-
question_answer1)
The order of reactivity of the following alcohols
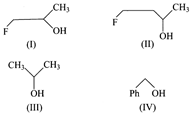
towards conc. HCl is:
A)
I> II> III>IV done
clear
B)
I> III> II,> IV done
clear
C)
IV> III> II>I done
clear
D)
IV> III> I> II done
clear
View Solution play_arrow
-
question_answer2)
Assign number 1 for least to 4 for most to indicate the relative base strength of the following:
| (I) \[{{C}_{6}}{{H}_{5}}N{{H}_{2}}\] |
| (II) \[p-N{{O}_{2}}{{C}_{6}}{{H}_{4}}N{{H}_{2}}\] |
| (III) \[m-N{{O}_{2}}{{C}_{6}}{{H}_{4}}N{{H}_{2}}\] |
| (IV) \[p-C{{H}_{3}}O{{C}_{6}}{{H}_{4}}N{{H}_{2}}\] |
A)
I - 3 II - 1 III - 2 IV - 4 done
clear
B)
I - 1 II - 2 III - 3 IV - 4 done
clear
C)
I - 2 II - 3 III - 4 IV - 1 done
clear
D)
I - 4 II - 1 III - 3 IV - 2 done
clear
View Solution play_arrow
-
question_answer3)
The correct order of +m effect of 'N' containing functional group on benzene ring, amongst the given compounds is
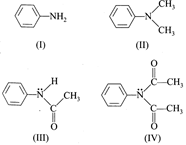
A)
I>II>IV>III done
clear
B)
II>I>III>IV done
clear
C)
I>II>III>IV done
clear
D)
IV>III>II>I done
clear
View Solution play_arrow
-
question_answer4)
The decreasing order of electron density on the ring is:
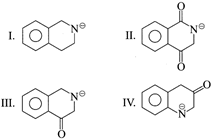
A)
(II)> (III)> (IV)> (I) done
clear
B)
(IV)> (I)> (II)> (III) done
clear
C)
(IV)> (I)> (III)> (II) done
clear
D)
(I)> (III)> (IV)> (II) done
clear
View Solution play_arrow
-
question_answer5)
Identify the aromatic compounds.
A)
B)
C)
D)
View Solution play_arrow
-
question_answer6)
The most stable carbocation is
A)
B)
C)
D)
View Solution play_arrow
-
question_answer7)
Arrange the following carbanions in decreasing order of stability.
A)
IV>III>I>II done
clear
B)
III>IV>II>I done
clear
C)
I>III>II>IV done
clear
D)
II>I>III>IV done
clear
View Solution play_arrow
-
question_answer8)
The stability order of following carbocation is
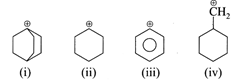
A)
i> ii> iv> iii done
clear
B)
iii> i> ii> iv done
clear
C)
iv> ii> iii> i done
clear
D)
ii> iv> iii> i done
clear
View Solution play_arrow
-
question_answer9)
Which one of the compound is not isomer of others?
A)
B)
C)
D)
View Solution play_arrow
-
question_answer10)
Which of the following is a pair of metamers?
A)
B)
C)
D)
View Solution play_arrow
-
question_answer11)
How many total structural isomers are obtained when three hydrogen atoms are replaced by chlorine atom from molecular formula \[{{C}_{3}}{{H}_{8}}\]
A)
5 done
clear
B)
4 done
clear
C)
6 done
clear
D)
3 done
clear
View Solution play_arrow
-
question_answer12)
A hydrocarbon with molecular formula \[{{C}_{6}}{{H}_{8}}\]is chiral but upon catalytic hydrogenation gives achiral hydrocarbon of molar mass 86. Which of the following could be the starting compound?
A)
3-methy1-1-pentene-4-yne done
clear
B)
1, 4-dimethylcyclobutadiene done
clear
C)
1-etheny1-3-methylcyclopropene done
clear
D)
5-methy1-1, 3-cyclopentadiene done
clear
View Solution play_arrow
-
question_answer13)
The correct IUPAC name of the compound
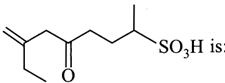
A)
6-Ethy1-1-methy1-4-oxohept-6-ene-1-suplphonic acid done
clear
B)
7-Ethy1-5-oxooct-7-ene-2-sulphonic acid done
clear
C)
2-Ethy1-7-sulphooct-1-ene-4-one done
clear
D)
7-Methylene-5-oxononane-2-sulphonic acid done
clear
View Solution play_arrow
-
question_answer14)
What is the correct IUPAC name of the following compound?
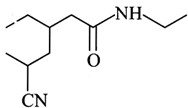
A)
N-Ethy1-5-cyano-3-ethy1 hexan-1-amide done
clear
B)
N-Ethy1-3-(2-cyanopropy1) pentanamide done
clear
C)
N, 3-Diethy1-5-cyanohexanamide done
clear
D)
3-(2-Cyanopropy1)-N-ethy1 pentanamide done
clear
View Solution play_arrow
-
question_answer15)
According to IUPAC convention, the systematic name of the compound given below is
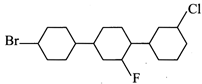
A)
4-(4-Bromocyclohexy1)-1-(3-chlorocy clohexy1)-2-flouorocyclohexane done
clear
B)
5-(4-Bromocyclohexy1)-2-(3-Chlorocy clohexy1)-1-flouorocyclohexane done
clear
C)
1-(3-chlorocyclohexy1)-4-(4-bromocy clohexy1)-2-flouorocyclohexane done
clear
D)
1-(4-Bromocyclohexy1)-4-(3-chlorocy clohexy1)-3-fluorocyclohexane done
clear
View Solution play_arrow
-
question_answer16)
What is the correct IUPAC name of the compound Shown below?
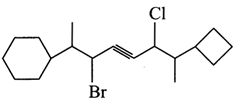
A)
5-Bromo-2-chloro-1-cyclobuty1-6cyclohexy1-1, 6-dimethy1-3-hexyne done
clear
B)
5-Bromo-2chloro-1-cyclobuty1-6-cyclohexy1-1-methy1-3-heptyne done
clear
C)
3-Bromo-6-chloro-7-cyclobuty1-2-cyclohexy1-4-octyne done
clear
D)
6-Bromo-3-chloro-2-cyclobuty1?7-cyclohexy1-4-oct-compound? done
clear
View Solution play_arrow
-
question_answer17)
What is the systematic IUPAC name of the following compound?
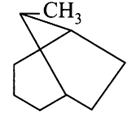
A)
1-Methy1 bicyclo [3, 2, 1] octane done
clear
B)
6-Methy1 bicyclo [3, 2, 1] octane done
clear
C)
8-Methy1 bicyclo [3, 2, 1] octane done
clear
D)
8-Methy1 tricyclo [3, 2, 1] octane done
clear
View Solution play_arrow
-
question_answer18)
For the detection of phosphorus, the organic compound after fusion with \[N{{a}_{2}}{{O}_{2}}\]is extracted with water, boiled with \[Na{{O}_{3}}\]and then ammonium molybdate is added to it, A yellow ppt. is obtained which is due to the formation of
A)
ammonium phosphate done
clear
B)
ammonium phosphomolybdate done
clear
C)
ferric phosphate done
clear
D)
disodium ammonium phosphate done
clear
View Solution play_arrow
-
question_answer19)
64 g of an organic compound contains 24 g of carbon, 8 g of hydrogen and the rest oxygen. The empirical formula of the compound is
A)
\[C{{H}_{2}}O\] done
clear
B)
\[{{C}_{2}}{{H}_{4}}O\] done
clear
C)
\[C{{H}_{4}}O\] done
clear
D)
\[{{C}_{2}}{{H}_{8}}{{O}_{2}}\] done
clear
View Solution play_arrow
-
question_answer20)
An organic compound of analysis gave c = 48 g H = 8 g and N = 56 g. Volume of 1.0 g of the compound was found to be 200 ml at NTP. Molecular formula of the compound is
A)
\[{{C}_{4}}{{H}_{8}}{{N}_{4}}\] done
clear
B)
\[{{C}_{2}}{{H}_{4}}{{N}_{2}}\] done
clear
C)
\[{{C}_{12}}{{H}_{24}}{{N}_{12}}\] done
clear
D)
\[{{C}_{16}}{{H}_{32}}{{N}_{16}}\] done
clear
View Solution play_arrow
-
question_answer21)
0.5 g of an organic compound on Kjeldahl's analysis gave enough ammonia to just neutralize 10 \[c{{m}^{3}}\]of M \[{{H}_{2}}S{{O}_{4}}\]The percentage of nitrogen in the compound is
View Solution play_arrow
-
question_answer22)
0.2595 g of an organic substance in a quantitative analysis yielded 0.35 g of the barium sulphate. The percentage of sulphur in the substance is
View Solution play_arrow
-
question_answer23)
The total number of contributing structures showing hyperconjugation for the following carbocation is
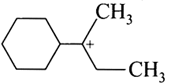
View Solution play_arrow
-
question_answer24)
Amongst the following, the total number of nucleophiles is: \[{{R}^{-}},\,\,O{{R}^{-}},\,\,{{H}_{2}}O,\text{ }S{{O}_{3}},\text{ }NH_{2}^{-},\text{ }C{{O}_{2}},\,\,ROH,\text{ }B{{F}_{3}},\,\,Al\overline{C}{{l}_{3}},\,\,{{H}^{-}}\]
View Solution play_arrow
-
question_answer25)
In 4-Formy1-2-methy1 benzoic acid, what is the maximum number of hydrogen atoms which lie in one single plane?
View Solution play_arrow
 towards conc. HCl is:
towards conc. HCl is: 
 done
clear
done
clear
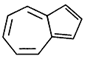 done
clear
done
clear
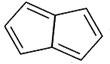 done
clear
done
clear
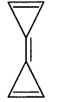 done
clear
done
clear
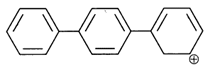 done
clear
done
clear
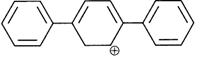 done
clear
done
clear
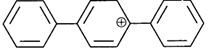 done
clear
done
clear
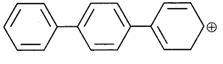 done
clear
done
clear


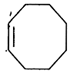 done
clear
done
clear
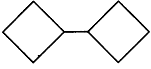 done
clear
done
clear
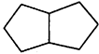 done
clear
done
clear
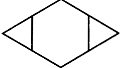 done
clear
done
clear
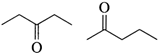 done
clear
done
clear
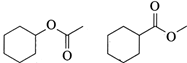 done
clear
done
clear
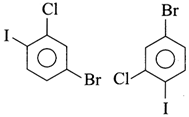 done
clear
done
clear





