A) \[l_{3}^{-}\]
B) \[l_{2}^{+}\]
C) \[{{H}_{2}}O\]
D) \[S{{O}_{2}}\]
Correct Answer: A
Solution :
The structure of \[I_{3}^{\Theta }\] is determined as follows \[I_{3}^{\Theta }\] has 2 bond pairs and 3 lone pairs. According to VSEPR theory, geometry is linear and can be represented as follows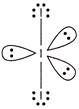 \[Geometry = linear\] \[\operatorname{Hybridisation}=s{{p}^{3}}d\]
\[Geometry = linear\] \[\operatorname{Hybridisation}=s{{p}^{3}}d\]
You need to login to perform this action.
You will be redirected in
3 sec
