| Direction: An ideal diatomic gas is confined in a cylinder A of volume \[{{V}_{0}},\] this cylinder is connected to another cylinder B with the help of tube of a negligible volume. The cylinder B is fitted with a movable piston which can be adjusted from outside. Initially, the piston is adjusted so that volume of B is the same as volume of A i.e., \[{{V}_{0}}\]. B is evacuated and the stopcork is opened so that gas expands and occupies the volume \[2{{V}_{0}}\]. [System is thermally isolated from the surroundings]. |
 |
| Work done on the gas is [for n moles of gas] |
A) \[nRT\text{ }\ln \text{ }2\]
B) \[-nRT\text{ }\ln \text{ }2\]
C) \[nRT\]
D) \[-nRT\]
Correct Answer: A
Solution :
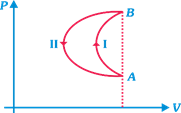
| [a]During compression, process is slow so it is isothermal at constant temperature T. |
| So, work done, |
| \[W\,=\,nRT\,\ln \,\frac{{{V}_{f}}}{{{V}_{i}}}\,=-nRT\,\,\ln \,\,2\] |
| This is the work done by the system on the surrounding, so work done by the surroundings on the system in nRT ln 2. |
You need to login to perform this action.
You will be redirected in
3 sec
