-
question_answer1)
Which of the following is not an example of addition polymer?
A)
Polystyrene done
clear
B)
Nylon done
clear
C)
PVC done
clear
D)
Polypropylene done
clear
View Solution play_arrow
-
question_answer2)
Which of the following is not a biopolymer?
A)
Proteins done
clear
B)
Rubber done
clear
C)
Cellulose done
clear
D)
RNA done
clear
View Solution play_arrow
-
question_answer3)
Thermosets are:
A)
cross-linked polymers done
clear
B)
don't melt or soften on heating done
clear
C)
cross-linking is usually developed at the time of moulding where they harden reversibly done
clear
D)
all of the above done
clear
View Solution play_arrow
-
question_answer4)
Which of the following statements is not correct for fibres?
A)
Fibres possess high tensile strength and high modulus. done
clear
B)
Fibres impart crystalline nature. done
clear
C)
Characteristic features of fibres are due to strong intermolecularforces like hydrogen bonding. done
clear
D)
All are correct. done
clear
View Solution play_arrow
-
question_answer5)
Perlon is
A)
Rubber done
clear
B)
Nylon-6 done
clear
C)
Terylene done
clear
D)
Orion done
clear
View Solution play_arrow
-
question_answer6)
Which one of the following is a chain growth polymer?
A)
Nucleic acid done
clear
B)
Polystyrene done
clear
C)
Protein done
clear
D)
Starch done
clear
View Solution play_arrow
-
question_answer7)
Structure of some important polymers are given. Which one represents Buna-S?
A)
\[-(C{{H}_{2}}-\overset{C{{H}_{3}}}{\mathop{\overset{|}{\mathop{C}}\,}}\,=CH-C{{H}_{2}}){{-}_{n}}\] done
clear
B)
\[-(C{{H}_{2}}-CH=CH-C{{H}_{2}}-\underset{{{C}_{6}}{{H}_{5}}}{\mathop{\underset{|}{\mathop{C}}\,}}\,H-C{{H}_{2}}){{-}_{n}}\] done
clear
C)
\[-(C{{H}_{2}}-CH=CH-C{{H}_{2}}-\underset{CN}{\mathop{\underset{|}{\mathop{C}}\,}}\,H-C{{H}_{2}}){{-}_{n}}\] done
clear
D)
\[-(C{{H}_{2}}-\overset{Cl}{\mathop{\overset{|}{\mathop{C}}\,}}\,=CH-C{{H}_{2}}){{-}_{n}}\] done
clear
View Solution play_arrow
-
question_answer8)
Low density polythene is prepared by
A)
Free radical polymerization done
clear
B)
cationic polymerization done
clear
C)
anionic polymerization done
clear
D)
Ziegler-Natta polymerisation done
clear
View Solution play_arrow
-
question_answer9)
The polymer used for optical lenses is :
A)
polypropylene done
clear
B)
polyvinyl chloride done
clear
C)
polythene done
clear
D)
polymethyl methacrylate done
clear
View Solution play_arrow
-
question_answer10)
Polymer used in bullet proof glass is
A)
lexan done
clear
B)
PMMA done
clear
C)
nomex done
clear
D)
kevlar done
clear
View Solution play_arrow
-
question_answer11)
Glyptals are chiefly employed in
A)
toymaking done
clear
B)
surface coating done
clear
C)
photofilm making done
clear
D)
electrical insulators done
clear
View Solution play_arrow
-
question_answer12)
Which of the following is currently used as a tyre cord?
A)
Terylene done
clear
B)
Polyethylene done
clear
C)
Polypropylene done
clear
D)
Nylon-6 done
clear
View Solution play_arrow
-
question_answer13)
The plastic household crockery is prepared by using
A)
melamine and tetrafluoroethane done
clear
B)
malonic acid and hexamethyleneamine done
clear
C)
melamine and vinyl acetate done
clear
D)
melamine and formaldehyde done
clear
View Solution play_arrow
-
question_answer14)
Mark out the most unlike form of polymerization of \[C{{H}_{2}}=CH-CH=C{{H}_{2}}\]
A)
B)
C)
D)
View Solution play_arrow
-
question_answer15)
The condensation of hexamethylenediamine with sebacoyl chloride at 525 K gives
A)
nylon-6, 20 done
clear
B)
nylon-6, 01 done
clear
C)
nylon-6, 10 done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer16)
Example of condensation polymer is
A)
Formaldehyde \[\to \] meta-formaldehyde done
clear
B)
Acetaldehyde \[\to \] para-aldehyde done
clear
C)
Acetone \[\to \] mesityl oxide done
clear
D)
Ethene \[\to \] polyethene done
clear
View Solution play_arrow
-
question_answer17)
The mass average molecular mass & number average molecular mass of a polymer are 40,000 and 30,000 respectively. The polydispersity index of polymer will be
A)
<1 done
clear
B)
>1 done
clear
C)
1 done
clear
D)
0 done
clear
View Solution play_arrow
-
question_answer18)
Given the polymers (i) Nylon-6, 6; (ii) Buna-S; (iii) Polythene. Arrange these in increasing order of their inter-molecular forces (lower to higher)
A)
(i)>(ii)>(iii) done
clear
B)
(ii)>(iii)>(i) done
clear
C)
(ii)<(iii)<(i) done
clear
D)
(iii)<(i)<(ii) done
clear
View Solution play_arrow
-
question_answer19)
What is the percentage of sulphur used in vulcanization of rubber?
A)
05% to 30% done
clear
B)
03% to 25% done
clear
C)
10% to 20% done
clear
D)
05% to 25% done
clear
View Solution play_arrow
-
question_answer20)
Which of the following is used in vulcanization of rubber?
A)
\[{{S}_{8}}\] done
clear
B)
\[C{{F}_{4}}~\] done
clear
C)
\[C{{l}_{2}}{{F}_{2}}~\] done
clear
D)
\[{{C}_{2}}{{F}_{2}}~\] done
clear
View Solution play_arrow
-
question_answer21)
Which one of the following statement is not true?
A)
In vulcanization the formation of sulphur bridges between different chains make rubber harder and stronger. done
clear
B)
Natural rubber has the trans -configuration at every double bond done
clear
C)
Buna-S is a copolymer of butadiene and styrene done
clear
D)
Natural rubber is a 1,4 - polymer of isoprene done
clear
View Solution play_arrow
-
question_answer22)
Which of the following organic compounds polymerizes to form the polyester Dacron?
A)
Propylene and para \[HO-({{C}_{6}}{{H}_{4}})-OH\] done
clear
B)
Benzoic acid and ethanol done
clear
C)
Terephthalic acid and ethylene glycol done
clear
D)
Benzoic acid and para \[HO-({{C}_{6}}{{H}_{4}})-OH\] done
clear
View Solution play_arrow
-
question_answer23)
Biodegradable polymer which can be produced from glycine and aminocaproic acid is:
A)
PHBV done
clear
B)
Buna-N done
clear
C)
Nylon 6, 6 done
clear
D)
Nylon 2-nylon 6 done
clear
View Solution play_arrow
-
question_answer24)
Caprolactum is used for the manufacture of:
A)
Nylon-6 done
clear
B)
Teflon done
clear
C)
Terylene done
clear
D)
Nylon-6, 6 done
clear
View Solution play_arrow
-
question_answer25)
If a polythene sample contains two monodisperse fractions in the ratio 2 : 3 with degree of polymerization 100 and 200, respectively, then its weight average molecular weight will be:
A)
4900 done
clear
B)
4600 done
clear
C)
4300 done
clear
D)
5200 done
clear
View Solution play_arrow
-
question_answer26)
Synthetic polymer bakelite can be prepared from following compounds
A)
Styrene and vinyl chloride done
clear
B)
Acrylonitrile and vinyl chloride done
clear
C)
Adipic acid and ethylene glycol done
clear
D)
Phenol and formaldehyde done
clear
View Solution play_arrow
-
question_answer27)
Among cellulose, polyvinyl chloride, nylon and natural rubber, the polymer in which the intermolecular force of attraction is weakest is
A)
Nylon done
clear
B)
Polyvinyl chloride done
clear
C)
Cellulose done
clear
D)
Natural Rubber done
clear
View Solution play_arrow
-
question_answer28)
Which one of the following is an example of thermosetting polymers?
A)
Neoprene done
clear
B)
Buna-N done
clear
C)
Nylon 6, 6 done
clear
D)
Bakelite done
clear
View Solution play_arrow
-
question_answer29)
Which of the following polymer is a polyamide?
A)
Terylene done
clear
B)
Nylon done
clear
C)
Rubber done
clear
D)
Vulcanised rubber done
clear
View Solution play_arrow
-
question_answer30)
Ebonite is a
A)
natural rubber done
clear
B)
synthetic rubber done
clear
C)
highly vulcanized rubber done
clear
D)
polypropene done
clear
View Solution play_arrow
-
question_answer31)
Melamine plastic crockery is a condensation polymer of
A)
HCHO and melamine done
clear
B)
HCHO and ethylene done
clear
C)
melamine and ethylene done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer32)
Nylon 6,6 is a polyamide obtained by the reaction of
A)
\[COOH{{(C{{H}_{2}})}_{4}}COOH+N{{H}_{2}}{{C}_{6}}{{H}_{4}}N{{H}_{2}}-(p)\] done
clear
B)
\[COOH{{(C{{H}_{2}})}_{4}}COOH+N{{H}_{2}}{{(C{{H}_{2}})}_{6}}N{{H}_{2}}\] done
clear
C)
\[COOH{{(C{{H}_{2}})}_{6}}COOH+N{{H}_{2}}{{(C{{H}_{2}})}_{4}}N{{H}_{2}}\] done
clear
D)
\[COOH{{C}_{6}}{{H}_{4}}COOH-(p)+N{{H}_{2}}{{(C{{H}_{2}})}_{6}}N{{H}_{2}}\] done
clear
View Solution play_arrow
-
question_answer33)
Which of the following is not correctly represented?
A)
B)
C)
D)
View Solution play_arrow
-
question_answer34)
The monomer of the polymer;
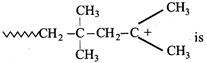
A)
B)
\[C{{H}_{3}}CH=CHC{{H}_{3}}\] done
clear
C)
\[C{{H}_{3}}CH=C{{H}_{2}}\] done
clear
D)
\[{{(C{{H}_{3}})}_{2}}C=C{{(C{{H}_{3}})}_{2}}\] done
clear
View Solution play_arrow
-
question_answer35)
Which of the following polymers do not involve cross linkages?
A)
Melmac done
clear
B)
Bakelite done
clear
C)
Polythene done
clear
D)
Vulcanised rubber done
clear
View Solution play_arrow
-
question_answer36)
Number average molecular mass, \[{{\overline{M}}_{n}}\] and weight average molecular mass \[\left( {{\overline{M}}_{w}} \right)\] of synthetic polymers are related as
A)
\[{{\overline{M}}_{n}}={{({{\overline{M}}_{w}})}^{1/2}}\] done
clear
B)
\[{{\overline{M}}_{n}}={{\overline{M}}_{w}}\] done
clear
C)
\[{{\overline{M}}_{w}}>{{\overline{M}}_{n}}\] done
clear
D)
\[{{\overline{M}}_{w}}<{{\overline{M}}_{n}}\] done
clear
View Solution play_arrow
-
question_answer37)
Polymer formation from monomers starts by
A)
condensation or addition reaction between monomers done
clear
B)
coordinate reaction between monomers done
clear
C)
conversion of monomer to monomer ions done
clear
D)
hydrolysis of monomers. done
clear
View Solution play_arrow
-
question_answer38)
\[n(C{{F}_{2}}=C{{F}_{2}})\xrightarrow[{{(N{{H}_{4}})}_{2}}{{S}_{2}}{{O}_{8}}]{Benzoyl\,peroxide\,or}X\] Here, X is:
A)
RMMA done
clear
B)
PVC done
clear
C)
PAN done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer39)
Which pair of polymers have similar properties?
A)
Nylon, PVC done
clear
B)
PAN, PTFE done
clear
C)
PCTFE, PTFE done
clear
D)
Bakelite, alkyl resin done
clear
View Solution play_arrow
-
question_answer40)
Among the following, the wrong statement is
A)
PMMA is plexiglass done
clear
B)
SBR is natural rubber done
clear
C)
PTFE is Teflon done
clear
D)
LDPE is low density polythene done
clear
View Solution play_arrow
-
question_answer41)
When condensation product of hexamethyiene- diamine and adipic acid is heated to 525 K in an atmosphere of nitrogen for about 4-5 hours, the product obtained is
A)
solid polymer of nylon 6, 6 done
clear
B)
liquid polymer of nylon 6, 6 done
clear
C)
gaseous polymer of nylon 6, 6 done
clear
D)
liquid polymer of nylon 6 done
clear
View Solution play_arrow
-
question_answer42)
In which of the following polymers, empirical formula resembles with monomer?
A)
Bakelite done
clear
B)
Teflon done
clear
C)
Nylon-6, 6 done
clear
D)
Dacron done
clear
View Solution play_arrow
-
question_answer43)
Which is a polymer of three different monomers?
A)
ABS done
clear
B)
SBR done
clear
C)
NBR done
clear
D)
Nylon-2, 6 done
clear
View Solution play_arrow
-
question_answer44)
The polymer which has conducting power is
A)
polyethylene done
clear
B)
polybutadiene done
clear
C)
polystyrene done
clear
D)
polyacetylene done
clear
View Solution play_arrow
-
question_answer45)
Which of the following compound is used for preparation of melamine formaldehyde polymer?
A)
B)
C)
D)
View Solution play_arrow
-
question_answer46)
Match the polymers in column - A with their main uses in column - B and choose the correct answer:
| Column - A |
Column - B |
| [A] Polystrene |
(i) Paints and lacquers |
| [B] Glyptal |
(ii) Rain coats |
| [C] Polyvinyl chloride |
(iii) Manufacture of toys |
| [D] Bakelite |
(iv) Computer discs |
A)
[A]-(ii), [B]-(i), [C]-(iii), [D]-(iv) done
clear
B)
[A]-(ii), [B]-(iv), [C]-(iii), [D]-(i) done
clear
C)
[A]-(iii), [B]-(iv), [C]-(ii), [D]-(i) done
clear
D)
[A]-(iii), [B]-(i), [C]-(ii), [D]-(iv) done
clear
View Solution play_arrow
-
question_answer47)
The compound which cannot be used as a plasticizer, is
A)
di-n-butylphthalate done
clear
B)
tricresyi phosphate done
clear
C)
di-ri-octyphthalate done
clear
D)
diethyl phthalate done
clear
View Solution play_arrow
-
question_answer48)
Which of the following statements is false?
A)
Artificial silk is derived from cellulose. done
clear
B)
Nylon-6, 6 is an example of elastomer. done
clear
C)
The repeat unit in natural rubber is isoprene. done
clear
D)
Both starch and cellulose are polymers of glucose. done
clear
View Solution play_arrow
-
question_answer49)
Which one of the following sets forms the biodegradable polymer?
A)
\[C{{H}_{2}}=CH-CN\] and \[C{{H}_{2}}=CH-CH=C{{H}_{2}}\] done
clear
B)
\[{{H}_{2}}N-C{{H}_{2}}-COOH\] and \[{{H}_{2}}N-{{(C{{H}_{2}})}_{5}}-COOH\] done
clear
C)
D)
View Solution play_arrow
-
question_answer50)
Which one of the following is an example of a thermosetting polymer?
A)
\[-(C{{H}_{2}}-\underset{Cl}{\mathop{\underset{|}{\mathop{C}}\,}}\,=CH-C{{H}_{2}}){{-}_{n}}\] done
clear
B)
\[-(C{{H}_{2}}-\underset{Cl}{\mathop{\underset{|}{\mathop{CH){{-}_{n}}}}\,}}\,\] done
clear
C)
\[-(\overset{H}{\mathop{\overset{|}{\mathop{N}}\,}}\,-{{(C{{H}_{2}})}_{6}}-\overset{H}{\mathop{\overset{|}{\mathop{N}}\,}}\,-\overset{O}{\mathop{\overset{||}{\mathop{C}}\,}}\,-{{(C{{H}_{2}})}_{4}}-\overset{O}{\mathop{\overset{||}{\mathop{C}}\,}}\,)-{{}_{n}}\] done
clear
D)
View Solution play_arrow
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
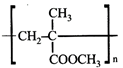 done
clear
done
clear

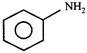 done
clear
done
clear
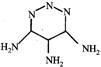 done
clear
done
clear
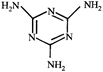 done
clear
done
clear
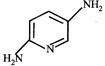 done
clear
done
clear
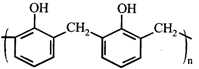 done
clear
done
clear
