and I
done
clear
View Answer play_arrow
A rod of length 10cm lies along the principal axis of a concave mirror of focal length 10cm in such a way that its end closer to the pole is 20cm away from the mirror length of image is:
A)
10cm
done
clear
B)
15cm
done
clear
C)
2.5cm
done
clear
D)
5cm
done
clear
View Answer play_arrow
A conveyor belt is moving horizontally at a speed of 4m/s. A box of mass 20kg is gently laid on it. It takes 0.1sec for the box to comes to rest, if the belt continuous to move uniformly, then the distance moved by the box on the conveyor belt is:
A)
Zero
done
clear
B)
0.2m
done
clear
C)
0.4m
done
clear
D)
0.8m
done
clear
View Answer play_arrow
The potential difference between A and B in A the following figure is.
A)
32v
done
clear
B)
48v
done
clear
C)
24v
done
clear
D)
44v
done
clear
View Answer play_arrow
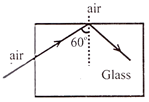
A light ray going air is incident at read of a glass fibre \[[\mu =1.5]\] making an incidence angle of \[{{60}^{{}^\circ }}\] on the lateral surface, it undergoes a TIR. How much time would it take to transverse the straight fibre of length 1km.
A)
\[3.3\mu s\]
done
clear
B)
\[5.7\mu s\]
done
clear
C)
\[6.6\mu s\]
done
clear
D)
\[3.85\mu s\]
done
clear
View Answer play_arrow
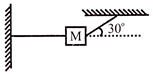
A body of mass m is placed on a rough surface with coefficient of friction \[\mu \] inclined \[\theta \]If the mass is in equilibrium, then:
A)
\[\theta ={{\tan }^{-1}}\mu \]
done
clear
B)
\[\theta ={{\tan }^{-1}}\frac{1}{\mu }\]
done
clear
C)
\[\theta ={{\tan }^{-1}}\frac{m}{\mu }\]
done
clear
D)
\[\theta ={{\tan }^{-1}}\frac{\mu }{m}\]
done
clear
View Answer play_arrow
A capacitor of capacitor C is connected with a batten- of potential V. the distance between plates is reduced to half, assuming that the battery remains the same. Then the new energy given by the battery will be:
A)
\[\frac{C{{V}^{2}}}{4}\]
done
clear
B)
\[\frac{C{{V}^{2}}}{2}\]
done
clear
C)
\[\frac{3C{{V}^{2}}}{4}\]
done
clear
D)
\[C{{V}^{2}}\]
done
clear
View Answer play_arrow
In a series LCR circuit the frequency of a 10V AC voltage source is adjusted in such a fashion that the reactance of the inductor measures\[15\Omega \] and that of the capacitor\[11\Omega \]. If\[R=3\Omega \], the potential difference across the series combination of L and C will be:
A)
8V
done
clear
B)
10V
done
clear
C)
22V
done
clear
D)
52V
done
clear
View Answer play_arrow
Two discs one of density \[7.2g/c{{m}^{3}}\]and the other of density\[8.9g/{{m}^{3}}\], are of same mass and thickness. Their moments of inertia are in the ratio:
A)
\[8.9:7.2\]
done
clear
B)
\[7.2:8.9\]
done
clear
C)
\[8.9\times 7.2:1~\]
done
clear
D)
\[1:8.9\times 7.2\]
done
clear
View Answer play_arrow
A string is wound round the rim of a mounted flywheel of mass 20kg and radius 20cm. A steady pull of 25N is applied on the cord, the angular acceleration of wheel is, [neglecting friction]
A)
\[50{{s}^{-2}}\]
done
clear
B)
\[25{{s}^{-2}}\]
done
clear
C)
\[2.5{{s}^{-2}}\]
done
clear
D)
\[6.25{{s}^{-2}}\]
done
clear
View Answer play_arrow
A thin plano-convex lens acts like a concave mirror of focal length 0.2m when silvered from its plane surface. The refractive index of the material of the lens is 1.5. The radius of curvature of the convex surface of the lens will be:
A)
0.1m
done
clear
B)
0.75m
done
clear
C)
0.4m
done
clear
D)
0.2m
done
clear
View Answer play_arrow
The relation between internal energy U, pressure F and volume V of a gas in an adiabatic process is: U = a + bPV Where a and b are positive constant. What is the value of Y:
A)
\[\frac{a}{b}\]
done
clear
B)
\[\frac{b+1}{b}\]
done
clear
C)
\[\frac{a+1}{a}\]
done
clear
D)
\[\frac{b}{a}\]
done
clear
View Answer play_arrow
A mass M is hung with light inextensible strings as shown in the figure. The tension in the horizontal string is:
A)
\[\sqrt{3}Mg\]
done
clear
B)
\[\sqrt{2}Mg\]
done
clear
C)
\[\frac{Mg}{\sqrt{3}}\]
done
clear
D)
\[\frac{Mg}{2}\]
done
clear
View Answer play_arrow
From the P-T graph what conclusion can be drawn:
A)
\[{{V}_{2}}={{V}_{1}}\]
done
clear
B)
\[{{V}_{2}}<{{V}_{1}}\]
done
clear
C)
\[{{V}_{2}}>{{V}_{1}}\]
done
clear
D)
\[None\]
done
clear
View Answer play_arrow
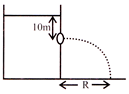
A large tank is filled water\[(density={{10}^{3}}Kg/{{m}^{3}})\]. A small hole is mode at a depth 10m below water surface. The range of water oozing out of the hole is R on ground. What extra pressure must be applied on the water surface, so that the range becomes 2R: \[(use1\,\,atm\,={{10}^{5}}Pa\,\,and\ g=10/{{s}^{2}})\]
A)
9 atm
done
clear
B)
4 atm
done
clear
C)
5 atm
done
clear
D)
3 atm
done
clear
View Answer play_arrow
An ideal transformer has a primary power input of 10kW. The secondary current when the transformer is on load is 25A. If the primary, secondary turns ratio is 8 : 1, then the potential difference applied to the primary coil is:
A)
\[\frac{{{10}^{4}}}{25\,{{(8)}^{2}}}\]
done
clear
B)
\[\frac{{{10}^{4}}}{25\,(8)}\]
done
clear
C)
\[\frac{{{(10)}^{4}}8}{25\,}\]
done
clear
D)
\[\frac{{{(10)}^{4}}\,{{(8)}^{2}}}{25\,}\]
done
clear
View Answer play_arrow
Superconductors are derived from the compounds of:
A)
p-block elements
done
clear
B)
Lanthanides
done
clear
C)
Actinides
done
clear
D)
Transition element
done
clear
View Answer play_arrow
The number of octahedral sites per sphere in fee structure is:
A)
8
done
clear
B)
4
done
clear
C)
2
done
clear
D)
1
done
clear
View Answer play_arrow
Mass percentage of benzene \[({{C}_{6}}{{H}_{6}})\] and \[(CC{{l}_{4}})\] carbon tetrachloride if 22g of benzene is dissolved in 122g of \[CC{{l}_{4}}\].
A)
84.72%
done
clear
B)
64%
done
clear
C)
92%
done
clear
D)
72%
done
clear
View Answer play_arrow
The average osmotic pressure of human blood is 7.8 bar at\[{{37}^{{}^\circ }}C\]. What is the concentration of an aqueous NaCI solution that could be used in the blood stream?
A)
0.15 mol/L
done
clear
B)
0.30 mol/L
done
clear
C)
0.60 mol/L
done
clear
D)
0.45 mol/L
done
clear
View Answer play_arrow
The rate law for reaction between substances A and B is given by \[Rate=k\,{{[A]}^{n}}\text{ }\!\![\!\!\text{ B}{{\text{ }\!\!]\!\!\text{ }}^{m}}\] On doubling the concentrations of A and halving the concentration of B, ratio of new rate to tha earlier rate will be:
A)
\[m+n\]
done
clear
B)
\[m-n\]
done
clear
C)
\[{{2}^{(n-m)}}\]
done
clear
D)
\[\frac{1}{{{2}^{(m-n)}}}\]
done
clear
View Answer play_arrow
The highest electrical conductivity of the following aqueous solutions is of:
A)
0.1 M acetic acid
done
clear
B)
0.1 M chloroacetic acid
done
clear
C)
0.1 M fluroacetic acid
done
clear
D)
0.1 M difluoroacetic acid
done
clear
View Answer play_arrow
The time taken for 90% of a first order reaction to complete is approximately:
A)
1.1 times that of half-life
done
clear
B)
2.2 times that of half-life
done
clear
C)
3.3 times that of half-life
done
clear
D)
4.4 times that of half-life
done
clear
View Answer play_arrow
The number of o-bonds in P4CL, is:
A)
6
done
clear
B)
16
done
clear
C)
20
done
clear
D)
7
done
clear
View Answer play_arrow
The basic character of the transition metal monoxide follows order:
A)
VO > CrO > TiO > FeO
done
clear
B)
CrO > VO > FeO > TiO
done
clear
C)
TiO > FeO > VO >CrO
done
clear
D)
TiO > VO > CrO > FeO
done
clear
View Answer play_arrow
Given below, catalyst and corresponding process/reaction are matched. The mismatch is:
A)
\[[RhCl\,{{(PP{{h}_{3}})}_{2}}]\]: hydrogenation
done
clear
B)
\[TiC{{l}_{4}}+Al\,{{({{C}_{2}}{{H}_{5}})}_{3}}\]: polymerization
done
clear
C)
\[{{V}_{2}}{{O}_{5}}\]: Haber - Bosch process
done
clear
D)
Nickel: Hydrogenation
done
clear
View Answer play_arrow
Silver is obtained from \[Na\,[Ag\,(C{{N}_{2}})]\] by reaction with:
A)
Fe
done
clear
B)
Na
done
clear
C)
Zn
done
clear
D)
Au
done
clear
View Answer play_arrow
Among \[A{{l}_{2}}\,{{O}_{3}}\],\[Si{{O}_{2}}\],\[{{P}_{2}}{{O}_{3}}\] and \[S{{O}_{2}}\], the correct order of acid strength is:
A)
\[S{{O}_{2}}<{{P}_{2}}{{O}_{3}}<Si{{O}_{2}}<A{{s}_{2}}{{O}_{3}}\]
done
clear
B)
\[Si{{O}_{2}}<S{{O}_{2}}<A{{s}_{2}}{{O}_{3}}<{{P}_{2}}{{O}_{3}}\]
done
clear
C)
\[A{{s}_{2}}{{O}_{3}}<Si{{O}_{2}}<S{{O}_{2}}<{{P}_{2}}{{O}_{3}}\]
done
clear
D)
\[A{{s}_{2}}{{O}_{3}}<Si{{O}_{2}}<{{P}_{2}}{{O}_{3}}<S{{O}_{2}}\]
done
clear
View Answer play_arrow
In the dichromate dianion:
A)
4 Cr - 0 bonds are equivalent
done
clear
B)
6 Cr - 0 bonds are equivalent
done
clear
C)
All Cr - 0 bonds are equivalent
done
clear
D)
All Cr - 0 bonds are non - equivalent
done
clear
View Answer play_arrow
How many EDTA (ethyenediaminetetracetic acid) molecules are required to make an octahedral complex with a \[C{{O}^{2+}}\] ion?
A)
One
done
clear
B)
Two
done
clear
C)
Six
done
clear
D)
Three
done
clear
View Answer play_arrow
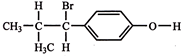
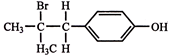
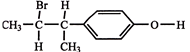
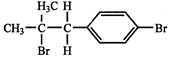
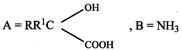
The compound, \[{{C}_{7}}{{H}_{8}}\xrightarrow{3C{{l}_{2}}/\Delta }\,A\,\xrightarrow{B{{r}_{2}}/Fe}\,B\,\xrightarrow{Zn/HCl}\,C\] The compound C is:
A)
o - Bromotoluene
done
clear
B)
m - bromotoluene
done
clear
C)
p - Bromotoluene
done
clear
D)
3 - Bromo - 2, 4, 6 - trichlorotoluene
done
clear
View Answer play_arrow
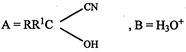
BL The reaction of:
With HBr gives predominantly
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Reduction of can be carried out with:
A)
Catalytic reduction
done
clear
B)
\[Na/{{C}_{2}}{{H}_{5}}OH\]
done
clear
C)
Wolff-kishner reduction
done
clear
D)
\[LiAl{{H}_{4}}\]
done
clear
View Answer play_arrow
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Which of the following is more basic than aniline?
A)
Benzylamine
done
clear
B)
Diphenylamine
done
clear
C)
Triphenylamine
done
clear
D)
p-nitroaniline
done
clear
View Answer play_arrow
In a set of reactions, propionic acid yielded a compound D\[C{{H}_{3}}C{{H}_{2}}COOH\xrightarrow{SOC{{l}_{2}}}\,B\,\xrightarrow{N{{H}_{3}}}\,C\,\xrightarrow{B{{r}_{2}}/KOH}\,d\] [A] The structure of D would be:
A)
\[C{{H}_{3}}C{{H}_{2}}N{{H}_{2}}\]
done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}N{{H}_{2}}\]
done
clear
C)
\[C{{H}_{3}}C{{H}_{2}}CON{{H}_{2}}\]
done
clear
D)
\[C{{H}_{3}}C{{H}_{2}}NHC{{H}_{3}}\]
done
clear
View Answer play_arrow
Subunits present in haemoglobin are:
A)
2
done
clear
B)
3
done
clear
C)
4
done
clear
D)
5
done
clear
View Answer play_arrow
Nylon - 6 is made from:
A)
1, 3-Butadiene
done
clear
B)
Chloroprene
done
clear
C)
Adipic acid
done
clear
D)
Caprolectam
done
clear
View Answer play_arrow
Among the following sweeteners which one has the lowest sweetness value?
A)
Alitame
done
clear
B)
Aspartame
done
clear
C)
Saccharin
done
clear
D)
Sucralose
done
clear
View Answer play_arrow
The isoelectronic pair is:
A)
\[C{{l}_{2}}\,O,\,ICl_{2}^{-}\]
done
clear
B)
\[ICl_{2}^{-},Cl{{O}_{2}}\]
done
clear
C)
\[IF_{2}^{+},I_{3}^{+}\]
done
clear
D)
\[ClO_{2}^{-},ClF_{2}^{+}\]
done
clear
View Answer play_arrow
In the reaction \[4N{{H}_{3}}(g)+{{50}_{2}}(g)\to 4NO\,(g)+6{{H}_{2}}O\,(l)\] When 1 mole of ammonia and 1 mole of \[{{O}_{2}}\] are made to react to completion:
A)
1.0 mole of \[{{H}_{2}}O\] is produced
done
clear
B)
1.0 mole of NO will be produced
done
clear
C)
All the oxygen will be consumed
done
clear
D)
All the ammonia will be consumed
done
clear
View Answer play_arrow
The ratio of area covered by second orbital to the first orbital is:
A)
1:1
done
clear
B)
1:16
done
clear
C)
8:1
done
clear
D)
16:1
done
clear
View Answer play_arrow
The ionic radii of isoelectronic species \[{{N}^{3-}}\],\[{{O}^{2-}}\], and F in \[\overset{o}{\mathop{A}}\,\] are in the order:
A)
1.36, 1.40, 1.71
done
clear
B)
1.36, 1.71, 1.40
done
clear
C)
1.71, 1.40, 1.36
done
clear
D)
1.71, 1.36, 1.40
done
clear
View Answer play_arrow
In which of the following molecular / ions are all the bonds not equal?
A)
\[Xe{{F}_{4}}\]
done
clear
B)
\[BF_{4}^{-}\]
done
clear
C)
\[SF_{4}^{{}}\]
done
clear
D)
\[SiF_{4}^{{}}\]
done
clear
View Answer play_arrow
AG for a reaction is 46.06 kcal/mole, K for the reaction at 300k is:
A)
\[{{10}^{8}}\]
done
clear
B)
\[{{10}^{2222}}\]
done
clear
C)
\[{{10}^{33.33}}\]
done
clear
D)
\[None\]
done
clear
View Answer play_arrow
How many bonds are there in
A)
\[14\sigma ,8\pi \]
done
clear
B)
\[18\sigma ,8\pi \]
done
clear
C)
\[19\sigma ,4\pi \]
done
clear
D)
\[14\sigma ,2\pi \]
done
clear
View Answer play_arrow
Containers A and B have same gaseous, pressure, volume and temperature of A are all twice that of B. Then ratio of the number of molecules of A and B are:
A)
1:2
done
clear
B)
2:1
done
clear
C)
1:4
done
clear
D)
4:1
done
clear
View Answer play_arrow
Among the compounds \[B{{F}_{3}}\], \[NC{{l}_{3}}\],\[{{H}_{2}}S\],\[S{{F}_{4}}\], and \[BeC{{l}_{2}}\] identify the ones in which, the central atom has the same type of hybridisation:
A)
\[B{{F}_{3}},NC{{l}_{3}}\And {{H}_{2}}S\]
done
clear
B)
\[{{H}_{2}}S\,\,andBeC{{l}_{2}}\]
done
clear
C)
\[NC{{l}_{3}}\,\,and\,{{H}_{2}}S\]
done
clear
D)
\[S{{F}_{4}}\,\,and\,BeC{{l}_{2}}\]
done
clear
View Answer play_arrow
In a regular octahedral molecule, \[M{{x}_{6}},\] the number X-M-X bonds at \[180{}^\circ \]is:
A)
Three
done
clear
B)
Two
done
clear
C)
Six
done
clear
D)
Four
done
clear
View Answer play_arrow
For the equilibrium \[MgC{{O}_{3}}(s)MgO(s)+C{{O}_{2}}(g)\] Which of the following expressions is correct?
A)
\[{{K}_{p}}=\frac{{{P}_{Mgo}}K\times {{P}_{C{{O}_{2}}}}}{{{P}_{MgC{{O}_{3}}}}}\]
done
clear
B)
\[{{K}_{p}}=\frac{[MgO][C{{O}_{2}}]}{[MgC{{O}_{3}}]}\]
done
clear
C)
\[{{K}_{p}}=\frac{{{P}_{Mg}}_{O}\times {{P}_{C{{O}_{2}}}}}{{{P}_{Mgc{{o}_{3}}}}}\]
done
clear
D)
\[{{K}_{p}}={{P}_{C{{O}_{2}}}}\]
done
clear
View Answer play_arrow
pH of water is 7. When a substance Y is dissolved in water, the pH becomes 13. The substance Y is a salt of:
A)
Weak acid and weak base
done
clear
B)
Strong acid and strong base
done
clear
C)
Strong acid and weak base
done
clear
D)
Weak acid and strong base
done
clear
View Answer play_arrow
For decolourisation of 1 mole of \[KMn{{O}_{4}}\], the moles of \[{{H}_{2}}{{O}_{2}}\] required is:
A)
1/2
done
clear
B)
3/2
done
clear
C)
5/2
done
clear
D)
7/2
done
clear
View Answer play_arrow
The structure of \[{{H}_{2}}{{O}_{2}}\] is:
A)
Planar
done
clear
B)
Non-planar
done
clear
C)
Spherical
done
clear
D)
Linear
done
clear
View Answer play_arrow
Photoelectric effect is maximum in:
A)
Cs
done
clear
B)
Na
done
clear
C)
K
done
clear
D)
Li
done
clear
View Answer play_arrow
The IUPAC name for the formula:
A)
2-methyl-2-butenoic acid
done
clear
B)
3-methyl-3-butenoic acid
done
clear
C)
3-methyl-2-butenoic acid
done
clear
D)
2-methyl-3-butenoic acid
done
clear
View Answer play_arrow
The compound \[C{{H}_{3}}-O-{{C}_{3}}{{H}_{7}}\] and \[{{C}_{2}}{{H}_{5}}-O-{{C}_{5}}{{H}_{5}}\] exhibit:
A)
Optical isomerism
done
clear
B)
Cis-trans isomerism
done
clear
C)
Metamerism
done
clear
D)
Chain isomerism
done
clear
View Answer play_arrow
Amorphous form of silica is:
A)
Triclymite
done
clear
B)
Cristobalite
done
clear
C)
Fumed silica
done
clear
D)
Asbestos
done
clear
View Answer play_arrow
Among the following the aromatic compound is:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
\[CH\equiv CH\xrightarrow{{{O}_{3}}}\xrightarrow{Zn/C{{H}_{3}}OH}Y,Y\]
A)
\[C{{H}_{2}}OH-C{{H}_{2}}OH\]
done
clear
B)
\[C{{H}_{3}}COOH\]
done
clear
C)
\[{{C}_{2}}{{H}_{5}}OH\]
done
clear
D)
\[C{{H}_{3}}-C{{H}_{3}}\]
done
clear
View Answer play_arrow
The ammonia evolved from the treatment of 0.30g of an organic compound for the estimation of nitrogen was passed in 100ml of 0.1 M Sulphuric acid. The excess of acid required 20ml of 0.5 M sodium hydroxide solution for complete neutraitization. The organic compound is:
A)
Acetamide
done
clear
B)
Benzamide
done
clear
C)
Urea
done
clear
D)
Thiourea
done
clear
View Answer play_arrow
In antartica, ozone depletion is due to the formation of the following compound:
A)
Acrolein
done
clear
B)
Peroxyaceltylnitrate
done
clear
C)
\[S{{O}_{2}}\,and\,S{{O}_{3}}\]
done
clear
D)
Chlorine nitrate
done
clear
View Answer play_arrow
Which of the following is the relatively most accurate method for dating of fossils?
A)
Radio - carbon method
done
clear
B)
Potassium - organ method
done
clear
C)
Electron - spin resonance method
done
clear
D)
Uranium - lead method
done
clear
View Answer play_arrow
Lactose is composed of:
A)
Glucose + glucose
done
clear
B)
Glucose + Fructose
done
clear
C)
K c. Fructose + galactose
done
clear
D)
Glucose + galactose
done
clear
View Answer play_arrow
Genes are packaged into a bacterial chromosome by:
A)
Histones
done
clear
B)
Acidic protein
done
clear
C)
Basic proteins
done
clear
D)
Actin
done
clear
View Answer play_arrow
Which of the following is the simplest amino acid?
A)
Alanine
done
clear
B)
Glycine
done
clear
C)
Asparagene
done
clear
D)
Tyrosine
done
clear
View Answer play_arrow
Ribosomes were discovered by:
A)
Golgi
done
clear
B)
De Roberti's
done
clear
C)
Porter
done
clear
D)
Palade
done
clear
View Answer play_arrow
Resing and terpentine are obtained from:
A)
Cycas
done
clear
B)
Pinus
done
clear
C)
Cedrus
done
clear
D)
Abies
done
clear
View Answer play_arrow
Largest sperms in the plant world are found in:
A)
Pinus
done
clear
B)
Banyan
done
clear
C)
Cycas
done
clear
D)
Tsuja
done
clear
View Answer play_arrow
The antherozoids of Funaria are:
A)
Aciliated
done
clear
B)
Biflagellated
done
clear
C)
Multiciliated
done
clear
D)
Monociliated
done
clear
View Answer play_arrow
Tube feet occurs in:
A)
Cockroach
done
clear
B)
Starfish
done
clear
C)
Cuttle fish
done
clear
D)
Cat fish
done
clear
View Answer play_arrow
Which of the following is not found in birds?
A)
Hindlimb
done
clear
B)
Pectoral girdle
done
clear
C)
Pelvic girdle
done
clear
D)
fore limb
done
clear
View Answer play_arrow
Tetradynamous stamens are found in family:
A)
Malavaceae
done
clear
B)
Solanaceae
done
clear
C)
Cruciferae
done
clear
D)
Lililaceae
done
clear
View Answer play_arrow
Edible part in litchi ia:
A)
Mesocarp
done
clear
B)
Fleshy aril
done
clear
C)
Endosperm
done
clear
D)
Peri corp
done
clear
View Answer play_arrow
Geocarpic fruit is:
A)
Potato
done
clear
B)
Ground nut
done
clear
C)
Ordon
done
clear
D)
Garlic
done
clear
View Answer play_arrow
Angiosperm to which the largest flowers belongs is:
A)
Total stem parasite
done
clear
B)
Partial stem parasite
done
clear
C)
Total root parasite
done
clear
D)
Partial root parasite
done
clear
View Answer play_arrow
Edible part in mango is:
A)
Mesocarp
done
clear
B)
Epicarp
done
clear
C)
Endocarp
done
clear
D)
Epidermis
done
clear
View Answer play_arrow
An example of axil placentation is:
A)
Argemone
done
clear
B)
Dianthus
done
clear
C)
Lemon
done
clear
D)
Marigold
done
clear
View Answer play_arrow
Bicarpellary gynoecium and oblique ovary occurs in:
A)
Mustard
done
clear
B)
Banana
done
clear
C)
Pisum
done
clear
D)
Brinjal
done
clear
View Answer play_arrow
Hypanthodium is:
A)
Thalamus
done
clear
B)
Fruit
done
clear
C)
Inflorescence
done
clear
D)
Ovary
done
clear
View Answer play_arrow
Which element is located at the centre of the porphyrin ring in chlorophyll?
A)
Manganese
done
clear
B)
Calcium
done
clear
C)
Magnesium
done
clear
D)
Potassium
done
clear
View Answer play_arrow
Stomata of CAM plants:
A)
Never open
done
clear
B)
Are always open
done
clear
C)
Open during the day time and closed at night
done
clear
D)
Open during the night and closed during the day.
done
clear
View Answer play_arrow
Krebs cycle occurs in:
A)
Mitochondria
done
clear
B)
Cytoplasm
done
clear
C)
Chloroplast
done
clear
D)
Ribosomes
done
clear
View Answer play_arrow
Ranthambore national park is situated in:
A)
Maharashtra
done
clear
B)
Rajasthan
done
clear
C)
Gujarat
done
clear
D)
Uttar Pradesh
done
clear
View Answer play_arrow
Deforestation does not lead to:
A)
Quick nutrient cycling
done
clear
B)
Soil erosion
done
clear
C)
Alteration of local weather conditions
done
clear
D)
Destruction of natural habitat of wild animals.
done
clear
View Answer play_arrow
Which one of the following is an example of ex-situ conservation?
A)
Wild life sancturary
done
clear
B)
Seed bank
done
clear
C)
Sacred groves
done
clear
D)
National park
done
clear
View Answer play_arrow
Land mass occupied by forests is about:
A)
60%
done
clear
B)
30%
done
clear
C)
22%
done
clear
D)
117o
done
clear
View Answer play_arrow
Glucose is taken back from glomerular filtrate through:
A)
Active transport
done
clear
B)
Passive transport
done
clear
C)
Osmosis
done
clear
D)
Diffusion
done
clear
View Answer play_arrow
Uric acid is the chief nitrogenous component of the excretory products of:
A)
Man
done
clear
B)
Earthworm
done
clear
C)
Cockroach
done
clear
D)
Frog
done
clear
View Answer play_arrow
Single - celled eukaryotes are included in:
A)
Protista
done
clear
B)
Fungi
done
clear
C)
Archaea
done
clear
D)
Monera
done
clear
View Answer play_arrow
Apomictic embryos in citrus arise from:
A)
Synergids
done
clear
B)
Maternal sporophytic tissue in ovule
done
clear
C)
Antipodal cells
done
clear
D)
Diploid egg
done
clear
View Answer play_arrow
Ringworm in Humans is caused by:
A)
Bacteria
done
clear
B)
Fungi
done
clear
C)
Nematodes
done
clear
D)
Viruses
done
clear
View Answer play_arrow
Widal test is used for the dignosis of:
A)
Malaria
done
clear
B)
Pneumonia
done
clear
C)
Tubeculosis
done
clear
D)
Typhoid
done
clear
View Answer play_arrow
Low \[C{{a}^{2+}}\] in the body fluid may be the cause of:
A)
Tetany
done
clear
B)
Anaemia
done
clear
C)
Angina pectoris
done
clear
D)
Gout
done
clear
View Answer play_arrow
Viruses envelope is known as:
A)
Capsid
done
clear
B)
Virion
done
clear
C)
Nucleo protein
done
clear
D)
Core
done
clear
View Answer play_arrow
The part of fallopian tube closest to the ovary is:
A)
Isthmus
done
clear
B)
Infundibulum
done
clear
C)
Cervix
done
clear
D)
Ampulla
done
clear
View Answer play_arrow
The first movements of the fetus and appearance of the hair on its head are usually observed during which month of pregnancy?
A)
Fourth month
done
clear
B)
Fifth month
done
clear
C)
Sixth month
done
clear
D)
Third month
done
clear
View Answer play_arrow
Vasa efferent are the ductless leading from:
A)
Testicular lobules to rete testis
done
clear
B)
Rete testis to vas deferens
done
clear
C)
Vas deference to epididymis
done
clear
D)
Epididymis to urethra
done
clear
View Answer play_arrow
Gas released during Bhopal tragedy was:
A)
Methyl isocyanate
done
clear
B)
Carbon monoxide
done
clear
C)
Sodium isothiocyanate
done
clear
D)
Chlorofluoro - carbon
done
clear
View Answer play_arrow
Pyramid of numbers in a grass land / true ecosystem is:
A)
Always inverted
done
clear
B)
Always upright
done
clear
C)
Both and
done
clear
D)
Spindle - shaped
done
clear
View Answer play_arrow
River water deposits:
A)
Loamy soil
done
clear
B)
Alluvial soil
done
clear
C)
Laterite soil
done
clear
D)
Sandy soil
done
clear
View Answer play_arrow
In a biotic community, the most important factor for survival of an animal is:
A)
Day length
done
clear
B)
Soil moisture
done
clear
C)
Green food
done
clear
D)
Predators
done
clear
View Answer play_arrow
Which of the following is not used for disinfection of drinking water:
A)
Phenyl
done
clear
B)
Chloramine
done
clear
C)
Chlarine
done
clear
D)
Ozone
done
clear
View Answer play_arrow
Frequency of an allele in an isolated population may be change due to:
A)
Genetic drift
done
clear
B)
Gene flow
done
clear
C)
Mutation
done
clear
D)
Natural selection
done
clear
View Answer play_arrow
What is the most important factor for the success of animal population?
A)
Natality
done
clear
B)
Unlimited food
done
clear
C)
Adaptability
done
clear
D)
Inter-species activity
done
clear
View Answer play_arrow
What is the best pH of the soil for cultivation of plants?
A)
3.4 - 5.4
done
clear
B)
6.5 - 7.5
done
clear
C)
4.5 - 8.5
done
clear
D)
5.5 - 6.5
done
clear
View Answer play_arrow
Introduction of food plants developed by genetic engineering is not desirable because:
A)
Economy of developing countries may
done
clear
B)
These products are less tasty as compared to the already existing products
done
clear
C)
This method is costly
done
clear
D)
There is danger of introduction viruses and toxins with introduced drop
done
clear
View Answer play_arrow
Which meristem helps in increasing girth?
A)
Lateral meristem
done
clear
B)
Intercalary meristem
done
clear
C)
Primary meristem
done
clear
D)
Apical meristem
done
clear
View Answer play_arrow
Angular collenchyma occurs in:
A)
Cucurbita
done
clear
B)
Tagetes
done
clear
C)
Althaea
done
clear
D)
Salvia
done
clear
View Answer play_arrow
Main function of lenticel is:
A)
Transpiration
done
clear
B)
Guttation
done
clear
C)
Gaseous
done
clear
D)
Bleeding
done
clear
View Answer play_arrow
Axillary bud and terminal bud are derived from the activity of:
A)
Lateral meristem
done
clear
B)
Intercalary meristem
done
clear
C)
Apical meristem
done
clear
D)
Parenchyma
done
clear
View Answer play_arrow
In a woody dicotyledonous tree, which of the following parts will mainly consists of primary tissues?
A)
All parts
done
clear
B)
Stem & Root
done
clear
C)
Flowers, Fruits and Leaves
done
clear
D)
Shoot tips and root tips
done
clear
View Answer play_arrow
In a terrestrial habitat which of the following is affected by temperature and rain fall condition?
A)
Translocation
done
clear
B)
Transpiration
done
clear
C)
Transformation
done
clear
D)
Thermo denaturation
done
clear
View Answer play_arrow
A plant requires magnesium for:
A)
Holding cells together
done
clear
B)
Protein synthesis
done
clear
C)
Chlorophyll synthesis
done
clear
D)
Cell wall development
done
clear
View Answer play_arrow
Leaf fall can be prevented with the help of:
A)
Abscisic acid
done
clear
B)
Auxins
done
clear
C)
Gibberellins
done
clear
D)
Cytokinins
done
clear
View Answer play_arrow
The response of different organisms to environmental rhythms of light and darkness is called:
A)
Phototaxis
done
clear
B)
Photoperiodism
done
clear
C)
Phototropism
done
clear
D)
Vernalization
done
clear
View Answer play_arrow
An improved variety of transgenic basmati rice:
A)
Does not require chemical fertilizers and growth hormones
done
clear
B)
Gives high yield and is rich in vitamin-A
done
clear
C)
Is completely resistant to all insect pests and diseases of paddy
done
clear
D)
Gives high yield but has no characteristic aroma
done
clear
View Answer play_arrow
Some of the characteristic of Bt. Cotton are:
A)
Long fibre and resistance to aphids
done
clear
B)
Medium yield, long fibre and resistance to battle pests
done
clear
C)
High yield and production of toxic protein crystals which kill dipteran pests
done
clear
D)
High yield and resistance to bollworms
done
clear
View Answer play_arrow
Breeding of crops with high levels of minerals, vitamins and proteins is called:
A)
Somatic hybridization
done
clear
B)
Bio fortification
done
clear
C)
Bio magnification
done
clear
D)
Micro propagation
done
clear
View Answer play_arrow
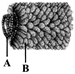
Identify the virus and name the structures A and B:
A)
TMV, A = ssRNA, B = Capsid
done
clear
B)
TMV, A = dsRNA, B = Capsid
done
clear
C)
TMV, A = Capsid, B =ssRNA
done
clear
D)
TMV, A = Capsid, B =dsRNA
done
clear
View Answer play_arrow
During on injury nasal septum gets damaged and for its recovery which cartilage is preferred?
A)
Hyaline cartilage
done
clear
B)
Elastic cartilage
done
clear
C)
Calcified cartilage
done
clear
D)
Fibrous cartilage
done
clear
View Answer play_arrow
The vitamin-C or ascorbic acid prevents:
A)
Rickets
done
clear
B)
Pellagra
done
clear
C)
Scurvy
done
clear
D)
Antibody synthesis
done
clear
View Answer play_arrow
Which one of the following is fat-soluble vitamin and its related deficiency disease?
A)
As corbie acid - Scurvy
done
clear
B)
Cobalamine - Beri-Beri
done
clear
C)
Retinol - Xerophthalmia
done
clear
D)
Calciferol - Pellagra
done
clear
View Answer play_arrow
Haploid plant cultures are got from:
A)
Leaves
done
clear
B)
Root tip
done
clear
C)
Pollen grain
done
clear
D)
Buds
done
clear
View Answer play_arrow
Endosperm is consumed by developing embryo is the seed of:
A)
Coconut
done
clear
B)
Castor
done
clear
C)
Pea
done
clear
D)
Maize
done
clear
View Answer play_arrow
Which one of the following is surrounded by a callose wall?
A)
Microspore mother cell
done
clear
B)
Male gametes
done
clear
C)
Egg
done
clear
D)
Pollen grain
done
clear
View Answer play_arrow
In the diagram given below, some of the algae have been labelled as 'A', 'B', 'C', 'D' and 'E'. These algae are respectively identified as:
A)
Dictyota, Polysiphonia, Porphyra, Fucus and Laminaria
done
clear
B)
Porphyra, Dictyota, Laminaria, Fucus and Polysiphonia
done
clear
C)
Dictyota, Polysiphonia, Porphyra, Laminaria and Fucus
done
clear
D)
Fucus, Porphyra, Dictyota, Polysiphonia and Laminaria
done
clear
View Answer play_arrow
Pollination occurs in:
A)
Bryophytes and angiosperms
done
clear
B)
Pteridophytes and angiosperms
done
clear
C)
Angiosperms and gymnosperms
done
clear
D)
Angiosperms and fungi
done
clear
View Answer play_arrow
DNA replication is:
A)
Conservative and discontinuous
done
clear
B)
Semi-conservative and semi discontinuous
done
clear
C)
Semi-conservative and discontinuous
done
clear
D)
Conservative
done
clear
View Answer play_arrow
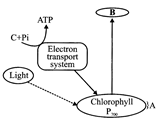
Observe the diagram given below and identify A. B and C:
A)
\[PS-II~~~~~~{{e}^{-}}acceptor~~~~~~ADP\]
done
clear
B)
\[ADP~~~~~~{{e}^{-}}acceptor~~~~~~PS-II\]
done
clear
C)
\[{{e}^{-}}\text{ }acceptor\text{ }PS-I~~~~~~~~~~~~ADP\]
done
clear
D)
\[PS-I~~~~~~~{{e}^{-}}acceptor~~~~~~ADP\]
done
clear
View Answer play_arrow
A dihybrid condition is:
A)
tt Rr
done
clear
B)
Tt rr
done
clear
C)
tt rr
done
clear
D)
Tt Rr
done
clear
View Answer play_arrow
An allele is dominant if it is expressed in:
A)
Both homozygous and heterozygous states
done
clear
B)
Second generation
done
clear
C)
Heterozygous combination
done
clear
D)
Nonozygous combination
done
clear
View Answer play_arrow
An organism with two identical alleles is:
A)
Dominant
done
clear
B)
Hybrid
done
clear
C)
Heterozygous
done
clear
D)
Homozygous
done
clear
View Answer play_arrow
Who proved that DNA is basic genetic material?
A)
Griffith
done
clear
B)
Boveri and Sutton
done
clear
C)
Watson
done
clear
D)
Hershey anbd chose
done
clear
View Answer play_arrow
The translation termination triplet is:
A)
UAU
done
clear
B)
UAA
done
clear
C)
UAC
done
clear
D)
UGC
done
clear
View Answer play_arrow
An enzyme that joins the ends of two strands of nucleic acid is a:
A)
Polymerase
done
clear
B)
Synthetase
done
clear
C)
Helicase
done
clear
D)
Ligase
done
clear
View Answer play_arrow
In a DNA percentage of thymine is 20. What is the percentage of guanine?
A)
20%
done
clear
B)
40%
done
clear
C)
30%
done
clear
D)
60%
done
clear
View Answer play_arrow
The book "Genera Plantarum" was written by:
A)
Bessey
done
clear
B)
Hutchinson
done
clear
C)
Engler and Pranh
done
clear
D)
Bentham and Hooker
done
clear
View Answer play_arrow
Tobacco mosaic virus is a tubular filament of size:
A)
\[700\times 300nm~~\]
done
clear
B)
\[300\times 10\text{ }nm\]
done
clear
C)
\[300\times 5nm\]
done
clear
D)
\[300\times 18nm\]
done
clear
View Answer play_arrow
The genotype of B group father of an group child is:
A)
\[{{I}^{O}}{{I}^{B}}\]
done
clear
B)
\[{{I}^{B}}{{I}^{B}}\]
done
clear
C)
\[{{I}^{A}}{{I}^{B}}\]
done
clear
D)
\[{{I}^{O}}{{I}^{B}}\]
done
clear
View Answer play_arrow
Law \[C{{a}^{2+}}\] in the body fluid may be the cause of:
A)
Tetany
done
clear
B)
Anaemia
done
clear
C)
Angina pectoris
done
clear
D)
Gout
done
clear
View Answer play_arrow
Which one of the following symbols and its representation used in human pedigree analysis is correct:
A)
D =0 = Mating between relatives
done
clear
B)
0 = Unaffected male
done
clear
C)
0 = Unaffected female
done
clear
D)
W = Male affected
done
clear
View Answer play_arrow
"Continuity of germplasm" theory was given by:
A)
De vries
done
clear
B)
Weismann
done
clear
C)
Darwin
done
clear
D)
Lamarck
done
clear
View Answer play_arrow
Enzymes are absent in:
A)
Algae
done
clear
B)
Fungi
done
clear
C)
Cyanobacteria
done
clear
D)
Viruses
done
clear
View Answer play_arrow
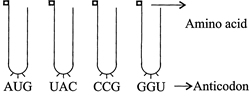
Find the sequence of binding of the following amino acyi t-RNA complexes during translation to a m-RNA transcribed by a DNA segment having the base sequence 3' TAC ATG GGT CCG 5'. Choose the answer showing the correct order of numerals: Amino acid
A)
ii, i, iv, iii
done
clear
B)
i, ii, iv, iii
done
clear
C)
ii, i, iii, iv
done
clear
D)
i, ii, iii, iv
done
clear
View Answer play_arrow
Which one of the following is a skull bone?
A)
Atlas
done
clear
B)
Coracoid
done
clear
C)
Arytenoid
done
clear
D)
Pterygoid
done
clear
View Answer play_arrow
Parkinson's disease (characterized by tremors and progressive rigidity of limbs) is caused by degeneration of brain neurons that are involved in movement control and make use of neurotransmitter:
A)
Acetylcholine
done
clear
B)
No repine phrine
done
clear
C)
Dopamine
done
clear
D)
GABA
done
clear
View Answer play_arrow
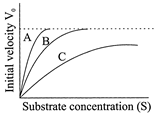
Figure given below shows three velocity -substrate concentration curves for an enzyme reaction. What do the curves depict?
A)
A = Normal enzyme action, B = competitive inhibition, C = Non-competitive inhibition
done
clear
B)
A = Enzyme with an allosteric modulator added B = Normal enzyme activity, C =Competitive inhibition.
done
clear
C)
A = Enzyme with an allosteric stimulator, B = Competitive inhibitor added, C = Normal enzyme reaction
done
clear
D)
A = Normal enzyme reaction, B = Non-competitive inhibitor added, C = Allosteric inhibitor added
done
clear
View Answer play_arrow
What does 'T' stands for in DPT vaccine?
A)
Tuberculosis
done
clear
B)
Typhoid
done
clear
C)
Trachoma
done
clear
D)
Tetanus
done
clear
View Answer play_arrow
Which animal has the longest gestation period?
A)
Shark
done
clear
B)
Walrus
done
clear
C)
Elephant
done
clear
D)
None of These
done
clear
View Answer play_arrow
Cell theory was proposed by:
A)
A botanist
done
clear
B)
Zoologist
done
clear
C)
A botanist and a zoologist
done
clear
D)
A psychologist
done
clear
View Answer play_arrow
PS-II occurs only in:
A)
Stroma
done
clear
B)
Granal thylakoids
done
clear
C)
Stromal lamella
done
clear
D)
Matrix
done
clear
View Answer play_arrow

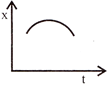
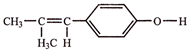 With HBr gives predominantly
With HBr gives predominantly