| Study the given figure carefully. |
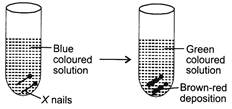
|
| Which of the following reactions explains the above change most appropriately? |
A) \[ZnS{{O}_{4}}+Cu\to CuS{{O}_{4}}+Zn\]
B) \[CuS{{O}_{4}}+Fe\to FeS{{O}_{4}}+Cu\]
C) \[FeS{{O}_{4}}+Cu\to CuS{{O}_{4}}+Fe\]
D) \[CuS{{O}_{4}}+Zn\to ZnS{{O}_{4}}+Cu\]
Correct Answer: B
Solution :
The blue coloured solution is \[CuS{{O}_{4}}\]solution. Iron (Fe) is more reactive than copper \[(Cu)\]. So, Fe displaces Cu from the \[CuS{{O}_{4}}\]solution and forming \[FeS{{O}_{4}}\] solution which is green in colour. Brown-red deposition in test tube is of copper. Such type of reactions are called displacement reactions.You need to login to perform this action.
You will be redirected in
3 sec
