-
question_answer1)
Which of the following statement is correct?
A)
Cathode is -ve terminal in both, galvanic and electrolytic cells. done
clear
B)
Anode is +ve terminal in both, galvanic and electrolytic cells. done
clear
C)
Cathode and anode are -ve terminal in electrolytic and galvanic cell. done
clear
D)
Cathode and anode are +ve terminal in electrolytic and galvanic cell. done
clear
View Solution play_arrow
-
question_answer2)
Based on the cell notation for a spontaneous reaction, at the anode: \[Ag\left( s \right)\left| AgCl\left( s \right) \right|C{{l}^{-}}\left( aq \right)\left| \left| B{{r}^{-}}\left( aq \right) \right|B{{r}_{2}}\left( l \right) \right|C\left( s \right)\]
A)
\[AgCl\] gets reduced done
clear
B)
Ag gets oxidized done
clear
C)
\[B{{r}^{-}}\] gets oxidized done
clear
D)
\[B{{r}_{2}}\] gets reduced done
clear
View Solution play_arrow
-
question_answer3)
For the electrochemical cell \[Pt(s)\underset{1\text{ }atm}{\mathop{|{{H}_{2}}(g)|}}\,{{H}^{+}}(1M)|,Cu(s)\] which one of the following statements is true?
A)
\[{{H}^{+}}\] ions are formed at anode and Cu is deposited at cathode. done
clear
B)
\[{{H}_{2}}\] is liberated at cathode and Cu is deposited at anode. done
clear
C)
Oxidation occurs at cathode. done
clear
D)
Reduction occurs at anode. done
clear
View Solution play_arrow
-
question_answer4)
The standard reduction potentials at 298K for the following half reactions are given against each
| \[Z{{n}^{2+}}(aq)+2{{e}^{-}}\rightleftharpoons Zn(s);\,\,-0.762\,V\] |
| \[C{{r}^{3+}}(aq)+3{{e}^{-}}~\rightleftharpoons Cr(s);\,\,-0.740\,V\] |
| \[2{{H}^{+}}(aq)+2{{e}^{-}}\rightleftharpoons {{H}_{2}}(g);\,\,\,0.00\,V\] |
| \[F{{e}^{3+}}(aq)+2{{e}^{-}}\rightleftharpoons F{{e}^{2+}}(aq);\,\,0.770\,V\] |
| Which is the strongest reducing agent? |
A)
\[Zn\left( s \right)\] done
clear
B)
\[Cr\left( s \right)\] done
clear
C)
\[{{H}_{2}}\left( g \right)\] done
clear
D)
\[F{{e}^{3+}}(aq)\] done
clear
View Solution play_arrow
-
question_answer5)
Zn gives \[{{H}_{2}}\] gas with \[{{H}_{2}}S{{O}_{4}}\] and \[HCl\] but not with \[HN{{O}_{3}}\] because
A)
Zn acts as oxidizing when reacts with \[HN{{O}_{3}}\]. done
clear
B)
\[HN{{O}_{3}}\] is weaker acid than \[{{H}_{2}}S{{O}_{4}}\] and \[HCl\]. done
clear
C)
In electrochemical series Zn is above hydrogen. done
clear
D)
\[N{{O}_{3}}^{-}\] is reduced in preference to hydronium. done
clear
View Solution play_arrow
-
question_answer6)
A smuggler could not carry gold by depositing iron on the gold surface since
A)
gold is denser done
clear
B)
iron rusts done
clear
C)
gold has higher reduction potential than iron done
clear
D)
gold has lower reduction potential than iron done
clear
View Solution play_arrow
-
question_answer7)
Standard electrode potential for \[S{{n}^{4+}}/S{{n}^{2+}}\] couple is + 0.15 V and that for the \[C{{r}^{3+}}/Cr\] couple is\[-0.74V\]. These two couples in their standard state are connected to make a cell. The cell potential will be:
A)
+1.19 V done
clear
B)
+0.89 V done
clear
C)
+0.18 V done
clear
D)
+1.83 V done
clear
View Solution play_arrow
-
question_answer8)
A solution contains \[F{{e}^{2+}},F{{e}^{3+}}\] and \[{{I}^{-}}\] ions. This solution was treated with iodine at\[35{}^\circ C\]. \[E{}^\circ \] for \[F{{e}^{3+}}/F{{e}^{2+}}\] is + 0.77 V and \[E{}^\circ \] for \[{{I}_{2}}/2{{I}^{-}}=0.536V.\] he favourable redox reaction is:
A)
\[{{I}_{2}}\] will be reduced to \[{{I}^{-}}\] done
clear
B)
There will be no redox reaction done
clear
C)
\[{{I}^{-}}\] will be oxidised to \[{{I}_{2}}\] done
clear
D)
\[F{{e}^{2+}}\] will be oxidised to \[F{{e}^{3+}}\] done
clear
View Solution play_arrow
-
question_answer9)
Consider the following four electrodes:
| \[P=C{{u}^{2+}}(0.0001M)/Cu(s)\] |
| \[Q=C{{u}^{2+}}\left( 0.1\text{ }M)/Cu(s \right)\] |
| \[R=C{{u}^{2+}}(0.01M/)Cu(s)\] |
| \[S=C{{u}^{2+}}(0.001M/)Cu(s)\] |
| If the standard reduction potential of \[C{{u}^{2+}}/Cu\] is +0.34 V, the reduction potentials in volts of the above electrodes follow the order. |
A)
\[P>S>R>Q\] done
clear
B)
\[S>R>Q>P\] done
clear
C)
\[R>S>Q>P\] done
clear
D)
\[Q>R>S>P\] done
clear
View Solution play_arrow
-
question_answer10)
Following cell has EMF 0.7995V. \[Pt|{{H}_{2}}(1\,atm)|HN{{O}_{3}}(1M)||AgN{{O}_{3}}(1M)|Ag\] If we add enough \[KCl\] to the Ag cell so that the final \[C{{l}^{-}}\] is 1M. Now the measured emf of the cell is 0.222V. The \[{{K}_{sp}}\] of \[AgCl\] would be -
A)
\[1\times {{10}^{-9.8}}\] done
clear
B)
\[1\times {{10}^{-19.6}}\] done
clear
C)
\[2\times {{10}^{-10}}\] done
clear
D)
\[2.64\times {{10}^{-14}}\] done
clear
View Solution play_arrow
-
question_answer11)
Standard cell voltage for the cell \[Pb|P{{b}^{2+}}||S{{n}^{2+}}\] is - 0.01 V. If the cell is to exhibit \[{{E}_{cell}}=0\], the value of \[[S{{n}^{2+}}]/[P{{b}^{2+}}]\] should be antilog of-
A)
+0.3 done
clear
B)
0.5 done
clear
C)
1.5 done
clear
D)
\[-0.5\] done
clear
View Solution play_arrow
-
question_answer12)
An electrochemical cell is set up as: \[Pt;{{H}_{2}}(1\,atm)|HCl(0.1M)||C{{H}_{3}}COOH(0.1M)|{{H}_{2}}\](1atm); Pt. The e.m.f of this cell will not be zero, because
A)
the temperature is constant done
clear
B)
e.m.f depends on molarities of acids used done
clear
C)
acids used in two compartments are different done
clear
D)
pH of \[0.1\text{ }M\text{ }HCl\] and \[0.1\,M\,C{{H}_{3}}COOH\] is not same done
clear
View Solution play_arrow
-
question_answer13)
A variable, opposite external potential \[({{E}_{ext}})\] is applied to the cell \[Zn|Z{{n}^{2+}}(1M)||C{{u}^{2+}}(1\,M)|Cu,\] of potential 1.1 V. When \[{{E}_{ext}}<1.1V\] and \[{{E}_{ext}}>1.1V\], respectively electrons flow from:
A)
anode to cathode in both cases done
clear
B)
cathode to anode and anode to cathode done
clear
C)
anode to cathode and cathode to anode done
clear
D)
cathode to anode in both cases done
clear
View Solution play_arrow
-
question_answer14)
The standard electrode potentials \[\left( E{{{}^\circ }_{{{M}^{+}}/M}} \right)\] of four metals A, B, C and D are\[-1.2V,\text{ }0.6V,\text{ }0.85V\] and \[-0.76\text{ }V,\] respectively. The sequence of deposition of metals on applying potential is:
A)
A, C, B, D done
clear
B)
B, D, C, A done
clear
C)
C, B, D, A done
clear
D)
D, A, B, C done
clear
View Solution play_arrow
-
question_answer15)
A current of 10.0 A flows for 2.00 h through an electrolytic cell containing a molten salt of metal X. This results in the decomposition of \[0.250\text{ }mol\] of metal X at the cathode. The oxidation state of X in the molten salt is: \[\left( F=96,500C \right)\]
A)
\[{{1}^{+}}\] done
clear
B)
\[{{2}^{+}}\] done
clear
C)
\[{{3}^{+}}\] done
clear
D)
\[{{4}^{+}}\] done
clear
View Solution play_arrow
-
question_answer16)
Which of the following will form a cell with the highest voltage?
A)
\[1\,MA{{g}^{+}},1\,MC{{o}^{2+}}\] done
clear
B)
\[2M\,A{{g}^{+}},2\,MC{{o}^{2+}}\] done
clear
C)
\[0.1\,M\,A{{g}^{+}},2\,MC{{o}^{2+}}\] done
clear
D)
\[2\,M\,A{{g}^{+}},0.1\,MC{{o}^{2+}}\] done
clear
View Solution play_arrow
-
question_answer17)
The standard EMF for the cell reaction, \[Zn+C{{u}^{2+}}\xrightarrow{{}}Cu+Z{{n}^{2+}}\] is 1.1 volt at \[25{}^\circ C.\] The EMF for the cell reaction, when \[0.1\,MC{{u}^{2+}}\] and \[0.1MZ{{n}^{2+}}\] solutions are used, at \[25{}^\circ C\] is
A)
1.10 V done
clear
B)
0.10 V done
clear
C)
\[-1.10\text{ }V\] done
clear
D)
\[-0.110\text{ }V\] done
clear
View Solution play_arrow
-
question_answer18)
Consider the following reactions
| (i) \[C{{d}^{2+}}\left( aq \right)+2{{e}^{-}}\xrightarrow{{}}Cd\left( s \right),E{}^\circ =-0.40V\] |
| (ii) \[A{{g}^{+}}(aq)+{{e}^{-}}\xrightarrow{{}}Ag(s),E{}^\circ =0.80V\] |
| For the galvanic cell involving the above reactions. Which of the following is not correct? |
A)
Molar concentration of the cation in the cathodic compartment changes faster than that of the cation in the anodic compartment. done
clear
B)
\[{{E}_{cell}}\] increase when \[C{{d}^{2+}}\] solution is diluted. done
clear
C)
Twice as many electrons pass through the cadmium electrode as through silver electrode. done
clear
D)
\[{{E}_{cell}}\] decreases when \[A{{g}^{+}}\] solution is diluted. done
clear
View Solution play_arrow
-
question_answer19)
Given that: \[E{{{}^\circ }_{A{{g}^{+}}/Ag}}~=0.80V\] and \[[A{{g}^{+}}]={{10}^{-3}}M;\] \[E{{{}^\circ }_{Hg_{2}^{2+}/Hg}}=0.785V\] and \[[Hg_{2}^{2+}]={{10}^{-1}}M\] which is true for the cell reaction \[2Hg(l)+2A{{g}^{+}}(aq)\to 2Ag(s)+Hg_{2}^{2+}(aq)?\]
A)
The forward reaction is spontaneous done
clear
B)
The backward reaction is spontaneous done
clear
C)
\[{{E}_{cell}}=0.163\,\nu \] done
clear
D)
\[{{E}_{cell}}=1.585\,\nu \] done
clear
View Solution play_arrow
-
question_answer20)
Use the following standand electrode potentials, calculate \[\Delta G{}^\circ \] in kJ/ mol for the indicated reaction:
| \[5C{{e}^{4+}}(aq)+M{{n}^{2+}}(aq)+4{{H}_{2}}O(l)\xrightarrow{{}}\] \[5C{{e}^{3+}}(aq)+MnO_{4}^{-}(aq)+8{{H}^{+}}(aq)\] |
| \[MnO_{4}^{-}\left( aq \right)+8{{H}^{+}}\left( aq \right)+5{{e}^{-}}\xrightarrow{{}}\] \[M{{n}^{2+}}(aq)+4{{H}_{2}}O(l);E{}^\circ =+1.51V\] |
| \[C{{e}^{4+}}(aq)+{{e}^{-}}\xrightarrow{{}}C{{e}^{3+}}(aq);E{}^\circ =+1.61\,V\] |
A)
\[-9.65\] done
clear
B)
\[-24.3\] done
clear
C)
\[-48.25\] done
clear
D)
\[-35.2\] done
clear
View Solution play_arrow
-
question_answer21)
If 0.01 M solution of an electrolyte has a resistance of 40 ohms in a cell having a cell constant of \[0.4\text{ }c{{m}^{-1}},\] then its molar conductance in \[oh{{m}^{-1}}c{{m}^{2}}mo{{l}^{-1}}\] is
A)
\[{{10}^{2}}\] done
clear
B)
\[{{10}^{4}}\] done
clear
C)
10 done
clear
D)
\[{{10}^{3}}\] done
clear
View Solution play_arrow
-
question_answer22)
When a concentrated solution of an electrolyte is diluted
A)
its specific conductance increases. done
clear
B)
its equivalent conductivity decreases. done
clear
C)
its specific conductivity decreases and equivalent conductivity increases. done
clear
D)
both specific and equivalent conductivity increase. done
clear
View Solution play_arrow
-
question_answer23)
The limiting equivalent conductivity of \[NaCl,~KCl\] and \[KBr\] are 126.5, 150.0 and\[151.5\text{ }S\text{ }c{{m}^{2}}e{{q}^{-1}}\], respectively. The limiting equivalent ionic conductivity for \[B{{r}^{-}}\] is\[78\,Sc{{m}^{2}}e{{q}^{-1}}\]. The limiting equivalent ionic conductivity for \[N{{a}^{+}}\] ions would be:
A)
128 done
clear
B)
125 done
clear
C)
49 done
clear
D)
50 done
clear
View Solution play_arrow
-
question_answer24)
The ionic conductivity of \[B{{a}^{2+}}\] and \[C{{l}^{-}}\] at infinite dilution are 127 and 76 \[oh{{m}^{-1}}\] \[c{{m}^{2}}e{{q}^{-1}}\] respectively. The equivalent conductivity of \[BaC{{l}_{2}}\] at infinity dilution (in \[oh{{m}^{-1}}\,C{{m}^{2}}e{{q}^{-1}}\]) would be:
A)
203 done
clear
B)
279 done
clear
C)
101.5 done
clear
D)
139.5 done
clear
View Solution play_arrow
-
question_answer25)
Aluminium oxide may be electrolysed at \[1000{}^\circ C\] to furnish aluminium metal (At. Mass = 27 amu; 1 Faraday = 96,500 Coulombs). The cathode reaction is - \[A{{l}^{3+}}+3{{e}^{-}}\to Al\] To prepare 5.12 kg of aluminium metal by this method we require electricity of
A)
\[5.49\times {{10}^{1}}C\] done
clear
B)
\[5.49\times {{10}^{4}}C\] done
clear
C)
\[1.83\times {{10}^{7}}C\] done
clear
D)
\[5.49\times {{10}^{7}}C\] done
clear
View Solution play_arrow
-
question_answer26)
When electric current is passed through acidified water, 112 mL of hydrogen gas at STP collected at the cathode in 965 seconds. The current passed in amperes is
A)
1.0 done
clear
B)
0.5 done
clear
C)
0.1 done
clear
D)
2.0 done
clear
View Solution play_arrow
-
question_answer27)
On passing current through two cells, connected in series containing solution of \[AgN{{O}_{3}}\] and \[CuS{{O}_{4}}\], 0.18 g of Ag is deposited. The amount of the Cu deposited is:
A)
0.529 g done
clear
B)
10.623 g done
clear
C)
0.0529 g done
clear
D)
1.2708 g done
clear
View Solution play_arrow
-
question_answer28)
If x is the specific resistance of the solution and N is the normality of the solution, the equivalent conductivity of the solution is given by
A)
\[\frac{1000x}{N}\] done
clear
B)
\[\frac{1000x}{N}\] done
clear
C)
\[\frac{1000N}{x}\] done
clear
D)
\[\frac{Nx}{1000}\] done
clear
View Solution play_arrow
-
question_answer29)
For an electrolyte solution of \[0.05\text{ }mol\text{ }{{L}^{-1}}\] the conductivity has been found to be \[0.0110\text{ }S\text{ }c{{m}^{-1}}\] The molar conductivity is
A)
\[0.055\text{ }S\text{ }c{{m}^{2}}mo{{l}^{-1}}\] done
clear
B)
\[550\text{ }S\text{ }c{{m}^{2}}mo{{l}^{-1}}\] done
clear
C)
\[0.22\text{ }S\text{ }c{{m}^{2}}mo{{l}^{-1}}\] done
clear
D)
\[220\text{ }S\text{ }c{{m}^{2}}mo{{l}^{-1}}\] done
clear
View Solution play_arrow
-
question_answer30)
The highest electrical conductivity of the following aqueous solutions is of
A)
0.1 M difluoroacetic acid done
clear
B)
0.1 M fluoroacetic acid done
clear
C)
0.1 M chloroacetic acid done
clear
D)
0.1 M acetic acid done
clear
View Solution play_arrow
-
question_answer31)
A current of 2.0 A passed for 5 hours through a molten metal salt deposits 22.2 g of metal (At wt. = 177). The oxidation state of the metal in the metal salt is
A)
+1 done
clear
B)
+2 done
clear
C)
+3 done
clear
D)
+4 done
clear
View Solution play_arrow
-
question_answer32)
The specific conductivity of \[N/10\text{ }KCl\] solution at \[20{}^\circ C\] is \[0.212\text{ }oh{{m}^{-1}}c{{m}^{-1}}\] and the resistance of the cell containing this solution at \[20{}^\circ C\] is 55 ohm. The cell constant is
A)
\[4.616\,c{{m}^{-1}}\] done
clear
B)
\[11.66\,\,c{{m}^{-1}}\] done
clear
C)
\[2.173\,c{{m}^{-1}}\] done
clear
D)
\[3.324\,c{{m}^{-1}}\] done
clear
View Solution play_arrow
-
question_answer33)
The equivalent conductance at infinite dilution of a weak acid such as HF
A)
can be determined by extrapolation of measurements of dilute solutions of \[HCl,HBr\] and HI done
clear
B)
can be determined by measurement of very dilute HF solutions done
clear
C)
can be determined from measurements of dilute solutions of \[NaF,NaCl\] and \[HCl\] done
clear
D)
is an undefined quantity done
clear
View Solution play_arrow
-
question_answer34)
Conductance of \[0.1\text{ }M\text{ }KCl\] (conductivity = \[X\,\,Oh{{m}^{-1}}c{{m}^{-1}}\] filled in a conductivity cell is \[Y\,\,Oh{{m}^{-1}}\]. If the conductance of \[0.1\text{ }M\text{ }NaOH\] filled in the same cell is \[Z\,\,Oh{{m}^{-1}},\] the molar conductance of \[NaOH\] will be
A)
\[{{10}^{3}}\frac{XZ}{Y}\] done
clear
B)
\[{{10}^{4}}\frac{XZ}{Y}\] done
clear
C)
\[10\frac{XZ}{Y}\] done
clear
D)
\[0.1\frac{XZ}{Y}\] done
clear
View Solution play_arrow
-
question_answer35)
In electrolytic reduction of a nitroarene with 50% current efficiency 20.50 g of the compound is reduced by \[2\times 96500\text{ }C\] of electric charge. The molar mass of the compound is
A)
123.0 g done
clear
B)
61.5 g done
clear
C)
10.2 g done
clear
D)
20.5 g done
clear
View Solution play_arrow
-
question_answer36)
Electrolysis of \[NaCl\] solution with inert electrodes for certain period of time gave \[600\text{ }c{{m}^{3}}\] of 1.0 M \[NaOH\] in the electrolytic cell. During the same period 31.80 g of copper was deposited in a copper voltmeter in series with the electrolytic cell. What is the percent current efficiency in the electrolytic cell? (At.wt. of Cu =63.6)
A)
40 done
clear
B)
25 done
clear
C)
60 done
clear
D)
50 done
clear
View Solution play_arrow
-
question_answer37)
Given the ionic conductance of
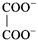
, \[{{K}^{+}}\], and \[N{{a}^{+}}\] are 74, 50, and \[73\text{ }c{{m}^{2}}oh{{m}^{-1}}\] \[e{{q}^{-1}}\] respectively. The equivalent conductance at infinite dilution of the salt
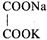
is
A)
\[197\text{ }c{{m}^{2}}oh{{m}^{-1}}e{{q}^{-1}}\] done
clear
B)
\[172\,c{{m}^{2}}oh{{m}^{-1}}e{{q}^{-1}}\] done
clear
C)
\[135.5\,c{{m}^{2}}oh{{m}^{-1}}e{{q}^{-1}}\] done
clear
D)
\[160.5\,c{{m}^{2}}oh{{m}^{-1}}e{{q}^{-1}}\] done
clear
View Solution play_arrow
-
question_answer38)
On passing a current of 1.0 ampere for 16 min and 5 sec through one litre solution of\[CuC{{l}_{2}}\], all copper of the solution was deposited at cathode. The strength of \[CuC{{l}_{2}}\] solution was (Molar mass of Cu= 63.5; Faraday constant\[=96500Cmo{{l}^{-1}}\])
A)
0.01 N done
clear
B)
0.01 M done
clear
C)
0.02 M done
clear
D)
0.2 N done
clear
View Solution play_arrow
-
question_answer39)
Electrode potential of the half cell \[Pt\left( s \right)|Hg\left( l \right)|\]\[H{{g}_{2}}C{{l}_{2}}(s)|C{{l}^{-}}(aq)\] can be increased by:
A)
Increasing \[\left[ C{{l}^{-}} \right]\] done
clear
B)
Decreasing \[\left[ C{{l}^{-}} \right]\] done
clear
C)
Increasing \[H{{g}_{2}}C{{l}_{2}}(s)\] done
clear
D)
Decreasing \[Hg\left( l \right)\] done
clear
View Solution play_arrow
-
question_answer40)
Identify the correct statement:
A)
Corrosion of iron can be minimized by forming a contact with another metal with a higher reduction potential done
clear
B)
Iron corrodes in oxygen free water done
clear
C)
Corrosion of iron can be minimized by forming an impermeable barrier at its surface done
clear
D)
Iron corrodes more rapidly in salt water because its electrochemical potential is higher done
clear
View Solution play_arrow
-
question_answer41)
During the charging of lead storage battery, the reaction at anode is represented by:
A)
\[P{{b}^{2+}}+SO_{4}^{2-}\xrightarrow{{}}PbS{{O}_{4}}\] done
clear
B)
\[PbS{{O}_{4}}+2{{H}_{2}}O\xrightarrow{{}}Pb{{O}_{2}}+SO_{4}^{2-}\]\[+4{{H}^{+}}+2{{e}^{-}}\] done
clear
C)
\[Pb\xrightarrow{{}}P{{b}^{2+}}+2{{e}^{-}}\] done
clear
D)
\[P{{b}^{2+}}+2{{e}^{-}}\xrightarrow{{}}Pb\] done
clear
View Solution play_arrow
-
question_answer42)
Which colourless gas evolves, when \[N{{H}_{4}}Cl\] reacts with zinc in a dry cell battery
A)
\[N{{H}_{4}}\] done
clear
B)
\[{{N}_{2}}\] done
clear
C)
\[{{H}_{2}}\] done
clear
D)
\[C{{l}_{2}}\] done
clear
View Solution play_arrow
-
question_answer43)
Given
| \[F{{e}^{3+}}(aq)+{{e}^{-}}\to F{{e}^{2+}}(aq);E{}^\circ =+0.77V\] |
| \[A{{l}^{3+}}(aq)+3{{e}^{-}}\to Al(s);E{}^\circ =-1.66V\] |
| \[B{{r}_{2}}(aq)+2{{e}^{-}}\to 2B{{r}^{-}};E{}^\circ =+1.09V\] |
| Considering the electrode potentials, which of the following represents the correct order of reducing power? |
A)
\[F{{e}^{2+}}<Al<B{{r}^{-}}\] done
clear
B)
\[B{{r}^{-}}<F{{e}^{2+}}<Al\] done
clear
C)
\[Al<B{{r}^{-}}<F{{e}^{2+}}\] done
clear
D)
\[Al<F{{e}^{2+}}<B{{r}^{-}}\] done
clear
View Solution play_arrow
-
question_answer44)
How many electrons would be required to deposit 6.35 g of copper at the cathode during the electrolysis of an aqueous solution of copper sulphate? (Atomic mass of copper \[=63.5u,{{N}_{A}}\] = Avogadro's constant):
A)
\[\frac{{{N}_{A}}}{20}\] done
clear
B)
\[\frac{{{N}_{A}}}{10}\] done
clear
C)
\[\frac{{{N}_{A}}}{5}\] done
clear
D)
\[\frac{{{N}_{A}}}{2}\] done
clear
View Solution play_arrow
-
question_answer45)
Given:
| \[E{{{}^\circ }_{\frac{1}{2}C{{l}_{2}}/C{{l}^{-}}}}=1.36V,\text{ }E{{{}^\circ }_{C{{r}^{3+}}/Cr}}~=-0.74V,\] |
| \[E{{{}^\circ }_{C{{r}_{2}}O_{7}^{2-}/C{{l}^{-}}}}=1.33V,\text{ }E{{{}^\circ }_{MnO_{4}^{-}/M{{n}^{2+}}}}~=1.51\,V\] |
| The correct order of reducing power of the species \[\left( Cr,C{{r}^{3+}},\text{ }M{{n}^{2+}}and\text{ }C{{l}^{-}} \right)\] will be: |
A)
\[M{{n}^{2+}}<C{{l}^{-}}<C{{r}^{3+}}<Cr\] done
clear
B)
\[M{{n}^{2+}}<C{{r}^{3+}}<C{{l}^{-}}<Cr\] done
clear
C)
\[C{{r}^{3+}}<C{{l}^{-}}<M{{n}^{2+}}<Cr\] done
clear
D)
\[C{{r}^{3+}}<C{{l}^{-}}<Cr<M{{n}^{2+}}\] done
clear
View Solution play_arrow
-
question_answer46)
The oxidation potential of a hydrogen electrode at pH = 10 and \[{{P}_{H}}_{_{2}}=1\,atm\] is
A)
\[-0.59V\] done
clear
B)
\[0.00\text{ }V\] done
clear
C)
\[+0.59V\] done
clear
D)
0.059 V done
clear
View Solution play_arrow
-
question_answer47)
If the \[E{{{}^\circ }_{cell}}\] for a given reaction has a negative value, then which of the following gives the correct relationships for the values of \[\Delta G{}^\circ \] and \[{{K}_{eq}}\] ?
A)
\[\Delta G{}^\circ >0;{{K}_{eq}}>1\] done
clear
B)
\[\Delta G{}^\circ <0;\,\,{{K}_{eq}}>1\] done
clear
C)
\[\Delta G{}^\circ <0;\,\,{{K}_{eq}}<1\] done
clear
D)
\[\Delta G{}^\circ >0;\,\,{{K}_{eq}}<1\] done
clear
View Solution play_arrow
-
question_answer48)
The EMF of the cell \[Tl/T{{l}^{+}}(0.001M)||C{{u}^{2+}}(0.01M)/Cu\,is\,0.83.\] The cell EMF can be increased by
A)
Increasing the concentration of \[T{{l}^{+}}\] ions. done
clear
B)
Increasing the concentration of\[C{{u}^{2+}}\] ions. done
clear
C)
Increasing the concentration of \[T{{l}^{+}}\] and \[C{{u}^{2+}}\]ions. done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer49)
Small quantities of solutions of compounds TX, TY and TZ are put into separate test tubes containing X, Y and Z solution. TX does not react with any of these. TY reacts with both X and Z. TZ reacts with X. The decreasing order of state of oxidation of the anions \[{{X}^{-}},{{Y}^{-}},{{Z}^{-}}\]is
A)
\[{{Y}^{-}},{{Z}^{-}},{{X}^{-}}\] done
clear
B)
\[{{Z}^{-}},{{X}^{-}},{{Y}^{-}}\] done
clear
C)
\[{{Y}^{-}},{{X}^{-}},{{Z}^{-}}\] done
clear
D)
\[{{X}^{-}},{{Z}^{-}},{{Y}^{-}}\] done
clear
View Solution play_arrow
-
question_answer50)
An unknown metal M displaces nickel from nickel (II) sulphate solution but does not displace manganese from manganese sulphate solution. Which order represents the correct order of reducing power?
A)
\[Mn>Ni>M\] done
clear
B)
\[Ni>Mn>M\] done
clear
C)
\[Mn>M>Ni\] done
clear
D)
\[M>Ni>Mn\] done
clear
View Solution play_arrow
-
question_answer51)
What is the e.m.f for the given cell?
| \[Cr|C{{r}^{3+}}(1.0M)||C{{o}^{2+}}(1.0M)|Co\] |
| \[(E{}^\circ for\text{ }C{{r}^{3+}}/Cr=-0.74\] volt and \[E{}^\circ \] for \[C{{o}^{2+}}/Co=-0.28\] volt) |
A)
\[-0.46\]volt done
clear
B)
\[-1.02\,~volt\] done
clear
C)
\[+0.46\] volt done
clear
D)
1.66 volt done
clear
View Solution play_arrow
-
question_answer52)
The standard electrode potential \[\left( E{}^\circ \right)\] for \[OC{{l}^{-}}/C{{l}^{-}}\] and \[C{{l}^{-}}/\frac{1}{2}C{{l}_{2}}\] respectively are 0.94 V and \[-1.36\text{ }V\]. The \[E{}^\circ \] value for \[OC{{l}^{-}}/\frac{1}{2}C{{l}_{2}}\] will be
A)
\[-0.42\,\,V\] done
clear
B)
\[-2.20\,\,V\] done
clear
C)
0.52 V done
clear
D)
1.04 V done
clear
View Solution play_arrow
-
question_answer53)
For the reduction of silver ions with copper metal, the standard cell potential was found to be \[+0.46\text{ }V\] at \[25{}^\circ C.\] The value of standard Gibbs energy, \[\Delta G{}^\circ \] will be \[\left( F=96500C\text{ }mo{{l}^{-1}} \right)\]
A)
\[-89.0\,kJ\] done
clear
B)
\[-89.0\,J\] done
clear
C)
\[-44.5\,kJ\] done
clear
D)
\[-98.0\,kJ\] done
clear
View Solution play_arrow
-
question_answer54)
In a cell that utilises the reaction \[Zn(s)+2{{H}^{+}}(aq)\to Z{{n}^{2+}}(aq)+{{H}_{2}}(g)\] addition of \[{{H}_{2}}S{{O}_{4}}\] to cathode compartment, will
A)
increase the E and shift equilibrium to the right done
clear
B)
lower the E and shift equilibrium to the right done
clear
C)
lower the E and shift equlibrium to the left done
clear
D)
increase the E and shift equilibrium to the left done
clear
View Solution play_arrow
-
question_answer55)
For a cell reaction involving two electron change, the standard EMF of the cell is 0.295 V at \[2{}^\circ C\]. The equilibrium constant of the reaction at \[25{}^\circ C\] will be:
A)
\[29.5\times {{10}^{-2}}\] done
clear
B)
10 done
clear
C)
\[1\times {{10}^{10}}\] done
clear
D)
\[2.95\times {{10}^{-10}}\] done
clear
View Solution play_arrow
-
question_answer56)
On the basis of the information available from the reaction \[\frac{4}{3}Al+{{O}_{2}}\to \frac{2}{3}A{{l}_{2}}{{O}_{3}},\Delta G\] \[=-827\text{ }kJ\text{ }mo{{l}^{-1}}\] of \[{{O}_{2}}\] the minimum e.m.f required to carry out an electrolysis of \[A{{l}_{2}}{{O}_{3}}\] is \[\left( F=96500C\text{ }mo{{l}^{-1}} \right)\]
A)
8.56 V done
clear
B)
2.14 V done
clear
C)
4.28 V done
clear
D)
6.42 V done
clear
View Solution play_arrow
-
question_answer57)
| The e.m.f. of a Daniell cell at 298 K is \[{{E}_{1}}.\] |
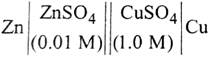 |
| When the concentration of \[ZnS{{O}_{4}}\] is 1.0 M and that of \[CuS{{O}_{4}}\] is 0.01 M, the e.m.f. changed to \[{{E}_{2}}\]. What is the relationship between \[{{E}_{1}}\] and\[{{E}_{2}}\]? |
A)
\[{{E}_{2}}=0\ne {{E}_{1}}\] done
clear
B)
\[{{E}_{1}}>{{E}_{2}}\] done
clear
C)
\[{{E}_{1}}<{{E}_{2}}\] done
clear
D)
\[{{E}_{1}}={{E}_{2}}\] done
clear
View Solution play_arrow
-
question_answer58)
Mark the false statement?
A)
A salt bridge is used to eliminate liquid junction potential done
clear
B)
The Gibbs free energy change, \[\Delta G\] is related with electromotive force E as \[\Delta G=-nFE\] done
clear
C)
Nernst equation for single electrode potential is \[E=E{}^\circ -\frac{RT}{nF}log{{a}_{{{M}^{n+}}}}\] done
clear
D)
The efficiency of a hydrogen-oxygen fuel cell is 23% done
clear
View Solution play_arrow
-
question_answer59)
The electrode potential \[{{E}_{(Z{{n}^{2+}}/Zn)}}\] of a zinc electrode at \[25{}^\circ C\] with an aqueous solution of \[0.1\text{ }M\text{ }ZnS{{O}_{4}}\] is\[[E{{{}^\circ }_{(Z{{n}^{2+}}/Zn)}}~=-0.76\text{ }V\]. Assume \[\frac{2.303RT}{F}=0.06\text{ }at\text{ }298\text{ }K].\]
A)
+0.73 done
clear
B)
\[-0.79\] done
clear
C)
\[-0.82\] done
clear
D)
\[-0.70\] done
clear
View Solution play_arrow
-
question_answer60)
The reduction potential (in volt) of a hydrogen electrode set up with a \[2\times {{10}^{-2}}M\] aqueous solution of a weak mono basic acid \[({{K}_{a}}=5\times {{10}^{-5}})\] at one atmosphere and \[25{}^\circ C\] is
A)
+0.09 done
clear
B)
+0.18 done
clear
C)
\[-0.09\] done
clear
D)
\[-0.18\] done
clear
View Solution play_arrow
-
question_answer61)
A solution of copper sulphate \[\left( CuS{{O}_{4}} \right)\] is electrolysed for 10 minutes with a current of 1.5 amperes. The mass of copper deposited at the cathode (at. mass of\[Cu=63u\]) is:
A)
0.3892 g done
clear
B)
0.2938 g done
clear
C)
0.2398 g done
clear
D)
0.3928 g done
clear
View Solution play_arrow
-
question_answer62)
A battery is constructed of Cr and \[N{{a}_{2}}C{{r}_{2}}{{O}_{7}}\]. The unbalanced chemical equation when such a battery discharges is following \[N{{a}_{2}}C{{r}_{2}}{{O}_{7}}+Cr+{{H}^{+}}\to C{{r}^{3+}}+{{H}_{2}}O+N{{a}^{+}}\] If one Faraday of electricity is passed through the battery during the charging, the number of moles of \[C{{r}^{\text{3+}}}\] removed from the solution is
A)
\[\frac{4}{3}\] done
clear
B)
\[\frac{1}{3}\] done
clear
C)
\[\frac{3}{3}\] done
clear
D)
\[\frac{2}{3}\] done
clear
View Solution play_arrow
-
question_answer63)
The standard potentials of \[A{{g}^{+}}/Ag,H{{g}_{2}}^{2+}/2Hg,C{{u}^{2+}}/Cu\] and \[M{{g}^{2+}}/Mg\] electrodes are 0.80,0.79, 0.34 and\[-2.37\text{ }V\], respectively. An aqueous solution which contains one mole per litre of the salts of each of the four metals is electrolyzed. With increasing voltage, the correct sequence of deposition of the metals at the cathode is
A)
\[Ag,Hg,Cu,Mg\] done
clear
B)
\[Cu,Hg,Ag\] only done
clear
C)
\[Ag,Hg,Cu\] only done
clear
D)
\[Mg,Cu,Hg,Ag\] done
clear
View Solution play_arrow
-
question_answer64)
When electric current is passed through a cell having an electrolytic solution, the cations move towards the cathode and anions towards the anode. If anode is pulled out from the solution
A)
the cations and anions will move towards the cathode done
clear
B)
the cations will continue to move towards cathode and anions will stop moving done
clear
C)
both the cations and anions will stop moving done
clear
D)
the cations and anions will start moving randomly. done
clear
View Solution play_arrow
-
question_answer65)
At 298K the standard free energy of formation of \[{{H}_{2}}O(l)\] is \[-237.20\text{ }kJ/mol\] while that of its ionisation into \[{{H}^{+}}\] ion and hydroxyl ions is 80 kJ/ mol, then the emf of the following cell at 298 K will be [Take Faraday constant F = 96500 C] \[{{H}_{2}}(g,1bar)|{{H}^{+}}(1M)||O{{H}^{-}}(1M)|{{O}_{2}}(g,1\,bar)\]
A)
0.40 V done
clear
B)
0.81 V done
clear
C)
1.23 V done
clear
D)
\[-0.40\text{ }V\] done
clear
View Solution play_arrow
-
question_answer66)
A concentration cell is a galvanic cell in which
A)
decrease in free energy in a spontaneous chemical process appears as electrical energy done
clear
B)
decrease in free energy in a spontaneous physical process appears as electrical energy done
clear
C)
decrease in free energy in a spontaneous physical or chemical process appears as electrical energy done
clear
D)
a non-spontaneous physical or chemical process produces electrical energy. done
clear
View Solution play_arrow
-
question_answer67)
In the silver plating of copper, \[K[Ag{{(CN)}_{2}}]\] is used instead of \[AgN{{O}_{3}}\]. The reason is
A)
a thin layer of Ag is formed on Cu done
clear
B)
more voltage is required done
clear
C)
\[A{{g}^{+}}\] ions are completely removed from solution done
clear
D)
less availability of \[A{{g}^{+}}\] ions, as Cu cannot displace Ag from \[{{[Ag{{\left( CN \right)}_{2}}]}^{-}}\] ion done
clear
View Solution play_arrow
-
question_answer68)
1.0 L each of a buffer containing 1 mole \[N{{H}_{3}}\] and 1 mol of \[NH_{4}^{+}\] were placed in the cathodic and anodic half-cells and 965 C of electricity was passed. If anodic and cathodic half cells reactions involve oxidation and reduction of water only as
| \[2{{H}_{2}}O\xrightarrow{{}}4{{H}^{+}}+{{O}_{2}}-4{{e}^{-}};\] |
| \[2{{H}_{2}}O+2{{e}^{-}}\xrightarrow{{}}{{H}_{2}}+2O{{H}^{-}}\] |
| Then pH of |
A)
cathodic solution will increase done
clear
B)
anodic solution will decrease done
clear
C)
both the solutions will remain practically constant done
clear
D)
both the solutions will increase done
clear
View Solution play_arrow
-
question_answer69)
The emf of a particular voltaic cell with the cell reaction \[Hg_{2}^{2+}+{{H}_{2}}\rightleftharpoons 2Hg+2{{H}^{+}}\] is 0.65 V. The maximum electrical work of this cell when 0.5 g of \[{{H}_{2}}\] is consumed.
A)
\[-3.12\times {{10}^{4}}J\] done
clear
B)
\[-1.25\times {{10}^{5}}J\] done
clear
C)
\[25.0\times {{10}^{6}}J\] done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer70)
A cell reaction would be spontaneous if the cell potential and \[{{\Delta }_{r}}G\] are respectively:
A)
positive and negative done
clear
B)
negative, negative done
clear
C)
zero, zero done
clear
D)
positive, zero done
clear
View Solution play_arrow
-
question_answer71)
The Nernst equation \[E=E{}^\circ -RT/nF\] In Q indicates that the Q will be equal to equilibrium constant \[{{K}_{c}}\] when:
A)
\[E=E{}^\circ \] done
clear
B)
\[RT/nF=1\] done
clear
C)
\[E=zero\] done
clear
D)
\[E{}^\circ =1\] done
clear
View Solution play_arrow
-
question_answer72)
Thermodynamic efficiency of a cell is given by:
A)
\[\frac{\Delta H}{\Delta G}\] done
clear
B)
\[\frac{nFE}{\Delta G}\] done
clear
C)
\[\frac{-nFE}{\Delta H}\] done
clear
D)
\[nFE{}^\circ \] done
clear
View Solution play_arrow
-
question_answer73)
A graph was plotted between molar conductivity of various electrolytes \[(NaCl,HCl\,and\,N{{H}_{4}}OH)\] and \[\sqrt{c}(in\,mol\,{{L}^{-1}}).\] Correct set is:
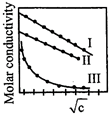
A)
\[I(NaCl),II(HCL),III,(N{{H}_{4}}OH)\] done
clear
B)
\[I\left( HCl \right),II\left( NaCl \right),III,\left( N{{H}_{4}}OH \right)\] done
clear
C)
\[I\left( N{{H}_{4}}OH \right),II\left( NaCl \right),III,\left( HCl \right)\] done
clear
D)
\[I(N{{H}_{4}}OH),II(HCl),III,(NaCl)\] done
clear
View Solution play_arrow
-
question_answer74)
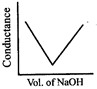
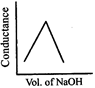
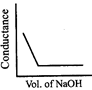
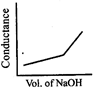
\[HN{{O}_{3}}\] (aq) is titrated with \[NaOH\left( aq \right)\] conductometrically, graphical representation of the titration is:
A)
B)
C)
D)
View Solution play_arrow
-
question_answer75)
In the electrolysis of water, one faraday of electrical energy would liberate
A)
one mole of oxygen done
clear
B)
one gram atom of oxygen done
clear
C)
8 g oxygen done
clear
D)
22.4 lit. of oxygen done
clear
View Solution play_arrow
-
question_answer76)
Resistance of a conductivity cell filled with a solution of an electrolyte of concentration 0.1 M is \[100\Omega .\]. The conductivity of this solution is \[1.29\text{ }S\text{ }{{m}^{-1}}\]. Resistance of the same cell when filled with 0.02 M of the same solution is \[520\Omega \]. The molar conductivity of 0.02 M solution of electrolyte will be
A)
\[1.24\times {{10}^{-4}}\,S{{m}^{2}}mo{{l}^{-1}}\] done
clear
B)
\[12.4\times {{10}^{-4}}\,S{{m}^{2}}mo{{l}^{-1}}\] done
clear
C)
\[124\times {{10}^{-4}}\,S{{m}^{2}}mo{{l}^{-1}}\] done
clear
D)
\[1240\times {{10}^{-4}}\,S{{m}^{2}}mo{{l}^{-1}}\] done
clear
View Solution play_arrow
-
question_answer77)
| \[{{\Lambda }_{ClC{{H}_{2}}COONa}}=224\,oh{{m}^{-1}}c{{m}^{2}}g\,e{{q}^{-1}},\] |
| \[{{\Lambda }_{NaCl}}=38.2\,oh{{m}^{-1}}c{{m}^{2}}g\,e{{q}^{-1}},\] |
| \[{{\Lambda }_{HCl}}=203\,oh{{m}^{-1}}c{{m}^{2}}ge{{q}^{-1}},\] |
| What is the value of \[{{\Lambda }_{CIC{{H}_{2}}COOH}}\] |
A)
\[288.5\,oh{{m}^{-1}}c{{m}^{2}}\,g\,e{{q}^{-1}}\] done
clear
B)
\[289.5\,oh{{m}^{-1}}c{{m}^{2}}ge{{q}^{-1}}\] done
clear
C)
\[388.8\,oh{{m}^{-1}}c{{m}^{2}}g\,e{{q}^{-1}}\] done
clear
D)
\[59.5\,oh{{m}^{-1}}c{{m}^{2}}g\,e{{q}^{-1}}\] done
clear
View Solution play_arrow
-
question_answer78)
1.08 g of pure silver was converted into silver nitrate and its solution was taken in a beaker. It was electrolysed using platinum cathode and silver anode. 0.01 Faraday of electricity was passed using 0.15 volt above the decomposition potential of silver. The silver content of the beaker after the above shall be
A)
0 g done
clear
B)
0.108 g done
clear
C)
1.08 g done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer79)
Specific conductance of 0.1 M sodium chloride solution is\[1.06\times {{10}^{-2}}oh{{m}^{-1}}c{{m}^{-1}}\]. Its molar conductance in \[oh{{m}^{-1}}c{{m}^{2}}mo{{l}^{-1}}\] is
A)
\[1.06\times {{10}^{2}}\] done
clear
B)
\[1.06\times {{10}^{3}}\] done
clear
C)
\[1.06\times {{10}^{4}}\] done
clear
D)
\[5.3\times {{10}^{2}}\] done
clear
View Solution play_arrow
-
question_answer80)
In the electrolysis of \[CuC{{l}_{2}}\] solution, the mass of the cathode increased by 3.2 g. What occured at the copper anode?
A)
0.12 litre of \[C{{l}_{2}}\] was liberated done
clear
B)
0.56 litre of \[{{O}_{2}}\] was liberated done
clear
C)
0.1 mol \[C{{u}^{2+}}\] passed into the solution. done
clear
D)
0.05 mol of \[C{{u}^{2+}}\] passed into the solution. done
clear
View Solution play_arrow
-
question_answer81)
Given \[E{{{}^\circ }_{C{{u}^{2+}}/Cu}}=0.34V,\,E{{{}^\circ }_{C{{u}^{2+}}/Cu}}=0.15V\] Standard electrode potential for the half cell \[C{{u}^{+}}/Cu\] is
A)
0.38 V done
clear
B)
0.53 V done
clear
C)
0.19 V done
clear
D)
0.49 V done
clear
View Solution play_arrow
-
question_answer82)
At 298 K, the standard reduction potentials are 1.51 V for \[MnO_{4}^{-}|M{{n}^{2+}},1.36V\,\text{for }C{{l}_{2}}|C{{l}^{-}},\,\,1.07\] \[V\,for\,B{{r}_{2}}/B{{r}^{-}}\], and 0.54 V for \[{{I}_{2}}/{{I}^{-}}.\text{ }At\text{ }pH=3,\] permanganate is expected to oxidize: \[\left( \frac{RT}{F}=0.059V \right)\]
A)
\[C{{l}^{-}},B{{r}^{-}}\] and \[{{I}^{-}}\] done
clear
B)
\[B{{r}^{-}}\] and \[{{I}^{-}}\] done
clear
C)
\[C{{l}^{-}}\] and \[B{{r}^{-}}\] done
clear
D)
\[{{I}^{-}}\] only done
clear
View Solution play_arrow
-
question_answer83)
Electrode potentials \[\left( E{}^\circ \right)\] are given below:
| \[C{{u}^{+}}/Cu=+0.52V,\] |
| \[F{{e}^{3+}}/F{{e}^{2+}}=+0.77V,\] |
| \[\frac{1}{2}{{I}_{2}}(s)/{{I}^{-}}=+0.54V,\] |
| \[A{{g}^{+}}/Ag=+0.88V\]. |
| Based on the above potentials, strongest oxidizing agent will be: |
A)
\[C{{u}^{+}}\] done
clear
B)
\[F{{e}^{3+}}\] done
clear
C)
\[A{{g}^{+}}\] done
clear
D)
\[{{I}_{2}}\] done
clear
View Solution play_arrow
-
question_answer84)
Which of the following is a highly corrosive salt?
A)
\[H{{g}_{2}}C{{l}_{2}}\] done
clear
B)
\[HgC{{l}_{2}}\] done
clear
C)
\[FeC{{l}_{2}}\] done
clear
D)
\[PbC{{l}_{2}}\] done
clear
View Solution play_arrow
-
question_answer85)
The Zn acts as sacrificial or cathodic protection to prevent rusting of iron because:
A)
\[{{E}^{{}^\circ }}_{OP}\,\text{of }Zn<{{E}^{{}^\circ }}_{OP}\text{of}\,Fe\] done
clear
B)
\[{{E}^{{}^\circ }}_{OP}\,\text{of}\,Zn>{{E}^{{}^\circ }}_{OP}\,\text{of}\,Fe\] done
clear
C)
\[{{E}^{{}^\circ }}_{OP}\text{of}\,Zn={{E}^{{}^\circ }}_{OP}\,\text{of}\,Fe\] done
clear
D)
Zn is cheaper than iron done
clear
View Solution play_arrow
-
question_answer86)
Among the following cells:
| (I) Leclanche cell |
| (II) Nickel-Cadmium cell |
| (III) Lead storage battery |
| (IV) Mercury cell primary cells are |
A)
I and II done
clear
B)
I and III done
clear
C)
II and III done
clear
D)
I and IV done
clear
View Solution play_arrow
-
question_answer87)
How long a current of 3 amperes has to be passed through a solution of \[AgN{{O}_{3}}\] to coat a metal surface of \[80c{{m}^{2}}\] and 0.005 mm thick layer. Density of Ag is \[10.5\text{ }g\,c{{m}^{-3}}\]
A)
125.1 seconds done
clear
B)
12.5 seconds done
clear
C)
155.2 seconds done
clear
D)
200 seconds done
clear
View Solution play_arrow
-
question_answer88)
\[A{{l}_{2}}{{O}_{3}}\] is reduced by electrolysis at low potentials and high currents. If \[4.0\times {{10}^{4}}\] amperes of current is passed through molten \[A{{l}_{2}}{{O}_{3}}\] for 6 hours, what mass of aluminium is produced? (Assume 100% current efficiency. At. mass of\[Al=27g\text{ }mo{{l}^{-1}}\])
A)
\[8.1\times {{10}^{4}}g\] done
clear
B)
\[2.4\times {{10}^{5}}g\] done
clear
C)
\[1.3\times {{10}^{4}}g\] done
clear
D)
\[9.0\times {{10}^{3}}g\] done
clear
View Solution play_arrow
-
question_answer89)
The resistance of 1 N solution of acetic acid is 250 ohm, when measured in a cell of cell constant\[1.15\text{ }c{{m}^{-1}}\]. The equivalent conductance (in \[oh{{m}^{-1}}c{{m}^{2}}equi{{v}^{-1}}\]) of 1 N acetic acid will be
A)
4.6 done
clear
B)
9.2 done
clear
C)
18.4 done
clear
D)
0.023 done
clear
View Solution play_arrow
-
question_answer90)
Specific conductance of 0.1 MHA is \[3.75\times {{10}^{-4}}oh{{m}^{-1}}c{{m}^{-1}}\]. If \[{{\lambda }^{\infty }}(HA)=250\,oh{{m}^{-1}}c{{m}^{2}}mo{{l}^{-1}}\], the dissociation constant \[{{K}_{a}}\] of HA is:
A)
\[1.0\times {{10}^{-5}}\] done
clear
B)
\[2.25\times {{10}^{-4}}\] done
clear
C)
\[2.25\times {{10}^{-5}}\] done
clear
D)
\[2.25\times {{10}^{-13}}\] done
clear
View Solution play_arrow
-
question_answer91)
Equivalent conductance at infinite dilution, \[{{\lambda }^{{}^\circ }}\] of \[N{{H}_{4}}Cl,\,NaOH\] and \[NaCl\] are 128.0,217.8 and \[109.3\text{ }oh{{m}^{-1}}c{{m}^{2}}\text{ }e{{q}^{-1}}\] respectively. The equivalent conductance of \[0.01\text{ }N\text{ }N{{H}_{2}}OH\] is \[9.30\text{ }oh{{m}^{-1}}\,c{{m}^{2}}\text{ }e{{q}^{-1}}\] then the degree of ionization of \[N{{H}_{4}}OH\] at this temperature would be
A)
0.04 done
clear
B)
0.1 done
clear
C)
0.39 done
clear
D)
0.62 done
clear
View Solution play_arrow
-
question_answer92)
\[{{I}_{2}}(s)|{{I}^{-}}(0.1M)\] half-cell is connected to a \[{{H}^{+}}\left( aq \right)\left| {{H}_{2}}\left( \text{1}bar \right) \right|Pt\] half cell and e.m.f. is found to be\[0.7714V.if\,{{E}^{{}^\circ }}_{{{I}_{2}}|{{I}^{-}}}=0.535\text{ }V\], find the pH of \[{{H}^{+}}|{{H}_{2}}\] half-cell.
A)
1 done
clear
B)
3 done
clear
C)
5 done
clear
D)
7 done
clear
View Solution play_arrow
-
question_answer93)
Consider the following standard electrode potentials and calculate the equilibrium constant at 25° C for the indicated disproportion nation reaction:
| \[3M{{n}^{2+}}(aq)\xrightarrow{{}}Mn(s)+2M{{n}^{3+}}(aq)\] |
| \[M{{n}^{3+}}\left( aq \right)+{{e}^{-}}\xrightarrow{{}}M{{n}^{2+}}\left( aq \right);{{E}^{{}^\circ }}=1.51\text{ }V\] |
| \[M{{n}^{2+}}(aq)+2{{e}^{-}}\xrightarrow{{}}Mn(s);E{}^\circ =-1.185V\] |
A)
\[1.2\times {{10}^{-43}}\] done
clear
B)
\[2.4\times {{10}^{-73}}\] done
clear
C)
\[6.3\times {{10}^{-92}}\] done
clear
D)
\[1.5\times {{10}^{-62}}\] done
clear
View Solution play_arrow
-
question_answer94)
In a hydrogen-oxygen fuel cell, combustion of hydrogen occurs to
A)
produce high purity water done
clear
B)
create potential difference between two electrodes done
clear
C)
generate heat done
clear
D)
remove adsorbed oxygen from electrode surfaces done
clear
View Solution play_arrow
-
question_answer95)
The standard reduction potentials at \[25{}^\circ C\] of \[L{{i}^{+}}/Li,B{{a}^{2+}}/Ba,N{{a}^{+}}/Na\] and \[M{{g}^{2+}}/Mg\] are \[-3.03,-2.73,-2.71\] and \[-2.37\] volt respectively Which one of the following is the strongest oxidising agent?
A)
\[N{{a}^{+}}\] done
clear
B)
\[L{{i}^{+}}\] done
clear
C)
\[B{{a}^{2+}}\] done
clear
D)
\[M{{g}^{2+}}\] done
clear
View Solution play_arrow
-
question_answer96)
The \[E{{{}^\circ }_{{{M}^{3+}}/{{M}^{2+}}}}\] values for \[Cr,Mn,Fe\] and Co are \[-0.41,+1.57,+0.77\] and + 1.97V respectively For which one of these metals the change in oxidation state from +2 to +3 is easiest?
A)
Fe done
clear
B)
Mn done
clear
C)
Cr done
clear
D)
Co done
clear
View Solution play_arrow
-
question_answer97)
The solution of \[CuS{{O}_{4}}\] in which copper rod is immersed is diluted to 10 times. The reduction electrode potential
A)
Increases by \[30m\text{ }V\] done
clear
B)
Decreases by \[30m\text{ }V\] done
clear
C)
Increases by \[59m\text{ }V\] done
clear
D)
Decreases by \[59m\text{ }V\] done
clear
View Solution play_arrow
-
question_answer98)
For a given reaction: \[M(x+n)+n{{e}^{-}}\xrightarrow{{}}{{M}^{x+}},{{E}^{-}}_{red}\] is known along with \[{{M}^{(x+n)}}\] and \[{{M}^{x+}}\] ion concentrations. Then
A)
n can be evaluated done
clear
B)
x can be evaluated done
clear
C)
\[\left( x+n \right)\] can be evaluated done
clear
D)
\[n,x,\left( x+n \right)\] can be evaluated done
clear
View Solution play_arrow
-
question_answer99)
The emf of the cell \[Pt\,C{{l}_{2}}(g)({{P}_{1}}atm)/C{{l}^{-}}(aq)\]\[(1M)/C{{l}_{2}}(g)({{P}_{2}}atm)Pt\] will be positive when
A)
\[{{P}_{1}}={{P}_{2}}\] done
clear
B)
\[{{P}_{1}}<{{P}_{2}}\] done
clear
C)
\[{{P}_{1}}>{{P}_{2}}\] done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer100)
Equivalent conductivity can be expressed in terms of specific conductance \[\kappa \] and concentration (N) in gram equivalent per \[d{{m}^{-3}}\] as:
A)
\[\kappa\times n\] done
clear
B)
\[\frac{\kappa\times 1000}{N}\] done
clear
C)
\[\frac{\kappa\times N}{1000}\] done
clear
D)
\[\kappa\times N\times 1000\] done
clear
View Solution play_arrow
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
