A)
B)
C)

D)

Correct Answer: A
Solution :
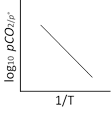
For the reaction, CaCO3(g) ⇌ CaO(s) + CO2(g) Kp = \[{{P}_{C{{O}_{2}}}}\] and KC = [CO2] (Q [CaCO3] = 1 and [CaO] = 1 for solids] According to Arrhenius equation we have \[K=A{{e}^{-\Delta H{{{}^\circ }_{r}}/RT}}\] Taking logarithm, we have \[\log {{K}_{p}}=\log A-\frac{\Delta H_{r}^{o}}{RT(2.303)}\] This is an equation of straight line. When log Kp is plotted against 1 / T. we get a straight line. The intercept of this line = log A, slope = ?∆H°r / 2.303 R Knowing the value of slope from the plot and universal gas constant R, ∆Hr° can be calculated. (Equation of straight line : Y = mx + C. Here, \[\log {{K}_{p}}=-\frac{\Delta H_{r}^{o}}{2.303R}\left( \frac{1}{T} \right)+\log A\] Y m x C
The intercept of this line = log A, slope = ?∆H°r / 2.303 R Knowing the value of slope from the plot and universal gas constant R, ∆Hr° can be calculated. (Equation of straight line : Y = mx + C. Here, \[\log {{K}_{p}}=-\frac{\Delta H_{r}^{o}}{2.303R}\left( \frac{1}{T} \right)+\log A\] Y m x C
You need to login to perform this action.
You will be redirected in
3 sec
