A) \[3.81\times {{10}^{5}}N/{{m}^{2}}\]
B) \[6\times {{10}^{6}}N/{{m}^{2}}\]
C) \[5\times {{10}^{7}}N/{{m}^{2}}\]
D) \[4.6\times {{10}^{2}}N/{{m}^{2}}\]
Correct Answer: A
Solution :
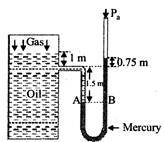
[a]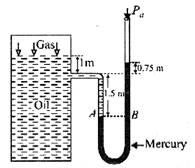 Suppose \[{{P}_{gas}}\] is the pressure of the gas on the oil. As the points A and B are at the same level in the mercury columns, so \[{{P}_{A}}={{P}_{B}}\] or \[{{P}_{gas}}+{{\rho }_{oil}}g{{h}_{oil}}={{P}_{a}}+{{\rho }_{Hg}}g{{h}_{^{Hg}}}\] or \[{{P}_{gas}}+820\times 9.8\times (1+1.50)\] \[={{P}_{a}}+13.6\times {{10}^{3}}\times 9.8\times (1.5+0.75)\] or \[{{P}_{gas}}+20.09\times {{10}^{3}}={{P}_{a}}+299.88\times {{10}^{3}}\] \[\therefore \,{{P}_{gas}}-Pa=299.88\times {{10}^{3}}-20.09\times {{10}^{3}}\] or \[{{[{{P}_{gas}}]}_{gauge}}=279.8\times {{10}^{3}}N/{{m}^{2}}=2.8\times {{10}^{5}}N/{{m}^{2}}\] Absolute pressure of gas \[{{[{{P}_{gas}}]}_{gauge}}={{[{{P}_{gas}}]}_{gauge}}+{{P}_{a}}\] \[=2.8\times {{10}^{5}}+1.01\times {{10}^{5}}=3.81\times {{10}^{5}}N/{{m}^{2}}\]
Suppose \[{{P}_{gas}}\] is the pressure of the gas on the oil. As the points A and B are at the same level in the mercury columns, so \[{{P}_{A}}={{P}_{B}}\] or \[{{P}_{gas}}+{{\rho }_{oil}}g{{h}_{oil}}={{P}_{a}}+{{\rho }_{Hg}}g{{h}_{^{Hg}}}\] or \[{{P}_{gas}}+820\times 9.8\times (1+1.50)\] \[={{P}_{a}}+13.6\times {{10}^{3}}\times 9.8\times (1.5+0.75)\] or \[{{P}_{gas}}+20.09\times {{10}^{3}}={{P}_{a}}+299.88\times {{10}^{3}}\] \[\therefore \,{{P}_{gas}}-Pa=299.88\times {{10}^{3}}-20.09\times {{10}^{3}}\] or \[{{[{{P}_{gas}}]}_{gauge}}=279.8\times {{10}^{3}}N/{{m}^{2}}=2.8\times {{10}^{5}}N/{{m}^{2}}\] Absolute pressure of gas \[{{[{{P}_{gas}}]}_{gauge}}={{[{{P}_{gas}}]}_{gauge}}+{{P}_{a}}\] \[=2.8\times {{10}^{5}}+1.01\times {{10}^{5}}=3.81\times {{10}^{5}}N/{{m}^{2}}\]
You need to login to perform this action.
You will be redirected in
3 sec
