-
question_answer1)
A large increase in the rate of a reaction for a rise in temperature is due to [EAMCET 1980; MP PET 1995]
A)
The decrease in the number of collisions done
clear
B)
The increase in the number of activated molecules done
clear
C)
The shortening of the mean free path done
clear
D)
The lowering of the activation energy done
clear
View Solution play_arrow
-
question_answer2)
Which of the following statements is not true according to collision theory of reaction rates
A)
Collision of molecules is a precondition for any reaction to occur done
clear
B)
All collisions result in the formation of the products done
clear
C)
Only activated collisions result in the formation of the products done
clear
D)
Molecules which have acquired the energy of activation can collide effectively done
clear
View Solution play_arrow
-
question_answer3)
According to the collision theory of chemical reactions
A)
A chemical reaction occurs with every molecular collision done
clear
B)
Rate is directly proportional to the number of collisions per second done
clear
C)
Reactions in the gas phase are always of zero order done
clear
D)
Reaction rates are of the order of molecular speeds done
clear
View Solution play_arrow
-
question_answer4)
According to the collision theory of reaction rates, rate of reaction increases with temperature due to
A)
Greater number of collisions done
clear
B)
Greater velocity of the reacting molecules done
clear
C)
Greater number of molecules have activation energy done
clear
D)
None of the above done
clear
View Solution play_arrow
-
question_answer5)
The reaction rate at a given temperature becomes slower, then [MP PMT 1993; DPMT 2000]
A)
The free energy of activation is higher done
clear
B)
The free energy of activation is lower done
clear
C)
The entropy changes done
clear
D)
The initial concentration of the reactants remains constant done
clear
View Solution play_arrow
-
question_answer6)
A rise in temperature increases the velocity of a reaction. It is because it results in
A)
An increased number of molecular collisions done
clear
B)
An increased momentum of colliding molecules done
clear
C)
An increase in the activation energy done
clear
D)
A decrease in the activation energy done
clear
View Solution play_arrow
-
question_answer7)
The number of collisions depend upon
A)
Pressure done
clear
B)
Concentration done
clear
C)
Temperature done
clear
D)
All the above done
clear
View Solution play_arrow
-
question_answer8)
If \[{{E}_{f}}\] and \[{{E}_{r}}\] are the activation energies of forward and reverse reactions and the reaction is known to be exothermic, then
A)
\[{{E}_{f}}>{{E}_{r}}\] done
clear
B)
\[{{E}_{f}}<{{E}_{r}}\] done
clear
C)
\[{{E}_{f}}={{E}_{r}}\] done
clear
D)
No relation can be given between \[{{E}_{f}}\] and \[{{E}_{r}}\] as data are not sufficient done
clear
View Solution play_arrow
-
question_answer9)
According to Arrhenius theory, the activation energy is
A)
The energy it should possess so that it can enter into an effective collision done
clear
B)
The energy which the molecule should possess in order to undergo reaction done
clear
C)
The energy it has to acquire further so that it can enter into a effective collision done
clear
D)
The energy gained by the molecules on colliding with another molecule done
clear
View Solution play_arrow
-
question_answer10)
The energy of activation is
A)
The energy associated with the activated molecules done
clear
B)
Threshold energy ? energy of normal molecules done
clear
C)
Threshold energy + energy of normal molecules done
clear
D)
Energy of products ? energy of reactants done
clear
View Solution play_arrow
-
question_answer11)
Which one of the following does not represent Arrhenius equation
A)
\[k=A{{e}^{-E/RT}}\] done
clear
B)
\[{{\log }_{e}}k={{\log }_{e}}A-\frac{E}{RT}\] done
clear
C)
\[{{\log }_{10}}k={{\log }_{10}}A-\frac{E}{2.303\,RT}\] done
clear
D)
\[k=A{{E}^{-RT}}\] done
clear
View Solution play_arrow
-
question_answer12)
On increasing the temperature, the rate of the reaction increases because of [MP PMT 1997]
A)
Decrease in the number of collisions done
clear
B)
Decrease in the energy of activation done
clear
C)
Decrease in the number of activated molecules done
clear
D)
Increase in the number of effective collisions done
clear
View Solution play_arrow
-
question_answer13)
Energy of activation of a reactant is reduced by
A)
Increased temperature done
clear
B)
Reduced temperature done
clear
C)
Reduced pressure done
clear
D)
Increased pressure done
clear
View Solution play_arrow
-
question_answer14)
The minimum energy a molecule should possess in order to enter into a fruitful collision is known as [Kurukshetra CEE 2002]
A)
Reaction energy done
clear
B)
Collision energy done
clear
C)
Activation energy done
clear
D)
Threshold energy done
clear
View Solution play_arrow
-
question_answer15)
Activation energy is
A)
The amount of energy to be added to the actual energy of a molecule so that the threshold energy is reached done
clear
B)
The amount of energy the molecule must contain so that it reacts done
clear
C)
The energy which a molecule should have in order to enter into an effective collision done
clear
D)
The average kinetic energy of the molecule done
clear
View Solution play_arrow
-
question_answer16)
The reason for almost doubling the rate of reaction on increasing the temperature of the reaction system by \[{{10}^{oC}}\] is [J & K 2005]
A)
The value of threshold energy increases done
clear
B)
Collision frequency increases done
clear
C)
The fraction of the molecule having energy equal to threshold energy or more increases done
clear
D)
Activation energy decreases done
clear
View Solution play_arrow
-
question_answer17)
The activation energy for a simple chemical reaction \[A\to B\] is \[{{E}_{a}}\] in forward direction. The activation energy for reverse reaction [CBSE PMT 2003]
A)
Is always double of \[{{E}_{a}}\] done
clear
B)
Is negative of \[{{E}_{a}}\] done
clear
C)
Is always less than \[{{E}_{a}}\] done
clear
D)
Can be less than or more than \[{{E}_{a}}\] done
clear
View Solution play_arrow
-
question_answer18)
Arrhenius equation is
A)
\[\frac{d\,\ln \,K}{dT}=\Delta {{E}^{*}}/RT\] done
clear
B)
\[\frac{d\,\ln \,K}{dT}=\Delta {{E}^{*}}/R{{T}^{2}}\] done
clear
C)
\[\frac{d\,\ln \,K}{dT}=-\Delta {{E}^{*}}/R{{T}^{2}}\] done
clear
D)
\[\frac{d\,\ln \,K}{dT}=-\Delta {{E}^{*}}/RT\] done
clear
View Solution play_arrow
-
question_answer19)
Activation energy of any reaction depends on
A)
Temperature done
clear
B)
Nature of reactants done
clear
C)
Number of collisions per unit time done
clear
D)
Concentration of reactants done
clear
View Solution play_arrow
-
question_answer20)
Relation between rate constant and temperature by Arrhenius equation is
A)
\[{{\log }_{e}}A={{\log }_{e}}K+\frac{{{E}_{a}}}{RT}\] done
clear
B)
\[\log \,K=A\,\frac{{{E}_{a}}}{RT}\] done
clear
C)
\[{{\log }_{e}}K={{\log }_{e}}A-\frac{{{E}_{a}}}{R{{T}^{2}}}\] done
clear
D)
\[\log A=RT\,\ln \,{{E}_{a}}-\ln \,K\] done
clear
View Solution play_arrow
-
question_answer21)
An endothermic reaction \[A\to B\] has an activation energy \[15\,kcal/mole\] and energy of reaction \[5\,kcal/mole\]. The activation energy of the reaction \[B\to A\] is [Pb. CET 1985]
A)
20 kcal/mole done
clear
B)
15 kcal/mole done
clear
C)
10 kcal/mole done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer22)
Which of the following plots is in accordance with the Arrhenius equation
A)
B)
C)
D)
View Solution play_arrow
-
question_answer23)
The Arrhenius equation expressing the effect of temperature on the rate constant of a reaction is [MP PET 1997]
A)
\[k={{e}^{-{{E}_{a}}/RT}}\] done
clear
B)
\[k={{E}_{a}}/RT\] done
clear
C)
\[k={{\log }_{e}}\,\frac{{{E}_{a}}}{RT}\] done
clear
D)
\[k=A{{e}^{-{{E}_{a}}/RT}}\] done
clear
View Solution play_arrow
-
question_answer24)
For a reaction, activation energy \[({{E}_{a}})=0\] and rate constant \[(K)=3.2\times {{10}^{6}}{{s}^{-1}}\]at 300 K. What is the value of the rate constant at 310 K [KCET (Med.) 1999]
A)
\[3.2\times {{10}^{-12}}{{s}^{-1}}\] done
clear
B)
\[3.2\times {{10}^{6}}{{s}^{-1}}\] done
clear
C)
\[6.4\times {{10}^{12}}{{s}^{-1}}\] done
clear
D)
\[6.4\times {{10}^{6}}{{s}^{-1}}\] done
clear
View Solution play_arrow
-
question_answer25)
Activation energy is given by the formula [DCE 1999]
A)
\[\log \frac{{{K}_{2}}}{{{K}_{1}}}=\frac{{{E}_{a}}}{2.303R}\left[ \frac{{{T}_{2}}-{{T}_{1}}}{{{T}_{1}}{{T}_{2}}} \right]\] done
clear
B)
\[\log \frac{{{K}_{1}}}{{{K}_{2}}}=-\frac{{{E}_{a}}}{2.303R}\left[ \frac{{{T}_{2}}-{{T}_{1}}}{{{T}_{1}}{{T}_{2}}} \right]\] done
clear
C)
\[\log \frac{{{K}_{1}}}{{{K}_{2}}}=-\frac{{{E}_{a}}}{2.303R}\left[ \frac{{{T}_{1}}-{{T}_{2}}}{{{T}_{1}}{{T}_{2}}} \right]\] done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer26)
A reaction having equal activation energies for forward and reverse reaction has [MP PMT 2002]
A)
\[\Delta H=0\] done
clear
B)
\[\Delta S=0\] done
clear
C)
Zero order done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer27)
Collision theory is applicable to [MP PMT 2002]
A)
First order reactions done
clear
B)
Zero order reactions done
clear
C)
Bimolecular reactions done
clear
D)
Intra molecular reactions done
clear
View Solution play_arrow
-
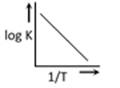
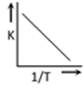
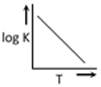
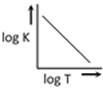
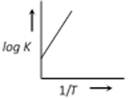
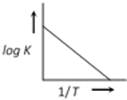
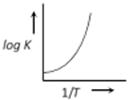
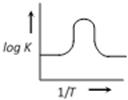
question_answer28)
A graph plotted between log K vs 1/T for calculating activation energy is shown by [MP PET 2002]
A)
B)
C)
D)
View Solution play_arrow
-
question_answer29)
The rate constant of a reaction at temperature 200K is 10 times less than the rate constant at 400 K. What is the activation energy \[({{E}_{a}})\]of the reaction (R = gas constant) [EAMCET 2003]
A)
1842.4 R done
clear
B)
921.2 R done
clear
C)
460.6 R done
clear
D)
230.3 R done
clear
View Solution play_arrow
-
question_answer30)
In respect of the equation \[k=A{{e}^{-{{E}_{a}}/RT}}\] in chemical kinetics, which one of the following statement is correct [AIEEE 2003]
A)
k is equilibrium constant done
clear
B)
A is adsorption factor done
clear
C)
\[{{E}_{a}}\]is energy of activation done
clear
D)
R is Rydberg?s constant done
clear
View Solution play_arrow
-
question_answer31)
The rate constant is doubled when temperature increases from 27°C to 37°C. Activation energy in kJ is [JEE Orissa 2004]
A)
34 done
clear
B)
54 done
clear
C)
100 done
clear
D)
50 done
clear
View Solution play_arrow
-
question_answer32)
The activation energy of a reaction is zero. The rate constant of this reaction
A)
Increases with increase of temperature done
clear
B)
Decreases with an increase of temperature done
clear
C)
Decreases with decrease of temperature done
clear
D)
Is independent of temperature done
clear
View Solution play_arrow
-
question_answer33)
The rate constant is given by the equation \[k=pz{{e}^{-E/RT}}\]. Which factor should register a decrease for the reaction to proceed more rapidly [MP PET/PMT 1998]
A)
\[T\] done
clear
B)
\[Z\] done
clear
C)
\[E\] done
clear
D)
\[p\] done
clear
View Solution play_arrow
-
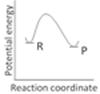
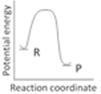
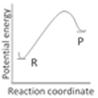
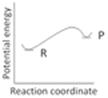
question_answer34)
An endothermic reaction with high activation energy for the forward reaction is given by the diagram: [AIIMS 2005]
A)
B)
C)
D)
View Solution play_arrow
-
question_answer35)
Consider an endothermic reaction \[X\to Y\] with the activation energies \[{{E}_{b}}\] and \[{{E}_{f}}\] for the backward and forward reactions, respectively, in general [AIEEE 2005]
A)
\[{{E}_{b}}<{{E}_{f}}\] done
clear
B)
\[{{E}_{b}}>{{E}_{f}}\] done
clear
C)
\[{{E}_{b}}={{E}_{f}}\] done
clear
D)
There is no definite relation between \[{{E}_{b}}\] and \[{{E}_{f}}\] done
clear
View Solution play_arrow
-
question_answer36)
Temperature dependent equation can be written as [Orrisa JEE 2005]
A)
\[\ln \,k=\ln A-{{e}^{{{E}_{a}}/RT}}\] done
clear
B)
\[\ln k=\ln A+{{e}^{{{E}_{a}}/RT}}\] done
clear
C)
\[\ln k=\ln A-{{e}^{RT/{{E}_{a}}}}\] done
clear
D)
All of these done
clear
View Solution play_arrow
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
