-
question_answer1)
Which of the following represents a redox reaction?
A)
\[NaOH+HCl\xrightarrow{{}}NaCl+{{H}_{2}}O\] done
clear
B)
\[BaC{{l}_{2}}+{{H}_{2}}S{{O}_{4}}\xrightarrow{{}}BaS{{O}_{4}}+2HCl\] done
clear
C)
\[CuS{{O}_{4}}+2{{H}_{2}}O\xrightarrow{{}}Cu{{(OH)}_{2}}+{{H}_{2}}S{{O}_{4}}\] done
clear
D)
\[Zn+2HCl\xrightarrow{{}}ZnC{{l}_{2}}+{{H}_{2}}\] done
clear
View Solution play_arrow
-
question_answer2)
Which reaction involves neither oxidation nor reduction?
A)
\[CrO_{4}^{2-}\xrightarrow{{}}C{{r}_{2}}O{{_{7}^{2-}}^{~}}\] done
clear
B)
\[Cr\xrightarrow{{}}CrC{{l}_{3}}\] done
clear
C)
\[Na\xrightarrow{{}}N{{a}^{+}}\] done
clear
D)
\[2{{S}_{2}}O_{3}^{2-}\xrightarrow{{}}{{S}_{4}}O_{6}^{2-}\] done
clear
View Solution play_arrow
-
question_answer3)
Zn gives \[{{H}_{2}}\] gas with \[{{B}_{2}}S{{O}_{4}}\] and \[HCl\] but not with \[HN{{O}_{3}}\] because
A)
Zn acts as an oxidising agent when it reacts with \[HN{{O}_{3}}\] done
clear
B)
\[HN{{O}_{3}}\] is weaker acid than \[{{H}_{2}}S{{O}_{4}}\]and \[HCl\] done
clear
C)
In electrochemical series, Zn is above hydrogen done
clear
D)
\[NO_{3}^{-}\] is reduced in preference to hydronium ion done
clear
View Solution play_arrow
-
question_answer4)
A compound of Xe and F is found to have 53.5% of Xe. What is oxidation number of Xe in this compound?
A)
- 4 done
clear
B)
0 done
clear
C)
+4 done
clear
D)
+6 done
clear
View Solution play_arrow
-
question_answer5)
Which of the following is not a disproportionation reaction?
A)
\[2PhCHO\xrightarrow{Al{{(OEt)}_{3}}}PhCOOC{{H}_{2}}Ph\] done
clear
B)
C)
\[NaH+{{H}_{2}}O\xrightarrow{{}}NaOH+{{H}_{2}}\] done
clear
D)
All of the above. done
clear
View Solution play_arrow
-
question_answer6)
A solution contains \[F{{e}^{2+}},F{{e}^{3+}}\] and \[{{I}^{-}}\] ions. This solution was treated with iodine at \[35{}^\circ C.\text{ }E{}^\circ \] for \[F{{e}^{3+}}/F{{e}^{2+}}\] is \[+0.77V\] and \[{{E}^{{}^\circ }}\]or \[{{I}_{2}}/2{{I}^{-}}=0.536V.\] The favourable redox reaction is:
A)
\[{{I}_{2}}\] will be reduced to \[{{I}^{-}}\] done
clear
B)
There will be no redox reaction done
clear
C)
\[{{I}^{-}}\] will be oxidised to \[{{I}_{2}}\] done
clear
D)
\[F{{e}^{2+}}\] will be oxidised to \[F{{e}^{3+}}\] done
clear
View Solution play_arrow
-
question_answer7)
Which of the following is correct code for x and y in the following reaction.
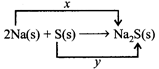 |
| (i) x = oxidation reaction, y = reduction reaction |
| (ii) x = gain of two electrons, y = loss of two electrons, |
| (iii) x = reduction reaction, y = oxidation reaction |
| (iv) x = loss of two electrons, y = gain of two electrons |
A)
(i) and (ii) done
clear
B)
(i) and (iv) done
clear
C)
(ii) and (iii) done
clear
D)
(iii) and (iv) done
clear
View Solution play_arrow
-
question_answer8)
One mole of \[{{N}_{2}}{{H}_{4}}\] loses 10 moles of electrons to form a new compound, y. Assuming that all nitrogen appear in the new compound, what is the oxidation state of nitrogen in y (There is no change in the oxidation state of hydrogen )
A)
\[-1\] done
clear
B)
\[-3\] done
clear
C)
+3 done
clear
D)
+5 done
clear
View Solution play_arrow
-
question_answer9)
The oxidation state of nitrogen is correctly given for
A)
B)
C)
D)
View Solution play_arrow
-
question_answer10)
The equivalent mass of oxidising agent in the following reaction is \[S{{O}_{2}}+2{{H}_{2}}S\xrightarrow{{}}3S+2{{H}_{2}}O\]
A)
32 done
clear
B)
64 done
clear
C)
16 done
clear
D)
8 done
clear
View Solution play_arrow
-
question_answer11)
Amongst the following, identify the species with an atom in + 6 oxidation state:
A)
\[{{[Mn{{O}_{4}}]}^{-}}\] done
clear
B)
\[{{\left[ Cr{{\left( CN \right)}_{6}} \right]}^{3-}}\] done
clear
C)
\[C{{r}_{2}}{{O}_{3}}\] done
clear
D)
\[Cr{{O}_{2}}C{{l}_{2}}\] done
clear
View Solution play_arrow
-
question_answer12)
Oxidation state of sulphur in anions \[SO_{3}^{2-},\,{{S}_{2}}O_{4}^{2-}\] and \[{{S}_{2}}O_{6}^{2-}\] increases in the orders:
A)
\[{{S}_{2}}O_{6}^{2-}<{{S}_{2}}O_{4}^{2-}<SO_{3}^{2-}~\] done
clear
B)
\[SO_{6}^{2-}<{{S}_{2}}O_{4}^{2-}<{{S}_{2}}O_{6}^{2-}~\] done
clear
C)
\[{{S}_{2}}O_{4}^{2-}<SO_{3}^{2-}<{{S}_{2}}O_{6}^{2-}~\] done
clear
D)
\[{{S}_{2}}O_{4}^{2-}<{{S}_{2}}O_{6}^{2-}<SO_{3}^{2-}~\] done
clear
View Solution play_arrow
-
question_answer13)
In the reaction \[3Mg+{{N}_{2}}\to M{{g}_{3}}{{N}_{2}}\]
A)
magnesium is reduced done
clear
B)
magnesium is oxidized done
clear
C)
nitrogen is oxidized done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer14)
One gas bleaches the colour of flowers by reduction, while the other by oxidation
A)
CO and\[C{{l}_{2}}\] done
clear
B)
\[S{{O}_{\text{2}}}\] and \[C{{l}_{2}}\] done
clear
C)
\[{{H}_{2}}S\] and\[B{{r}_{\text{2}}}\] done
clear
D)
\[N{{H}_{3}}\] and \[S{{O}_{2}}\] done
clear
View Solution play_arrow
-
question_answer15)
In the reaction: \[C+4HN{{O}_{3}}\to C{{O}_{2}}+2{{H}_{2}}O+4N{{O}_{2}}\] \[HN{{O}_{3}}\] act as
A)
an oxidizing agent done
clear
B)
an acid done
clear
C)
an acid as well as oxidizing agent done
clear
D)
a reducing agent. done
clear
View Solution play_arrow
-
question_answer16)
Which of the following represents redox reactions?
| I. \[C{{r}_{2}}O_{7}^{2-}+2\overline{O}H\xrightarrow{{}}2Cr_{4}^{2-}+{{H}_{2}}O\] |
| II. \[Zn+CuS{{O}_{4}}\xrightarrow{{}}\text{ }ZnS{{O}_{4}}+Cu\] |
| III. \[2MnO_{4}^{-}+3M{{n}^{2+}}+4\overline{O}H\xrightarrow{{}}\] \[5Mn{{O}_{2}}+2{{H}_{2}}O\] |
| IV. \[2C{{u}^{+}}\xrightarrow{{}}Cu+C{{u}^{2+}}\] |
A)
I, II done
clear
B)
I, III done
clear
C)
III, IV done
clear
D)
II, III, IV done
clear
View Solution play_arrow
-
question_answer17)
The standard reduction potentials at 298K for the following half reactions are given against each
| \[Z{{n}^{2+}}(aq)+2e\rightleftharpoons Zn(s);-0.762V\] |
| \[C{{r}^{3+}}(aq)+3e\rightleftharpoons Cr(s);-0.740V\] |
| \[2{{H}^{+}}(aq)+2e\rightleftharpoons {{H}_{2}}(g);0.00V\] |
| \[F{{e}^{3+}}(aq)+e\rightleftharpoons F{{e}^{2+}}(aq);0.770V\] |
| Which is the strongest reducing agent? |
A)
Zn(s) done
clear
B)
Cr(s) done
clear
C)
\[{{H}_{2}}\left( g \right)\] done
clear
D)
\[F{{e}^{3+}}(aq)\] done
clear
View Solution play_arrow
-
question_answer18)
The correct decreasing order of oxidation number of oxygen in compounds \[Ba{{O}_{2}},{{O}_{3}},K{{O}_{2}}\] and \[O{{F}_{2}}\] is:
A)
\[Ba{{O}_{2}}>K{{O}_{2}}>{{O}_{3}}>O{{F}_{2}}\] done
clear
B)
\[O{{F}_{2}}>{{O}_{3}}>K{{O}_{2}}>Ba{{O}_{2}}\] done
clear
C)
\[K{{O}_{2}}>O{{F}_{2}}>{{O}_{3}}>Ba{{O}_{2}}\] done
clear
D)
\[Ba{{O}_{2}}>{{O}_{3}}>O{{F}_{2}}>K{{O}_{2}}\] done
clear
View Solution play_arrow
-
question_answer19)
In the balanced chemical reaction \[IO_{3}^{-}+a{{l}^{-}}+b{{H}^{-}}\xrightarrow{{}}c{{H}_{2}}O+d{{I}_{2}}\] a, b, c and d, respectively, correspond to
A)
5, 6, 3, 3 done
clear
B)
5, 3, 6, 3 done
clear
C)
3, 5, 3, 6 done
clear
D)
5, 6, 5, 5 done
clear
View Solution play_arrow
-
question_answer20)
In the reaction shown below, oxidation state of the carbon in reactant and product are (i) and (ii) respectively? Is the given reaction a redox reaction?
| \[N{{a}_{2}}C{{O}_{3}}(aq)+HCl(aq)\xrightarrow{{}}\] |
| \[N{{a}^{+}}(aq)+C{{l}^{-}}(aq)+{{H}_{2}}O(l)+C{{O}_{2}}(g)\] |
A)
(i) 6, (ii) 4, yes done
clear
B)
(i) 6, (ii) 6, No done
clear
C)
(i) 4, (ii) 4, No done
clear
D)
(i) 4, (ii) 4, yes done
clear
View Solution play_arrow
-
question_answer21)
In the disproportionation reaction \[3HCl{{O}_{3}}\xrightarrow{{}}HCl{{O}_{4}}+C{{l}_{2}}+2{{O}_{2}}+{{H}_{2}}O,\] the equivalent mass of the oxidizing agent is (molar mass of \[HCl{{O}_{3}}=84.45\])
A)
16.89 done
clear
B)
32.22 done
clear
C)
84.45 done
clear
D)
28.15 done
clear
View Solution play_arrow
-
question_answer22)
The oxidation number of phosphorus in \[Ba{{({{H}_{2}}P{{O}_{2}})}_{2}}\] is
A)
+3 done
clear
B)
+2 done
clear
C)
+1 done
clear
D)
- 1 done
clear
View Solution play_arrow
-
question_answer23)
How many electrons are involved in the following redox reaction?
| \[C{{r}_{2}}O_{7}^{2-}+F{{e}^{2+}}+{{C}_{2}}O_{4}^{2-}\to C{{r}^{3+}}+F{{e}^{3+}}+C{{O}_{2}}\](Unbalanced) |
A)
3 done
clear
B)
4 done
clear
C)
6 done
clear
D)
5 done
clear
View Solution play_arrow
-
question_answer24)
Given:
| \[XN{{a}_{2}}HAs{{O}_{3}}+YNaBr{{O}_{3}}+ZHCl\to \] |
| \[NaBr+{{H}_{3}}As{{O}_{4}}+NaCl\] |
| The values of X, Y and Z in the above redox reaction are respectively: |
A)
2, 1, 2 done
clear
B)
2, 1, 3 done
clear
C)
3, 1, 6 done
clear
D)
3, 1, 4 done
clear
View Solution play_arrow
-
question_answer25)
When \[S{{O}_{2}}\] is passed through acidified solution of potassium dichromate, then chromium sulphate is formed. The change in valency of chromium is
A)
+ 4 to + 2 done
clear
B)
+ 5 to + 3 done
clear
C)
+ 6 to + 3 done
clear
D)
+ 7 to + 2 done
clear
View Solution play_arrow
-
question_answer26)
In which of the following transition metal complexes does the metal exhibit zero oxidation state?
A)
\[[Co{{\left( N{{H}_{3}} \right)}_{6}}]C{{l}_{3}}\] done
clear
B)
\[[Fe{{({{H}_{2}}O)}_{6}}]S{{O}_{4}}\] done
clear
C)
\[Ni{{\left( CO \right)}_{4}}\] done
clear
D)
\[\left[ Fe{{\left( {{H}_{2}}O \right)}_{6}} \right]{{X}_{3}}\] done
clear
View Solution play_arrow
-
question_answer27)
In which of the following pairs, there is greatest difference in the oxidation number of the underlined elements?
A)
\[\underline{N}{{O}_{2}}\] and \[{{\underline{N}}_{2}}{{O}_{4}}\] done
clear
B)
\[{{\underline{P}}_{2}}{{O}_{5}}\] and \[{{\underline{P}}_{4}}{{O}_{10}}\] done
clear
C)
\[{{\underline{N}}_{2}}O\] and \[\underline{N}O\] done
clear
D)
\[\underline{S}{{O}_{2}}\] and \[\underline{S}{{O}_{3}}\] done
clear
View Solution play_arrow
-
question_answer28)
Which of the following statements is not correct?
A)
The oxidation number of S in \[{{(N{{H}_{4}})}_{2}}{{S}_{2}}{{O}_{8}}\]is +6. done
clear
B)
The oxidation number of Os in \[Os{{O}_{4}}\] is +8. done
clear
C)
The oxidation number of S in \[{{H}_{2}}S{{O}_{5}}\] is +8. done
clear
D)
The oxidation number of O in \[K{{O}_{2}}\] is -1/2. done
clear
View Solution play_arrow
-
question_answer29)
Among \[N{{H}_{3}},HN{{O}_{3}},Na{{N}_{3}}\] and \[M{{g}_{3}}{{N}_{2}}\] the number of molecules having nitrogen in negative oxidation state is
A)
1 done
clear
B)
2 done
clear
C)
3 done
clear
D)
4 done
clear
View Solution play_arrow
-
question_answer30)
\[a{{K}_{2}}C{{r}_{2}}{{O}_{7}}+bKCl+c{{H}_{2}}S{{O}_{4}}\xrightarrow{{}}\] \[xCr{{O}_{2}}C{{l}_{2}}+yKHS{{O}_{4}}+z{{H}_{2}}O\] The above equation balances when
A)
a = 2, b = 4, c = 6 and x = 2, y = 6, z = 3 done
clear
B)
a = 4, b = 2, c = 6 and x = 6, y = 2, z = 3 done
clear
C)
a = 6, b = 4, c = 2 and x = 6, y = 3, z = 2 done
clear
D)
a = 1, b = 4, c = 6 and x = 2, y = 6, z = 3 done
clear
View Solution play_arrow
-
question_answer31)
The number of electrons involved in the reduction of one nitrate ion to hydrazine is
A)
8 done
clear
B)
5 done
clear
C)
3 done
clear
D)
7 done
clear
View Solution play_arrow
-
question_answer32)
Which of the following do not show disproportionation reaction? \[ClO_{4}^{-},\,{{F}_{2}},\,C{{l}_{2}},\,ClO_{2}^{-},\,ClO_{2}^{-},\,{{P}_{4}},\,{{S}_{8}}\] and \[Cl{{O}^{-}}\]
A)
\[ClO_{2}^{-},\,ClO_{4}^{-},\] and \[Cl{{O}^{-}}\] done
clear
B)
\[{{F}_{2}}\] only done
clear
C)
\[{{F}_{2}}\] and \[ClO_{4}^{-}\] done
clear
D)
\[ClO_{4}^{-}\] only done
clear
View Solution play_arrow
-
question_answer33)
In the following balanced reaction, \[XMnO_{4}^{-}+Y{{C}_{2}}O_{4}^{2-}+Z{{H}^{+}}\] \[\rightleftharpoons XM{{n}^{2+}}+2YC{{O}_{2}}+\frac{Z}{2}{{H}_{2}}O\] values of X, Y and Z respectively are
A)
2, 5, 16 done
clear
B)
8, 2, 5 done
clear
C)
5, 2, 16 done
clear
D)
5, 8, 4 done
clear
View Solution play_arrow
-
question_answer34)
Consider the reaction:
| \[{{H}_{2}}S{{O}_{3}}\left( aq \right)+S{{n}^{4+}}\left( aq \right)+{{H}_{2}}O\left( I \right)\] |
| \[\to S{{n}^{2+}}(aq)+HSO_{4}^{-}(aq)+3{{H}^{+}}(aq)\] |
| Which of the following statements is correct? |
A)
\[S{{n}^{4+}}\] is the oxidizing agent because it undergoes oxidation done
clear
B)
\[S{{n}^{4+}}\] is the reducing agent because it undergoes oxidation done
clear
C)
\[{{H}_{2}}S{{O}_{3}}\] is the reducing agent because it undergoes oxidation done
clear
D)
\[{{H}_{2}}S{{O}_{3}}\] is the reducing agent because it undergoes reduction done
clear
View Solution play_arrow
-
question_answer35)
Which of the following statements is not correct about the reaction given below? \[{{K}_{4}}[Fe{{(CN)}_{6}}]\xrightarrow{oxidation}F{{e}^{3+}}+C{{O}_{2}}+NO_{3}^{-}\]
A)
Fe is oxidised from \[F{{e}^{2+}}\] to \[F{{e}^{3+}}.\] done
clear
B)
Carbon is oxidised from \[{{C}^{2+}}\] to \[{{C}^{4+}}\]. done
clear
C)
N is oxidised from \[{{N}^{3-}}\] to \[{{N}^{5+}}\]. done
clear
D)
Carbon is not oxidised. done
clear
View Solution play_arrow
-
question_answer36)
Which of the following is not an intermolecular redox reaction?
A)
\[MgC{{O}_{3}}\xrightarrow{{}}MgO+C{{O}_{2}}\] done
clear
B)
\[{{O}_{2}}+2{{H}_{2}}\xrightarrow{{}}2{{H}_{2}}O\] done
clear
C)
\[K+{{H}_{2}}O\xrightarrow{{}}KOH+(1/2){{H}_{2}}\] done
clear
D)
\[MnB{{r}_{3}}\xrightarrow{{}}MnB{{r}_{2}}+(1/2)B{{r}_{2}}\] done
clear
View Solution play_arrow
-
question_answer37)
Which of the following is not a disproportionation reaction?
A)
\[{{P}_{4}}+5O{{H}^{-}}\xrightarrow{{}}{{H}_{2}}PO_{2}^{-}+P{{H}_{3}}\] done
clear
B)
\[C{{l}_{2}}+O{{H}^{-}}\xrightarrow{{}}C{{l}^{-}}+ClO\] done
clear
C)
\[2{{H}_{2}}{{O}_{2}}\xrightarrow{{}}2{{H}_{2}}O+{{O}_{2}}\] done
clear
D)
\[Pb{{O}_{2}}+{{H}_{2}}O\xrightarrow{{}}PbO+{{H}_{2}}{{O}_{2}}\] done
clear
View Solution play_arrow
-
question_answer38)
In a balance equation \[{{H}_{2}}S{{O}_{4}}+xHI\to {{H}_{2}}S+y{{I}_{2}}+z{{H}_{2}}O,\] the values of x, y, z are
A)
x=3, y=5, z=2 done
clear
B)
x=4, y=8, z=5 done
clear
C)
x=8, y=4, z=4 done
clear
D)
x=5, y=3, z=4 done
clear
View Solution play_arrow
-
question_answer39)
Given
| \[F{{e}^{3+}}(aq)+{{e}^{-}}\to F{{e}^{2+}}(aq);E{}^\circ =+0.77V\] |
| \[A{{l}^{3+}}(aq)+3{{e}^{-}}\to Al(s);E{}^\circ =-1.66V\] |
| \[B{{r}_{2}}(aq)+2{{e}^{-}}\to 2B{{r}^{-}}(aq);E{}^\circ =+1.09V\] |
| Considering the electrode potentials, which of the following represents the correct order of reducing power? |
A)
\[F{{e}^{2+}}<Al<B{{r}^{-}}\] done
clear
B)
\[B{{r}^{-}}<F{{e}^{2+}}<Al\] done
clear
C)
\[Al<B{{r}^{-}}<F{{e}^{2+}}\] done
clear
D)
\[Al<F{{e}^{2+}}<B{{r}^{-}}\] done
clear
View Solution play_arrow
-
question_answer40)
For the reaction: \[N{{H}_{3}}+OC{{l}^{-}}\xrightarrow{{}}{{N}_{2}}{{H}_{4}}+C{{l}^{-}}\] in basic medium, the coefficients of \[N{{H}_{3}},OC{{l}^{-}}\] and \[{{N}_{2}}{{H}_{4}}\] for the balanced equation are respectively
A)
2, 2, 2 done
clear
B)
2, 2, 1 done
clear
C)
2, 1, 1 done
clear
D)
4, 4, 2 done
clear
View Solution play_arrow
-
question_answer41)
Which of the following act as reducing agents?
| (i) \[PO_{4}^{3-}\] |
| (ii) \[S{{O}_{3}}\] |
| (iii) \[PO_{3}^{2-}\] |
| (iv) \[N{{H}_{3}}\] |
A)
(i), (ii) and (iii) done
clear
B)
Only (iii) done
clear
C)
(i), (iii) and (iv) done
clear
D)
(iii) and (iv) done
clear
View Solution play_arrow
-
question_answer42)
In the reaction
| \[C{{r}_{2}}O_{7}^{2-}+14{{H}^{+}}+{{6I}^{-}}\xrightarrow{{}}2C{{r}^{3+}}+7{{H}_{2}}O+3{{I}_{2}}\] |
| Which element is reduced? |
A)
I done
clear
B)
O done
clear
C)
H done
clear
D)
Cr done
clear
View Solution play_arrow
-
question_answer43)
Which of the following substances acts as an oxidising as well as a reducing agent?
A)
\[N{{a}_{2}}O\]\[\] done
clear
B)
\[SnC{{l}_{2}}\] done
clear
C)
\[N{{a}_{2}}{{O}_{2}}\] done
clear
D)
\[NaN{{O}_{2}}\] done
clear
View Solution play_arrow
-
question_answer44)
Stronger is oxidising agent, more is
A)
standard reduction potential of that species done
clear
B)
the tendency to get itself oxidized done
clear
C)
the tendency to lose electrons by that species done
clear
D)
standard oxidation potential of that species done
clear
View Solution play_arrow
-
question_answer45)
Which of the following involves transfer of five electrons?
A)
\[MnO_{4}^{-}\to M{{n}^{2+}}\] done
clear
B)
\[CrO_{4}^{2-}\to C{{r}^{3+}}\] done
clear
C)
\[MnO_{4}^{2-}\to Mn{{O}_{2}}\] done
clear
D)
\[C{{r}_{2}}O_{7}^{2-}\to 2C{{r}^{3+}}\] done
clear
View Solution play_arrow
-
question_answer46)
The brown ring complex is formulated as\[[Fe{{({{H}_{2}}O)}_{5}}NO]S{{O}_{4}}\]. The oxidation number of iron is
A)
1 done
clear
B)
2 done
clear
C)
3 done
clear
D)
0 done
clear
View Solution play_arrow
-
question_answer47)
In which of the following reactions, there is no change in valency?
A)
\[4KCl{{O}_{3}}\to 3KCl{{O}_{4}}+KCl\] done
clear
B)
\[S{{O}_{2}}+2{{H}_{2}}\text{S}\to 2{{H}_{2}}O+3S\] done
clear
C)
\[Ba{{O}_{2}}+{{H}_{2}}S{{O}_{4}}\to BaS{{O}_{4}}+{{H}_{2}}{{O}_{2}}\] done
clear
D)
\[2BaO+{{O}_{2}}\to 2Ba{{O}_{2}}\] done
clear
View Solution play_arrow
-
question_answer48)
The reaction, \[2{{H}_{2}}O\left( l \right)\to 4{{H}^{+}}\left( aq \right)+{{O}_{2}}\left( g \right)+4{{e}^{-}}\] is
A)
an oxidation reaction done
clear
B)
a reduction reaction done
clear
C)
a redox reaction done
clear
D)
a hydrolysis reaction done
clear
View Solution play_arrow
-
question_answer49)
In the reaction \[2FeC{{l}_{3}}+{{H}_{2}}S\to 2FeC{{l}_{2}}+2HCl+S\]
A)
\[FeC{{l}_{3}}\] acts as an oxidising agent done
clear
B)
Both \[{{H}_{2}}S\] are \[FeC{{l}_{3}}\] are oxidized done
clear
C)
\[FeC{{l}_{3}}\] is oxidised while \[{{H}_{2}}S\] is reduced done
clear
D)
\[{{H}_{2}}S\] acts as an oxidising agent. done
clear
View Solution play_arrow
-
question_answer50)
The oxidation number of S in \[N{{a}_{2}}{{S}_{4}}{{O}_{6}}\] is
A)
+0.5 done
clear
B)
2.5 done
clear
C)
+4 done
clear
D)
+6 done
clear
View Solution play_arrow
-
question_answer51)
In the following reaction \[4P+3KOH+3{{H}_{2}}O\xrightarrow{{}}3K{{H}_{2}}P{{O}_{2}}+P{{H}_{3}}\]
A)
phosphorus is both oxidised and reduced. done
clear
B)
only phosphorus is reduced. done
clear
C)
phosphorus is not oxidized done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer52)
The standard electrode potentials of four elements A, B, C and D are -3.05, -1.66, -0.40 and +0.80. The highest chemical reactivity will be exhibited by:
A)
A done
clear
B)
B done
clear
C)
C done
clear
D)
D done
clear
View Solution play_arrow
-
question_answer53)
The oxidation number of Fe in \[N{{a}_{2}}[Fe{{(CN)}_{5}}NO]\]is
A)
+2 done
clear
B)
+1 done
clear
C)
+3 done
clear
D)
-2 done
clear
View Solution play_arrow
-
question_answer54)
Which of the following is the strongest reducing agent in aqueous medium?
A)
Mg done
clear
B)
Na done
clear
C)
Li done
clear
D)
Ca done
clear
View Solution play_arrow
-
question_answer55)
The oxidation state of Fe in \[F{{e}_{3}}{{O}_{8}}\] is
A)
3/2 done
clear
B)
4/5 done
clear
C)
5/4 done
clear
D)
16/3 done
clear
View Solution play_arrow
-
question_answer56)
Which substance serves as reducing agent in the following reaction? \[14{{H}^{+}}+C{{r}_{2}}O_{7}^{2-}+3Ni\xrightarrow{{}}\] \[2C{{r}^{3+}}+7{{H}_{2}}O+3N{{i}^{2+}}\]
A)
\[{{H}_{2}}O\] done
clear
B)
Ni done
clear
C)
\[{{H}^{+}}\] done
clear
D)
\[C{{r}_{2}}O_{7}^{2-}\] done
clear
View Solution play_arrow
-
question_answer57)
In the reaction \[3B{{r}_{2}}+6CO_{3}^{2-}+3{{H}_{2}}O\xrightarrow{{}}\] \[5B{{r}^{-}}+BrO_{3}^{-}+6HCO_{3}^{-}\]
A)
bromine is oxidised and carbonate is reduced. done
clear
B)
bromine is reduced and water is oxidized done
clear
C)
bromine is neither reduced nor oxidized done
clear
D)
bromine is both reduced and oxidized done
clear
View Solution play_arrow
-
question_answer58)
Which one of the following cannot function as an oxidising agent?
A)
\[{{I}^{-}}\] done
clear
B)
\[S\left( s \right)\] done
clear
C)
\[NO_{3}^{-}(aq)\] done
clear
D)
\[C{{r}_{2}}O_{7}^{2-}\] done
clear
View Solution play_arrow
-
question_answer59)
The oxidation number of Pt in \[{{[Pt({{C}_{2}}{{H}_{4}})C{{l}_{3}}]}^{-}}\] is
A)
+1 done
clear
B)
+2 done
clear
C)
+3 done
clear
D)
+4 done
clear
View Solution play_arrow
-
question_answer60)
Standard electrode potentials of redox couples \[{{A}^{2+}}/A,{{B}^{2+}}/B,{{C}^{2+}}/C\] and \[{{D}^{2+}}/D\] are \[0.3V,-0.5V,-0.75V\] and 0.9V respectively. Which of these is best oxidising agent and reducing agent respectively?
A)
\[{{D}^{2+}}/D\] and \[{{B}^{2+}}/B\] done
clear
B)
\[{{B}^{2+}}/B\] and \[{{D}^{2+}}/D\] done
clear
C)
\[{{D}^{2+}}/D\] and \[{{C}^{2+}}/C\] done
clear
D)
\[{{C}^{2+}}/C\] and \[{{D}^{2+}}/D\] done
clear
View Solution play_arrow
