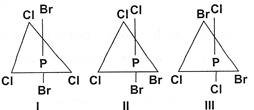
| (i) Liquid at room temperature. |
| (ii) Decolourises aqueous bromine. |
| (iii) Undergoes addition polymerization. |
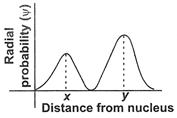 If the above radial probability curve indicates 2s orbital, the distance between the peak points x and y is
If the above radial probability curve indicates 2s orbital, the distance between the peak points x and y is
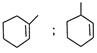
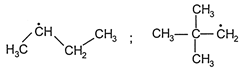
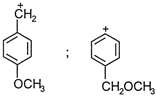
 The product (X) is
The product (X) is
You need to login to perform this action.
You will be redirected in
3 sec
