-
question_answer1)
Which of the following would not give 2-phenylbutane as the major product in a Friedel- Crafts alkylation reaction on benzene ring?
A)
1-butene+HF done
clear
B)
2-butanol \[+{{H}_{2}}S{{O}_{4}}\] done
clear
C)
Butanoyl chloride \[+AlC{{l}_{3}}\] then \[Zn,HCl\] done
clear
D)
Butyl chloride \[+AlC{{l}_{3}}\] done
clear
View Solution play_arrow
-
question_answer2)
Which of the following compounds are antiaromatic
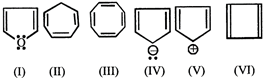
A)
(l) and (V) done
clear
B)
(II) and (V) done
clear
C)
(I) and (IV) done
clear
D)
(V) and (VI) done
clear
View Solution play_arrow
-
question_answer3)
Which one of the following reactions is expected to readily give a hydrocarbon product in good yields?
A)
\[RCOOK~\xrightarrow[oxidation]{Electrolytic}\] done
clear
B)
\[RCO{{O}^{-}}A{{g}^{+}}\xrightarrow{B{{r}_{2}}}\] done
clear
C)
\[C{{H}_{3}}C{{H}_{3}}\xrightarrow[hv]{C{{l}_{2}}}\] done
clear
D)
\[{{(C{{H}_{3}})}_{3}}CCl\ \xrightarrow{{{C}_{2}}{{H}_{5}}OH}\] done
clear
View Solution play_arrow
-
question_answer4)
[A]
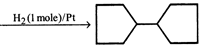
Double bond equivalent (degree of unsaturation) of [A] is:
A)
1 done
clear
B)
2 done
clear
C)
3 done
clear
D)
4 done
clear
View Solution play_arrow
-
question_answer5)
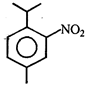
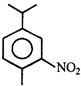
An alkene having molecular formula \[{{C}_{7}}{{H}_{14}}\] was subjected to ozonolysis in the presence of zinc dust. An equimolar amount of the following two compounds was obtained
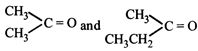
The IUPAC name of the alkene is
A)
3, 4-dimethyl-3-pentene done
clear
B)
3, 4-dimethyl-2-pentene done
clear
C)
2, 3-dimethyl-3-pentene done
clear
D)
2, 3-dimethyl-2-pentene done
clear
View Solution play_arrow
-
question_answer6)
Which of the following will yield a mixture of 2-chlorobutene and 3-chlorobutene on treatment with\[HCl\]?
A)
\[C{{H}_{2}}=C=CH-C{{H}_{3}}\] done
clear
B)
\[{{H}_{2}}C=\underset{C{{H}_{3}}}{\mathop{\underset{|}{\mathop{C}}\,}}\,-CH=C{{H}_{2}}\] done
clear
C)
\[C{{H}_{2}}=CH-CH=C{{H}_{2}}\] done
clear
D)
\[HC\equiv C-CH=C{{H}_{2}}\] done
clear
View Solution play_arrow
-
question_answer7)
Which of the following statements is incorrect regarding dehydrohalogenation of alkenes?
A)
During the reaction hydrogen atom is eliminated from the P - carbon atom. done
clear
B)
Rate of reaction for same alkyi group; Iodine > Bromine > Chlorine done
clear
C)
Rate of reaction; \[{{(C{{H}_{3}})}_{3}}C->{{(C{{H}_{3}})}_{2}}CH->C{{H}_{3}}C{{H}_{2}}-\] done
clear
D)
Only nature of halogen atom determine rate of the reaction. done
clear
View Solution play_arrow
-
question_answer8)
Which of the following will form alkynide?
A)
B)
C)
D)
\[C{{H}_{3}}-C\equiv C-C{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer9)
\[C{{H}_{3}}-\overset{C{{H}_{3}}}{\mathop{\overset{|}{\mathop{CH}}\,}}\,C\equiv CH~\xrightarrow{excess\,\,HBr}\] The product of the above reaction is:
A)
\[C{{H}_{3}}-\overset{C{{H}_{3}}}{\mathop{\overset{|}{\mathop{CH}}\,}}\,-\overset{Br}{\mathop{\overset{|}{\mathop{C}}\,}}\,={{\overset{Br}{\mathop{\overset{|}{\mathop{CH}}\,}}\,}_{2}}\] done
clear
B)
\[C{{H}_{3}}-\overset{C{{H}_{3}}}{\mathop{\overset{|}{\mathop{CH}}\,}}\,-\overset{Br}{\mathop{\overset{|}{\mathop{C}}\,}}\,=C{{H}_{2}}\] done
clear
C)
\[C{{H}_{3}}-\overset{C{{H}_{3}}}{\mathop{\overset{|}{\mathop{CH}}\,}}\,-\underset{Br}{\mathop{\underset{|}{\mathop{\overset{Br}{\mathop{\overset{|}{\mathop{C}}\,}}\,}}\,}}\,=C{{H}_{3}}\] done
clear
D)
\[C{{H}_{3}}-\overset{C{{H}_{3}}}{\mathop{\overset{|}{\mathop{CH}}\,}}\,-C{{H}_{2}}-\underset{Br}{\mathop{\underset{|}{\mathop{\overset{Br}{\mathop{\overset{|}{\mathop{CH}}\,}}\,}}\,}}\,\] done
clear
View Solution play_arrow
-
question_answer10)
What is the end product of the following sequences of operations? \[Ca{{C}_{2}}\xrightarrow{{{H}_{2}}O}A\xrightarrow[H{{g}^{2+}}]{dil.\,{{H}_{2}}S{{O}_{4}}}B\xrightarrow[{{H}_{2}}]{Ni}C\]
A)
Methyl alcohol done
clear
B)
Acetaldehyde done
clear
C)
\[{{C}_{2}}{{H}_{5}}OH\] done
clear
D)
\[{{C}_{2}}{{H}_{4}}\] done
clear
View Solution play_arrow
-
question_answer11)
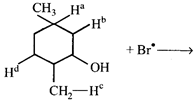
\[B{{r}^{\bullet }}\] will abstract which of the hydrogen most readily?
A)
a done
clear
B)
b done
clear
C)
c done
clear
D)
d done
clear
View Solution play_arrow
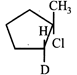
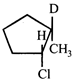
-
question_answer12)
Consider the following reaction \[{{H}_{3}}C-\underset{D}{\mathop{\underset{|}{\mathop{CH}}\,}}\,-\underset{C{{H}_{3}}}{\mathop{\underset{|}{\mathop{CH}}\,}}\,-C{{H}_{3}}+\overset{\bullet }{\mathop{B}}\,r\xrightarrow{{}}'X'+HBr\] Identify the structure of the major product 'X'
A)
\[{{H}_{3}}C-\underset{D}{\mathop{\underset{|}{\mathop{C}}\,}}\,H-\underset{C{{H}_{3}}}{\mathop{\underset{|}{\mathop{C}}\,}}\,H-\overset{\bullet }{\mathop{C}}\,{{H}_{2}}\] done
clear
B)
\[{{H}_{3}}C-\underset{D}{\mathop{\underset{|}{\mathop{C}}\,}}\,H-\overset{\bullet }{\mathop{\underset{C{{H}_{3}}}{\mathop{\underset{|}{\mathop{C}}\,}}\,}}\,-\overset{{}}{\mathop{C}}\,{{H}_{2}}\] done
clear
C)
\[{{H}_{3}}C-\overset{\bullet }{\mathop{\underset{D}{\mathop{\underset{|}{\mathop{C}}\,}}\,}}\,-\overset{{}}{\mathop{\underset{C{{H}_{3}}}{\mathop{\underset{|}{\mathop{CH}}\,}}\,}}\,-\overset{{}}{\mathop{C}}\,{{H}_{3}}\] done
clear
D)
\[{{H}_{3}}C-\overset{\bullet }{\mathop{CH}}\,-\overset{{}}{\mathop{\underset{C{{H}_{3}}}{\mathop{\underset{|}{\mathop{CH}}\,}}\,}}\,-\overset{{}}{\mathop{C}}\,{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer13)
Predict the product [A] of the following reaction
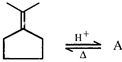
A)
B)
C)
D)
View Solution play_arrow
-
question_answer14)
Consider the following sequence of reactions \[C{{H}_{3}}CH=C{{H}_{2}}\xrightarrow[700k]{C{{l}_{2}}}A\xrightarrow[420k,\,12atm]{N{{a}_{2}}C{{O}_{3}}}\] \[B\xrightarrow[(ii)NaOH]{(i)\,HOCl}C\]
A)
B)
C)
\[HOC{{H}_{2}}-CH=C{{H}_{2}}\] done
clear
D)
\[C{{H}_{3}}\underset{OH}{\mathop{\underset{|}{\mathop{C}}\,}}\,OCl\] done
clear
View Solution play_arrow
-
question_answer15)
Choose the correct alkyne and reagents for the preparation of
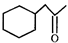
A)
B)
C)
D)
View Solution play_arrow
-
question_answer16)
The major product obtained in the following reaction
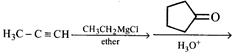
A)
B)
C)
D)
View Solution play_arrow
-
question_answer17)
A compound (X) \[({{C}_{5}}{{H}_{8}})\] reacts with ammonical \[AgN{{O}_{3}}\] to give a white precipitate, and on oxidation with hot alkaline \[KMn{{O}_{4}}\] gives the acid, \[{{(C{{H}_{3}})}_{2}}CHCOOH\], therefore X is -
A)
\[C{{H}_{2}}=CH-CH=CH-C{{H}_{3}}\] done
clear
B)
\[C{{H}_{3}}-CH=CH-C{{H}_{2}}-C{{H}_{3}}\] done
clear
C)
\[{{(C{{H}_{3}})}_{2}}CH-C=CH\] done
clear
D)
\[{{(C{{H}_{3}})}_{2}}C=C\equiv C{{H}_{2}}\] done
clear
View Solution play_arrow
-
question_answer18)
The major product of the following reaction
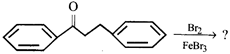
A)
B)
C)
D)
View Solution play_arrow
-
question_answer19)
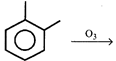
Which of the following products cannot be obtained in ozonolysis of o-xylene?
A)
B)
C)
D)
View Solution play_arrow
-
question_answer20)
Compare rate of dehydration of (I), (II) and (III) by cone.\[{{H}_{2}}S{{O}_{4}}\].
A)
(I) > (III) > (II) done
clear
B)
(I) > (II) > (Ill) done
clear
C)
(II) > (I) > (III) done
clear
D)
(II) > (III) > (I) done
clear
View Solution play_arrow
-
question_answer21)
In which step of the following reaction, \[s{{p}^{3}}\] carbon changes to \[s{{p}^{2}}\] carbon?
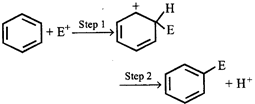
A)
Step 1 done
clear
B)
Step 2 done
clear
C)
Both done
clear
D)
None done
clear
View Solution play_arrow
-
question_answer22)
Toluene reacts with excess of \[C{{l}_{2}}\] in presence of sun light to give a product which on hydrolysis by reaction with \[NaOH\] gives
A)
B)
C)
D)
None of these done
clear
View Solution play_arrow
-
question_answer23)
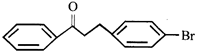
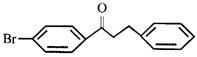
The major product formed in the reaction is:
A)
B)
C)
D)
View Solution play_arrow
-
question_answer24)
The major product obtained in the photo catalysed bromination of 2-methylbutane is:
A)
1-bromo-2-methylbutane done
clear
B)
1-bromo-3-methylbutane done
clear
C)
2-bromo-3-methylbutane done
clear
D)
2-bromo-2-methylbutane done
clear
View Solution play_arrow
-
question_answer25)
In the hydroboration - oxidation reaction of propene with diborane, \[{{H}_{2}}{{O}_{2}}\] and \[NaOH,\] the organic compound formed is:
A)
\[C{{H}_{3}}C{{H}_{2}}OH\] done
clear
B)
\[C{{H}_{3}}CHOHC{{H}_{3}}\] done
clear
C)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}OH\] done
clear
D)
\[{{(C{{H}_{3}})}_{3}}COH\] done
clear
View Solution play_arrow
-
question_answer26)
In the given reaction,
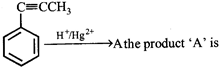
A)
B)
C)
D)
View Solution play_arrow
-
question_answer27)
The product of the reaction between ethyl benzene and N-bromosuccinamide is
A)
B)
C)
D)
View Solution play_arrow
-
question_answer28)
In preparation of alkene from alcohol using \[A{{l}_{2}}{{O}_{3}}\] which is effective factor?
A)
Porosity of \[A{{l}_{2}}{{O}_{3}}\] done
clear
B)
Temperature done
clear
C)
Concentration done
clear
D)
Surface area of \[A{{l}_{2}}{{O}_{3}}\] done
clear
View Solution play_arrow
-
question_answer29)
Which one of the following compounds would have the highest heat of hydrogenation?
A)
\[C{{H}_{2}}=C{{H}_{2}}\] done
clear
B)
\[C{{H}_{3}}-C{{H}_{2}}-CH=C{{H}_{2}}\] done
clear
C)
\[C{{H}_{3}}CH=CHC{{H}_{3}}\] done
clear
D)
\[{{\left( C{{H}_{3}} \right)}_{2}}C=C{{\left( C{{H}_{3}} \right)}_{2}}\] done
clear
View Solution play_arrow
-
question_answer30)
Which of the following types of reaction occur when a reactant has got a double bond?
| (i) Addition |
| (ii) Photolysis |
| (iii) Nucleophilic substitution |
| (iv) Polymerization |
A)
(i) and (iv) done
clear
B)
(i) and (ii) done
clear
C)
(i), (ii) and (iv) done
clear
D)
(i),(ii),(iii) and (iv) done
clear
View Solution play_arrow
-
question_answer31)
\[HOCl\] reacts on 3-methyl-2-pentene, the main product will be:
A)
\[C{{H}_{3}}-CH=\underset{Cl}{\mathop{\underset{|}{\mathop{C}}\,}}\,-CH-(OH)-C{{H}_{3}}\] done
clear
B)
\[C{{H}_{3}}-\overset{OH}{\mathop{\overset{|}{\mathop{C}}\,}}\,H-\overset{C{{H}_{3}}}{\mathop{\overset{|}{\mathop{\underset{Cl}{\mathop{\underset{|}{\mathop{C}}\,}}\,}}\,}}\,-C{{H}_{2}}-C{{H}_{3}}\] done
clear
C)
\[C{{H}_{3}}-\overset{Cl}{\mathop{\overset{|}{\mathop{C}}\,}}\,H-\overset{C{{H}_{3}}}{\mathop{\overset{|}{\mathop{\underset{OH}{\mathop{\underset{|}{\mathop{C}}\,}}\,}}\,}}\,-C{{H}_{2}}-C{{H}_{3}}\] done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer32)
The major product of the following reaction sequence is:
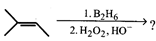
A)
B)
C)
D)
View Solution play_arrow
-
question_answer33)
All the hydrocarbons shown are very weak acids. One, however, is far more acidic than the others. Which one is the strongest acid?
A)
B)
C)
D)
View Solution play_arrow
-
question_answer34)
Which one amongst the following carbocations is most stable?
A)
\[{{C}_{6}}{{H}_{5}}-\overset{+}{\mathop{C}}\,H-{{C}_{6}}{{H}_{5}}\] done
clear
B)
C)
\[C{{H}_{3}}-\underset{C{{H}_{3}}}{\mathop{\underset{|}{\overset{+}{\mathop{C}}}\,}}\,-C{{H}_{3}}\] done
clear
D)
View Solution play_arrow
-
question_answer35)
Toluene on treatment with \[Cr{{O}_{3}}\] and \[{{(C{{H}_{3}}CO)}_{2}}O\] followed by hydrolysis with dil. \[HCl\] gives
A)
benzaldehyde done
clear
B)
benzoicacid done
clear
C)
phenol done
clear
D)
phenylacetaldehyde done
clear
View Solution play_arrow
-
question_answer36)
What is the correct product of reaction?
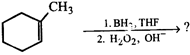
A)
B)
C)
D)
View Solution play_arrow
-
question_answer37)
Of the three isomeric \[{{C}_{3}}{{H}_{4}}\] hydrocarbons shown below how many can exist with all carbon and hydrogen nuclei located in a single plane
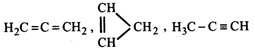
A)
0 done
clear
B)
1 done
clear
C)
2 done
clear
D)
3 done
clear
View Solution play_arrow
-
question_answer38)
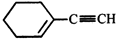
\[\xrightarrow[(ii)\,C{{H}_{3}}Br]{(i)\,NaN{{H}_{2}};N{{H}_{3}}}(A)\xrightarrow[Lindlar\,catalyst]{{{H}_{2}}}(B);\] Product (B) is:
A)
B)
C)
D)
View Solution play_arrow
-
question_answer39)
Among the following free radical bromination reactions, select those in which \[2{}^\circ \] halide is the major product
A)
P, Q, R, S done
clear
B)
P, R, U done
clear
C)
P, R, S, T done
clear
D)
P, Q, R, S, T done
clear
View Solution play_arrow
-
question_answer40)
The gas liberated by the electrolysis of dipotassium succinate solution is:
A)
Ethane done
clear
B)
Ethyne done
clear
C)
Ethene done
clear
D)
Propene done
clear
View Solution play_arrow
-
question_answer41)
In the presence of peroxide, \[HCl\] and HI do not give anti-Markownikoff's addition of alkenes because:
A)
One of the steps is endothermic in \[HCl\] and HI done
clear
B)
Both \[HCl\] and HI are strong acids done
clear
C)
\[HCl\] is oxidizing and the HI is reducing done
clear
D)
All the steps are exothermic is \[HCl\] and HI done
clear
View Solution play_arrow
-
question_answer42)
Consider the following sequence of reactions \[C{{H}_{3}}CH=C{{H}_{2}}\xrightarrow[700k]{C{{l}_{2}}}A\xrightarrow[420k,\,12\,atm]{N{{a}_{2}}C{{O}_{3}}}B\] Compound 'B' is
A)
B)
\[C{{H}_{3}}CH\underset{OH}{\mathop{\underset{|}{\mathop{C}}\,}}\,OONa\] done
clear
C)
\[HOC{{H}_{2}}-CH=C{{H}_{2}}\] done
clear
D)
\[C{{H}_{3}}\underset{OH}{\mathop{\underset{|}{\mathop{C}}\,}}\,HOCl\] done
clear
View Solution play_arrow
-
question_answer43)
How many monochlorolmtanes will be obtained on chlorination of n-butane?
A)
5 done
clear
B)
2 done
clear
C)
3 done
clear
D)
4 done
clear
View Solution play_arrow
-
question_answer44)
The cycloalkane having the lowest heat of combustion per \[C{{H}_{2}}\] group
A)
B)
C)
D)
View Solution play_arrow
-
question_answer45)
Which one of the following conformations of cyclohexane is chiral?
A)
Boat done
clear
B)
Twist boat done
clear
C)
Rigid done
clear
D)
Chair done
clear
View Solution play_arrow
-
question_answer46)
Which one of the following compounds gives acetone \[{{(C{{H}_{3}})}_{2}}C=0\] as one of the product of its ozonolysis?
A)
B)
C)
D)
View Solution play_arrow
-
question_answer47)
A)
\[{{H}_{2}}O/{{H}^{+}}\] done
clear
B)
\[B{{H}_{3}},THF/{{H}_{2}}{{O}_{2}}-O{{H}^{-}}\] done
clear
C)
\[Hg{{(OCOC{{H}_{3}})}_{2}},{{H}_{2}}O/NaB{{H}_{4}}.NaOH\] done
clear
D)
All are possible done
clear
View Solution play_arrow
-
question_answer48)
In reaction sequence \[C{{H}_{2}}=C{{H}_{2}}\xrightarrow[acid]{Hypochlorous}M\xrightarrow{R}\underset{C{{H}_{2}}OH}{\overset{C{{H}_{2}}OH}{\mathop{|}}}\,\] molecule 'M' and reagent 'R' respectively are
A)
\[C{{H}_{3}}C{{H}_{2}}Cl\] and \[NaOH\] done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}OH\] and \[{{H}_{2}}S{{O}_{4}}\] done
clear
C)
\[C{{H}_{2}}(Cl)C{{H}_{2}}OH\] and aqueous \[NaHC{{O}_{3}}\] done
clear
D)
View Solution play_arrow
-
question_answer49)
What is the major product expected from the following reaction?
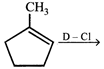
Where D is an isotope of hydrogen
A)
B)
C)
D)
View Solution play_arrow
-
question_answer50)
The reagent needed for converting

A)
Cat. Hydrogenation done
clear
B)
\[{{H}_{2}}\]/Lindlar Cat. done
clear
C)
\[Li/N{{H}_{3}}\] done
clear
D)
\[LiAl{{H}_{4}}\] done
clear
View Solution play_arrow
-
question_answer51)
Addition of HI to double bond of propene yields isopropyl iodide and not n-propyl iodide as the major product, because addition proceeds through
A)
a more stable carbonium ion done
clear
B)
a more stabi e carban ion done
clear
C)
a more stable free radical done
clear
D)
homolysis done
clear
View Solution play_arrow
-
question_answer52)
The principal organic product formed in the reaction, \[C{{H}_{2}}=CH{{(C{{H}_{2}})}_{8}}COOH+HBr\,\xrightarrow{peroxide}\] is
A)
\[C{{H}_{3}}CHBr{{(C{{H}_{2}})}_{8}}COOH\] done
clear
B)
\[C{{H}_{2}}=CH{{(C{{H}_{2}})}_{8}}COBr\] done
clear
C)
\[C{{H}_{2}}BrC{{H}_{2}}{{(C{{H}_{2}})}_{8}}COOH\] done
clear
D)
\[C{{H}_{2}}=CH{{(C{{H}_{2}})}_{7}}CHBrCOOH\] done
clear
View Solution play_arrow
-
question_answer53)
Acid catalyzed hydration of alkenes except ethane leads to the formation of
A)
mixture of secondary and tertiary alcohols done
clear
B)
mixture of primary and secondary alcohols done
clear
C)
secondary or tertiary alcohol done
clear
D)
primary alcohol done
clear
View Solution play_arrow
-
question_answer54)
Identify the reagent from the following list which can easily distinguish between 1-butyne and 2-butyne
A)
bromine, \[CC{{l}_{4}}\] done
clear
B)
\[{{H}_{2}}\], Lindlar catalyst done
clear
C)
dilute \[{{H}_{2}}S{{O}_{4}}\], \[HgS{{O}_{4}}\] done
clear
D)
ammonical \[C{{u}_{2}}C{{l}_{2}}\] solution done
clear
View Solution play_arrow
-
question_answer55)
An unknown compound A has a molecular formula \[{{C}_{2}}{{H}_{6}}\], when A is treated with an excess of\[B{{r}_{2}}\], a new substance B with formula \[{{C}_{4}}{{H}_{6}}B{{r}_{2}}\]is formed. A forms a white precipitate with ammonical silver nitrate solution. A may be
A)
Butyne-1 done
clear
B)
Butyne-2 done
clear
C)
Butene-1 done
clear
D)
Butene-2 done
clear
View Solution play_arrow
-
question_answer56)
The major product formed in the reaction is:
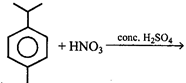
A)
B)
C)
D)
View Solution play_arrow
-
question_answer57)
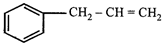
On mercuration-demercuration produces the major product:
A)
B)
C)
D)
View Solution play_arrow
-
question_answer58)
Which one of the following class of compounds is obtained by polymerization of acetylene?
A)
Poly-yne done
clear
B)
Poly-ene done
clear
C)
Poly-ester done
clear
D)
Poly-amine done
clear
View Solution play_arrow
-
question_answer59)
The hydration of propyne results in formation of
A)
Acetone done
clear
B)
Propanol-1 done
clear
C)
Propene done
clear
D)
Propanal done
clear
View Solution play_arrow
-
question_answer60)
A)
B)
C)
D)
View Solution play_arrow
-
question_answer61)
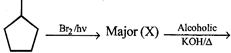
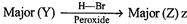
is:
A)
B)
C)
D)
View Solution play_arrow
-
question_answer62)
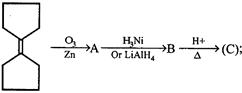
Product of the reaction is:
A)
B)
C)
D)
View Solution play_arrow
-
question_answer63)
Which of the following change is correct
A)
B)
C)
D)
View Solution play_arrow
-
question_answer64)
Which of the following change is correct
A)
B)
C)
Both [a] and [b] one correct done
clear
D)
Neither [a] nor [b] done
clear
View Solution play_arrow
-
question_answer65)
Choose the correct product of the following reaction
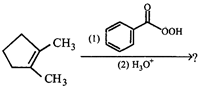
A)
B)
C)
D)
View Solution play_arrow
-
question_answer66)
The two compounds A and B obtained from 1-butyne can be distinguished by \[B\xleftarrow[(ii)\,{{H}_{2}}{{O}_{2}}]{(i)\,B{{H}_{3}}}C{{H}_{3}}C{{H}_{2}}C\equiv CH\xrightarrow{{{H}^{+}}/H{{g}^{2+}}}A\]
A)
\[NaHS{{O}_{3}}\] done
clear
B)
litmus solution done
clear
C)
iodoform test done
clear
D)
2, 4-DNP done
clear
View Solution play_arrow
-
question_answer67)
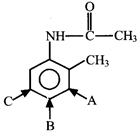
Identify the position where electrophilic aromatic substitution (EAS) is most favourable.
A)
A done
clear
B)
B done
clear
C)
C done
clear
D)
A and C done
clear
View Solution play_arrow
-
question_answer68)
Arrange the following in decreasing order of reactivity towards EAS (electrophilic aromatic substitution)
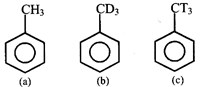
A)
a > b > c done
clear
B)
c > b > a done
clear
C)
a > c > b done
clear
D)
c > a > b done
clear
View Solution play_arrow
-
question_answer69)
Which of the following is an aromatic species?
A)
B)
C)
D)
All of the three done
clear
View Solution play_arrow
-
question_answer70)
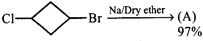
Product [A] of above reaction is:
A)
B)
C)
D)
View Solution play_arrow

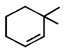
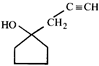
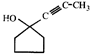
![]() Double bond equivalent (degree of unsaturation) of [A] is:
Double bond equivalent (degree of unsaturation) of [A] is:  done
clear
done
clear
 done
clear
done
clear
 \[B{{r}^{\bullet }}\] will abstract which of the hydrogen most readily?
\[B{{r}^{\bullet }}\] will abstract which of the hydrogen most readily? 
 done
clear
done
clear
![]()
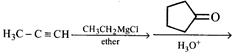
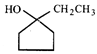 done
clear
done
clear
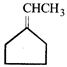 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear

 done
clear
done
clear
 done
clear
done
clear
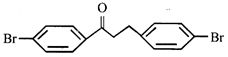 done
clear
done
clear
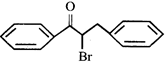 done
clear
done
clear
 Which of the following products cannot be obtained in ozonolysis of o-xylene?
Which of the following products cannot be obtained in ozonolysis of o-xylene? 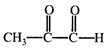 done
clear
done
clear
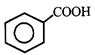 done
clear
done
clear
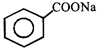 done
clear
done
clear
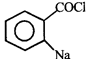 done
clear
done
clear

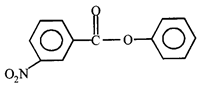 done
clear
done
clear
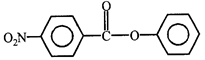 done
clear
done
clear
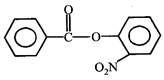 done
clear
done
clear
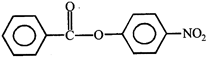 done
clear
done
clear
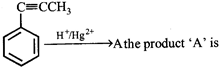
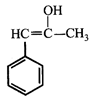 done
clear
done
clear
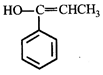 done
clear
done
clear
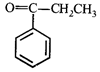 done
clear
done
clear
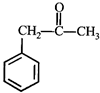 done
clear
done
clear
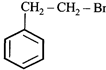 done
clear
done
clear
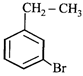 done
clear
done
clear
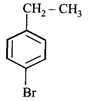 done
clear
done
clear
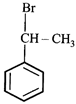 done
clear
done
clear
![]()
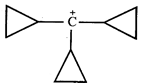 done
clear
done
clear
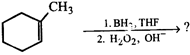
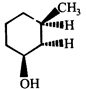 done
clear
done
clear
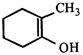 done
clear
done
clear
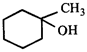 done
clear
done
clear
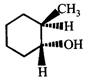 done
clear
done
clear
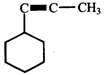 done
clear
done
clear
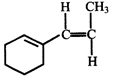 done
clear
done
clear
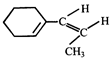 done
clear
done
clear
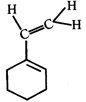 done
clear
done
clear
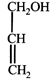 done
clear
done
clear
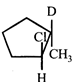 done
clear
done
clear
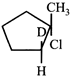 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
![]()

 done
clear
done
clear
 done
clear
done
clear
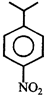 done
clear
done
clear
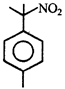 done
clear
done
clear
![]() On mercuration-demercuration produces the major product:
On mercuration-demercuration produces the major product: 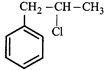 done
clear
done
clear
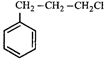 done
clear
done
clear
 done
clear
done
clear
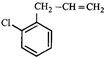 done
clear
done
clear
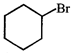 done
clear
done
clear
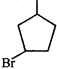 done
clear
done
clear
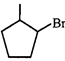 done
clear
done
clear
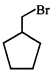 done
clear
done
clear
 Product of the reaction is:
Product of the reaction is: 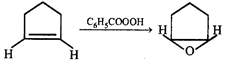 done
clear
done
clear
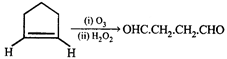 done
clear
done
clear
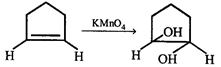 done
clear
done
clear
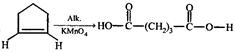 done
clear
done
clear
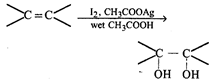 done
clear
done
clear
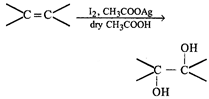 done
clear
done
clear

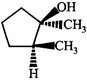 done
clear
done
clear
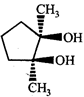 done
clear
done
clear
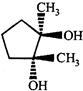 done
clear
done
clear
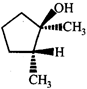 done
clear
done
clear
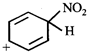 done
clear
done
clear
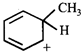 done
clear
done
clear
![]() Product [A] of above reaction is:
Product [A] of above reaction is: 