-
question_answer1)
Which of the following solids are not correctly matched with the bonds found between the constituent particles?
A)
Solid \[C{{O}_{2}}\]: van der Waals done
clear
B)
Graphite: Covalent and van der Waals done
clear
C)
Grey Cast Iron: Ionic done
clear
D)
Metal alloys: lons-delocalised electrons done
clear
View Solution play_arrow
-
question_answer2)
Copper crystallises in a structure of face centered cubic unit cell. The atomic radius of copper is 1.28 \[\overset{\text{o}}{\mathop{\text{A}}}\,\]. What is the axial length on an edge of copper?
A)
2.16\[\overset{\text{o}}{\mathop{\text{A}}}\,\] done
clear
B)
3.62\[\overset{\text{o}}{\mathop{\text{A}}}\,\] done
clear
C)
3.94\[\overset{\text{o}}{\mathop{\text{A}}}\,\] done
clear
D)
4.15\[\overset{\text{o}}{\mathop{\text{A}}}\,\] done
clear
View Solution play_arrow
-
question_answer3)
In a face centered lattice of X and Y, X atoms are present at the comers while Y atoms are at face centers. Then the formula of the compound would be if two atoms of X are missing from the comers:
A)
\[{{X}_{4}}\]Y done
clear
B)
\[{{X}_{3}}\]\[{{Y}_{4}}\] done
clear
C)
X\[{{Y}_{4}}\] done
clear
D)
\[{{X}_{2}}\]\[{{Y}_{4}}\] done
clear
View Solution play_arrow
-
question_answer4)
Consider a cube 1 of body centered cubic unit cell of edge length a. Now atom at the body centre can be viewed to be lying on the comer of another cube 2. Find the volume common to cube 1 and cube 2.
A)
\[\frac{{{a}^{3}}}{27}\] done
clear
B)
\[\frac{{{a}^{3}}}{64}\] done
clear
C)
\[\frac{{{a}^{3}}}{2\sqrt{2}}\] done
clear
D)
\[\frac{{{a}^{3}}}{8}\] done
clear
View Solution play_arrow
-
question_answer5)
How many tetrahedral holes are occupied in a diamond?
A)
25% done
clear
B)
50% done
clear
C)
75% done
clear
D)
100% done
clear
View Solution play_arrow
-
question_answer6)
A mineral has molecular formula \[X{{Y}_{2}}{{O}_{4}}\cdot {{O}^{2-}}\] ions are arranged in ccp and cations of X are present in octahedral voids. Cations of Y are equally distributed among octahedral and tetrahedral voids. The fraction of the octahedral voids occupied will be
A)
\[\frac{1}{6}\] done
clear
B)
\[\frac{1}{8}\] done
clear
C)
\[\frac{1}{4}\] done
clear
D)
\[\frac{1}{2}\] done
clear
View Solution play_arrow
-
question_answer7)
A crystalline solid is made of X, Y and Z. Atoms of X are present in ccp lattice; Y occupies all the octahedral voids and Z occupies all the tetrahedral voids. If all atoms along with body are diagonally removed then formula of solid will be:
A)
\[{{X}_{5}}{{Y}_{4}}{{Z}_{8}}\] done
clear
B)
\[{{X}_{8}}{{Y}_{4}}{{Z}_{5}}\] done
clear
C)
\[{{X}_{2}}Y{{Z}_{2}}\] done
clear
D)
\[XY{{Z}_{2}}\] done
clear
View Solution play_arrow
-
question_answer8)
Which of the following statement is not true about the hexagonal close packing?
A)
The coordination number is 12 done
clear
B)
It has 74% packing efficiency done
clear
C)
Tetrahedral voids of the second layer are covered by the spheres of the third layer done
clear
D)
In this arrangement, spheres of the fourth layer are exactly aligned with those of the first layer done
clear
View Solution play_arrow
-
question_answer9)
PbS has NaCl type structure. The distance between \[P{{b}^{2+}}\] and \[{{S}^{2-}}\] ions is 297 pm. What is the volume of unit cell of PbS?
A)
\[209.6\times {{10}^{-24}}c{{m}^{3}}\] done
clear
B)
\[207.8\times {{10}^{-23}}c{{m}^{3}}\] done
clear
C)
\[22.3\times {{10}^{-23}}c{{m}^{3}}\] done
clear
D)
\[209.8\times {{10}^{-23}}c{{m}^{3}}\] done
clear
View Solution play_arrow
-
question_answer10)
\[MgA{{l}_{2}}{{O}_{4}}\], is found in the spinel structure in which \[{{O}^{2-}}\] ions constitute CCP lattice, \[M{{g}^{2+}}\] ions occupy l/8th of the tetrahedral voids and \[A{{l}^{3+}}\] ions occupy 1/2 of the octahedral voids. Find the total +ve charge contained in one unit cell.
A)
+7/4 electronic charge done
clear
B)
+6 electronic charge done
clear
C)
+2 electronic charge done
clear
D)
+8 electronic charge done
clear
View Solution play_arrow
-
question_answer11)
A mineral of iron contains an oxide containing 72.36% iron by mass and has a density of 5.2 g/cc. Its unit cell is cubic with edge length of 839 pm. What is the total number of atoms (ions) present in each unit cell? (Fe-56, O-16)
A)
58 done
clear
B)
57 done
clear
C)
56 done
clear
D)
55 done
clear
View Solution play_arrow
-
question_answer12)
LiCl has NaCl type structure with edge length 512 pm. Assuming that the \[L{{i}^{+}}\] ion is small enough that the chloride ions are in contact, then radius of \[C{{I}^{-}}\] ion is
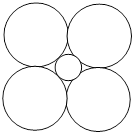
A)
223 pm done
clear
B)
224 pm done
clear
C)
181 pm done
clear
D)
362 pm done
clear
View Solution play_arrow
-
question_answer13)
The density of solid argon (Ar = 40 g/mol) is 1.68 g/mL at 40 K. If the argon atom is assumed to be a sphere of radius \[1.50\times {{10}^{-8}}\] cm, what % of solid Ar is apparently empty space? (use \[{{N}_{A}}\] = \[6\times {{10}^{23}}\])
A)
35.64 done
clear
B)
64.36 done
clear
C)
74 done
clear
D)
none of these done
clear
View Solution play_arrow
-
question_answer14)
The number of unit cells in 58.5 g of NaCl is nearly
A)
\[6\times {{10}^{20}}\] done
clear
B)
\[1.5\times {{10}^{23}}\] done
clear
C)
\[3\times {{10}^{22}}\] done
clear
D)
\[0.5\times {{10}^{24}}\] done
clear
View Solution play_arrow
-
question_answer15)
The spinel structure (\[A{{B}_{2}}{{O}_{4}}\]) consists of an fee array of \[{{O}^{2-}}\] ions in which the:
A)
A cation occupies one-eighth of the tetrahedral holes and B cation occupies half of octahedral holes done
clear
B)
A cation occupies one-fourth of the tetrahedral holes and the B cations the octahedral holes done
clear
C)
A cation occupies one-eighth of the octahedral hole and the B cation the tetrahedral holes done
clear
D)
A cation occupies one-fourth of the octahedral holes and the B cations the tetrahedral holes done
clear
View Solution play_arrow
-
question_answer16)
If NaCl is doped with \[{{10}^{-3}}\] mole% of \[SrC{{l}_{2}}\], the number of cationic vacancies is
A)
\[6.02\times {{10}^{-18}}mo{{l}^{-1}}\] done
clear
B)
\[{{10}^{-5}}mo{{l}^{-1}}\] done
clear
C)
\[6.02\times {{10}^{20}}mo{{l}^{-1}}\] done
clear
D)
\[6.02\times {{10}^{18}}mo{{l}^{-1}}\] done
clear
View Solution play_arrow
-
question_answer17)
Which of the following statements are correct in context of point defects in a crystal?
A)
AgCl has anion Frenkel defect and \[Ca{{F}_{2}}\] has Schottky defects done
clear
B)
AgCl has cation Frenkel defects and \[Ca{{F}_{2}}\] has anion Frenkel defects done
clear
C)
AgCl and \[Ca{{F}_{2}}\] have anion Frenkel defects done
clear
D)
AgCl and \[Ca{{F}_{2}}\] have Schottky defects done
clear
View Solution play_arrow
-
question_answer18)
Which of the following pairs contain ferromagnetic and ferrimagnetic solids respectively?
A)
\[F{{e}_{2}}{{O}_{3}},F{{e}_{3}}{{O}_{4}}\] done
clear
B)
\[F{{e}_{3}}{{O}_{4}},C{{r}_{2}}{{O}_{3}}\] done
clear
C)
\[Cr{{O}_{2}},F{{e}_{3}}{{O}_{4}}\] done
clear
D)
\[C{{r}_{2}}{{O}_{3}},Cr{{O}_{2}}\] done
clear
View Solution play_arrow
-
question_answer19)
In Bragg's X-ray diffraction studies. 100 pm X-ray second order diffraction at glancing angle of\[30{}^\circ \]. Its interplanar spacing will be
A)
200 pm done
clear
B)
150 pm. done
clear
C)
100 pm done
clear
D)
144 pm done
clear
View Solution play_arrow
-
question_answer20)
In orthorhombic, the value of a, b and c are respectively 4.2\[\overset{\text{o}}{\mathop{\text{A}}}\,\], 8.6\[\overset{\text{o}}{\mathop{\text{A}}}\,\] and 8.3\[\overset{\text{o}}{\mathop{\text{A}}}\,\]. Given the molecular mass off solute is 155 gm \[mo{{l}^{-1}}\] and that of density is 3.3 g/cc, the number of formula units per unit cell is
A)
2 done
clear
B)
3 done
clear
C)
4 done
clear
D)
6 done
clear
View Solution play_arrow
-
question_answer21)
Calcium crystallises in a face-centred cubic unit cell wit! 0.556 nm and density 1.4848\[g\text{/}c{{m}^{3}}\]. Percentage of Schottky defects in this crystal is ....
View Solution play_arrow
-
question_answer22)
The number of square faces that are present in a truncated octahedron is .......
View Solution play_arrow
-
question_answer23)
An ionic compound AB has a rock salt structure with A : B = 1 : 1. The formula mass of AB is 6.023 y amu and the closest A - B distance is \[{{y}^{1/2}}\] nm. Calculate the distance of lattice.
View Solution play_arrow
-
question_answer24)
A second order Bragg's diffraction of X-rays from a set of parallel planes separated by 1.155\[\overset{\text{o}}{\mathop{\text{A}}}\,\] occurs at an angle \[60{}^\circ \]using wavelength of\[x\,\overset{\text{o}}{\mathop{\text{A}}}\,\]. What is the value of x?
View Solution play_arrow
-
question_answer25)
Number of unit cells in 240 g of element X (atomic ma 40) which crystallises in bcc pattern in y\[{{N}_{A}}\]. What is the value of y?
View Solution play_arrow
