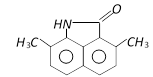
 is [IIT-JEE Screening 2004]
is [IIT-JEE Screening 2004]
A)
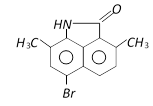
B)
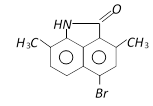
C)
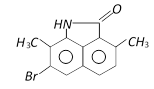
D)
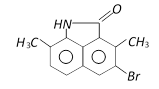
Correct Answer: A
Solution :
The phenyl ring having H - N< group is activated while another one is deactivated due to \[-\underset{O}{\mathop{\underset{||}{\mathop{C}}\,}}\,-\], so electrophilic aromatic bromination will occur at para position with respect to H - N< group inactivated ring.You need to login to perform this action.
You will be redirected in
3 sec
