| The correct order of acidic strength of the following alcohols is |
| I. \[C{{H}_{3}}OH\] |
| II. \[{{C}_{2}}{{H}_{5}}OH\] |
| III. \[C{{H}_{3}}-CH(OH)-C{{H}_{3}}\] |
IV. 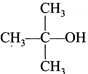 |
A) I<II<III<IV
B) III<II<IV<I
C) III<IV<I<III
D) IV<III<II<I
Correct Answer: D
Solution :
[d] More stable is alkoxide ion \[R-{{O}^{\Theta }}\], more is the acidic nature of corresponding alcohol. Electron donating (+I-effect) alkyl groups decreases the acidic strength.You need to login to perform this action.
You will be redirected in
3 sec
