-
question_answer1)
For the reaction
| \[{{N}_{2}}{{O}_{5}}(g)\xrightarrow{{}}2N{{O}_{2}}(g)+1/2\,{{O}_{2}}(g)\] the value of rate of disappearance of \[{{N}_{2}}{{O}_{5}}\] is given as \[6.25\times {{10}^{-3}}mol\,{{L}^{-1}}{{s}^{-1}}\]. The rate of formation of \[N{{O}_{2}}\] and \[{{O}_{2}}\] is given respectively as: |
A)
\[6.25\times {{10}^{-3}}mol\text{ }{{L}^{-1}}{{s}^{-1}}\] and \[6.25\times {{10}^{-3}}mol\text{ }{{L}^{-1}}{{s}^{-1}}\] done
clear
B)
\[1.25\times {{10}^{-2}}mol\text{ }{{L}^{-1}}{{s}^{-1}}\] and \[3.125\times {{10}^{-3}}mol\text{ }{{L}^{-1}}{{s}^{-1}}\] done
clear
C)
\[6.25\times {{10}^{-3}}mol\text{ }{{L}^{-1}}{{s}^{-1}}\] and \[3.125\times {{10}^{-3}}mol\text{ }{{L}^{-1}}{{s}^{-1}}\] done
clear
D)
\[1.125\times {{10}^{-2}}mol\text{ }{{L}^{-1}}{{s}^{-1}}\]and \[6.25\times {{10}^{-3}}mol\text{ }{{L}^{-1}}{{s}^{-1}}\] done
clear
View Solution play_arrow
-
question_answer2)
The reactions rate \[{{N}_{2}}\left( g \right)+3{{H}_{2}}\left( g \right)\to 2N{{H}_{3}}\left( g \right)\]was measured\[\frac{d[N{{H}_{3}}]}{dt}=2\times {{10}^{-4}}mol\,{{\sec }^{-1}}\]. The rates of reactions expressed in terms of \[{{N}_{2}}\]and \[{{H}_{2}}\] are
A)
B)
C)
D)
View Solution play_arrow
-
question_answer3)
For the reaction\[A+B\xrightarrow{{}}C+D\]. The variation of the concentration of the products is given by the curve
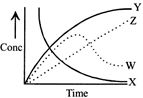
A)
Z done
clear
B)
Y done
clear
C)
W done
clear
D)
X done
clear
View Solution play_arrow
-
question_answer4)
For the reaction \[{{H}_{2}}\left( g \right)+B{{r}_{2}}\left( g \right)\to 2HBr\left( g \right),\] the experimental data suggest, rate\[=k[{{H}_{2}}]{{[B{{r}_{2}}]}^{1/2}}\]. The molecularity and order of the reaction are respectively
A)
\[2,\frac{3}{2}\] done
clear
B)
\[\frac{3}{2},\frac{3}{2}\] done
clear
C)
1,1 done
clear
D)
\[1,\frac{1}{2}\] done
clear
View Solution play_arrow
-
question_answer5)
For a chemical reaction \[{{t}_{1/2}}\] is 2.5 hours at room temperature. How much of the reactant will be left after 7.5 hours if initial weight of reactant was 160g?
A)
10 g done
clear
B)
40 g done
clear
C)
80 g done
clear
D)
20 g done
clear
View Solution play_arrow
-
question_answer6)
The rate law for a reaction between the substances A and B is given by rate \[=k{{[A]}^{n}}{{[B]}^{m}}\] On doubling the concentration of A and halving the concentration of B, the ratio of the new rate to the earlier rate of the reaction will be as
A)
\[\left( m+n \right)\] done
clear
B)
\[\left( n-m \right)\] done
clear
C)
\[{{2}^{\left( n+m \right)}}\] done
clear
D)
\[\frac{1}{{{2}^{\left( m+n \right)}}}\] done
clear
View Solution play_arrow
-
question_answer7)
The order of a reaction, with respect to one of the reacting component Y, is zero. It implies that:
A)
the reaction is going on at a constant rate done
clear
B)
the rate of reaction does not vary with temperature done
clear
C)
the reaction rate is independent of the concentration of Y done
clear
D)
the rate of formation of the activated complex is zero done
clear
View Solution play_arrow
-
question_answer8)
For a first order reaction, a plot of log \[\left( a-x \right)\] against time is a straight line with a negative slope equal to
A)
\[\frac{-k}{2.303}\] done
clear
B)
\[-2.303k\] done
clear
C)
\[\frac{2.303}{k}\] done
clear
D)
\[-\frac{{{E}_{a}}}{2.303R}\] done
clear
View Solution play_arrow
-
question_answer9)
A following mechanism has been proposed for a reaction
| \[2A+B\to D+E\] |
| \[A+B\to C+D\left( slow \right)\] |
| \[A+C\to E\left( fast \right)\] |
| The rate law expression for the reaction is: |
A)
\[r=k{{\left[ A \right]}^{2}}\left[ B \right]\] done
clear
B)
\[r=k\left[ A \right]\left[ B \right]\] done
clear
C)
\[r=k{{\left[ A \right]}^{2}}\] done
clear
D)
\[r=k\left[ A \right]\left[ C \right]\] done
clear
View Solution play_arrow
-
question_answer10)
The rate law for the reaction below is given by the expression k [A] [B] \[A+B\to Product\] If the concentration of B is increased from 0.1 to 0.3 mole, keeping the value of A at 0.1 mole, the rate constant will be:
A)
3k done
clear
B)
9k done
clear
C)
k/3 done
clear
D)
k done
clear
View Solution play_arrow
-
question_answer11)
The half-life of a radioactive element is 20 min. The time interval between the stages of its 33% and 67% decay is
A)
40 min done
clear
B)
20 min done
clear
C)
30min done
clear
D)
25 min done
clear
View Solution play_arrow
-
question_answer12)
Which of the following reactions is not of the first order?
A)
Inversion of sucrose in the presence of acid done
clear
B)
Acid-catalyzed hydrolysis of ethyl acetate done
clear
C)
Hydrolysis of tertiary butyl halide using alkali done
clear
D)
Oxidation of \[{{I}^{-}}\] ion by \[{{S}_{2}}O_{8}^{2-}\] ion done
clear
View Solution play_arrow
-
question_answer13)
Rate constant \[k=1.2\times {{10}^{3}}mo{{l}^{-1}}\,L\,{{s}^{-1}}\]and \[{{E}_{a}}=2.0\times {{10}^{2}}\,kJ\,mo{{l}^{-1}}\] When \[T\to \infty :\]
A)
\[A=2.0\times {{10}^{2}}kJ\,mo{{l}^{-1}}\] done
clear
B)
\[A=1.2\times {{10}^{3}}\,mo{{l}^{-1}}L\,{{S}^{-1}}\] done
clear
C)
\[A=1.2\times {{10}^{3}}mol\,{{L}^{-1}}{{s}^{-1}}\] done
clear
D)
\[A=2.4\times {{10}^{3}}kJ\,mo{{l}^{-1}}{{s}^{-1}}\] done
clear
View Solution play_arrow
-
question_answer14)
A catalyst lowers the activation enegy of a reaction from \[20kJ\text{ }mo{{l}^{-1}}\] to\[10kJ\text{ }mo{{l}^{-1}}\]. The temperature at which the uncatalyzed reaction will have the same rate as that of the catalyzed at \[27{}^\circ C\] is
A)
\[-123{}^\circ C\] done
clear
B)
\[327{}^\circ C\] done
clear
C)
\[32.7{}^\circ C\] done
clear
D)
\[+23{}^\circ C\] done
clear
View Solution play_arrow
-
question_answer15)
For a reaction, the rate constant is expressed as\[k=A{{e}^{-40000/T}}\]. The energy of the activation is
A)
40000 cal done
clear
B)
88000 cal done
clear
C)
80000 cal done
clear
D)
8000 cal done
clear
View Solution play_arrow
-
question_answer16)
The activation energy of the reaction, \[A+B\to C+D+38\] kcal is 20 kcal. What would be the activation energy of the following reaction \[C+D\to A+B\]
A)
20 kcal done
clear
B)
-20 kcal done
clear
C)
18 kcal done
clear
D)
58 kcal done
clear
View Solution play_arrow
-
question_answer17)
The half-life of a reaction is inversely proportional to the square of the initial concentration of the reactant. Then the order of the reaction is
A)
0 done
clear
B)
1 done
clear
C)
2 done
clear
D)
3 done
clear
View Solution play_arrow
-
question_answer18)
For a reaction\[A+2B\to C\], the amount of C formed by starting the reaction with 5 moles of A and 8 moles of B is
A)
5 moles done
clear
B)
8 moles done
clear
C)
16 moles done
clear
D)
4 moles done
clear
View Solution play_arrow
-
question_answer19)
The rate law for the reaction \[2X+Y\to Z\] is Rate \[=k\left[ X \right]\left[ Y \right].\] The correct statement with regard to this relation is
A)
the unit of k is \[{{s}^{-1}}\] done
clear
B)
the rate of the reaction is independent of [X] and [Y] done
clear
C)
for this reaction \[{{t}_{1/2}}\] is independent of initial concentrations of reactant done
clear
D)
the rate of formation of Z is half of the rate of disappearance of X done
clear
View Solution play_arrow
-
question_answer20)
In a reaction, \[2A\to \] products, the concentration of A decreases from 0.50 M to 0.38 M in 10 min. What is the rate of reaction (in\[M\text{ }{{s}^{-1}}\]) during this interval?
A)
0.012 done
clear
B)
0.024 done
clear
C)
\[2\times {{10}^{-3}}\] done
clear
D)
\[2\times {{10}^{-4}}\] done
clear
View Solution play_arrow
-
question_answer21)
A chemical reaction is catalyzed by a catalyst X. Hence X
A)
reduces enthalpy of the reaction done
clear
B)
decreases rate constant of the reaction done
clear
C)
increases activation energy of the reaction done
clear
D)
does not affect equilibrium constant of the reaction done
clear
View Solution play_arrow
-
question_answer22)
In respect of the equation \[k=A{{e}^{-Ea/RT}}\] in chemical kinetics, which one of the following statements is correct?
A)
A is adsorption factor done
clear
B)
\[{{E}_{a}}\]is energy of activation done
clear
C)
R is Rydberg's constant done
clear
D)
k is equilibrium constant done
clear
View Solution play_arrow
-
question_answer23)
The minimum energy a molecule should possess in order to enter into a fruitful collision is known as
A)
reaction energy done
clear
B)
collision energy done
clear
C)
activation energy done
clear
D)
threshold energy done
clear
View Solution play_arrow
-
question_answer24)
For a reaction, activation energy \[\left( {{E}_{a}} \right)=0\] and rate constant \[(k)=3.2\times {{10}^{6}}{{s}^{-1}}\] at 300 K. What is the value of the rate constant at 310 K
A)
\[3.2\times {{10}^{-12}}{{s}^{-1}}\] done
clear
B)
\[3.2\times {{10}^{6}}{{s}^{-1}}\] done
clear
C)
\[6.4\times {{10}^{12}}{{s}^{-1}}\] done
clear
D)
\[6.4\times {{10}^{6}}{{s}^{-1}}\] done
clear
View Solution play_arrow
-
question_answer25)
In the Arrhenius plot of ln k Vs\[\frac{1}{T}\], a linear plot is obtained with a slope of \[-2\times {{10}^{4}}K.\] The energy of activation of the reaction (in\[kJ\text{ }mol{{e}^{-1}}\]) is (R value is\[8.3J\text{ }{{K}^{-1}}mo{{l}^{-1}}\])
A)
83 done
clear
B)
166 done
clear
C)
249 done
clear
D)
332 done
clear
View Solution play_arrow
-
question_answer26)
The rate of reaction is doubled for every \[10{}^\circ C\] rise in temperature. The increase in reaction rate as a result of temperature rise from \[10{}^\circ C\] to \[100{}^\circ C\] is
A)
112 done
clear
B)
512 done
clear
C)
400 done
clear
D)
614 done
clear
View Solution play_arrow
-
question_answer27)
Consider the reaction \[A\to 2B+C,\Delta H=-15\]kcal. The energy of activation of backward reaction is\[20\text{ }kcal\text{ }mo{{l}^{-1}}\]. In presence of catalyst the energy of activation of forward reaction is \[3\text{ }kcal\text{ }mo{{l}^{-1}}\]. At 400 K. the catalyst causes the rate of the reaction to increase by the number of times equal to
A)
\[{{e}^{3.5}}\] done
clear
B)
\[{{e}^{2.5}}\] done
clear
C)
\[{{e}^{-\,2.5}}\] done
clear
D)
\[{{e}^{2.303}}\] done
clear
View Solution play_arrow
-
question_answer28)
A reaction takes place in various steps. The rate constant for first, second, third and fifth steps are \[{{k}_{1}},{{k}_{2}},{{k}_{3}}\] and \[{{k}_{4}}\] respectively The overall rate constant is given by \[k=\frac{{{k}_{2}}}{{{k}_{3}}}{{\left( \frac{{{k}_{1}}}{{{k}_{5}}} \right)}^{1/2}}\]. If activation energy are 40,60,50 and 10 kJ/mol respectively, the overall energy of activation (kJ/ mol) is:
A)
10 done
clear
B)
20 done
clear
C)
25 done
clear
D)
none of these done
clear
View Solution play_arrow
-
question_answer29)
Consider the two hypothetical reactions given below:
| I \[a\text{ }A\to \]Products, \[k=x\,mo{{l}^{-1}}\text{ }L\text{ }mi{{n}^{-1}}\] |
| II \[bB\to \]Products, \[k=y\,mi{{n}^{-1}}\] |
| The half-lives of both the reactions are the same, equal to 1 hr when molar concentration of the reactant is 1.0 M in each case. If these reactions are started at the same time taking 1M of the reactant in each case, the ratio [A]/[B] after 3 hr will be: |
A)
0.5 done
clear
B)
4 done
clear
C)
1 done
clear
D)
2 done
clear
View Solution play_arrow
-
question_answer30)
Consider the consecutive reactions: \[A\xrightarrow{k=2\times {{10}^{-5}}{{s}^{-1}}}B\xrightarrow{k=8\times {{10}^{-6}}{{s}^{-1}}}\]\[C\xrightarrow{k=3\times {{10}^{-3}}{{s}^{-1}}}D\] The rate determining step of the reaction is:
A)
\[A\to B\] done
clear
B)
\[C\to D\] done
clear
C)
\[B\to C\] done
clear
D)
\[A\to D\] done
clear
View Solution play_arrow
-
question_answer31)
\[A+2B\to C\], the rate equation for this reaction is given as Rate \[=K\left[ A \right]\left[ B \right].\] If the concentration of A is kept the same but that of B is doubled what will happen to the rate itself?
A)
halved done
clear
B)
the same done
clear
C)
doubled done
clear
D)
quadrupled done
clear
View Solution play_arrow
-
question_answer32)
In the reaction of formation of sulphur trioxide by contact process \[2S{{O}_{2}}+{{O}_{2}}\rightleftharpoons 2S{{O}_{3}}\] the rate of reaction was measured as \[\frac{d[{{O}_{2}}]}{dt}=-2.5\times {{10}^{-4}}mol{{L}^{-1}}{{s}^{-1}}\]. The rate of reaction is terms of \[[S{{O}_{2}}]\] in \[mol\text{ }{{L}^{-1}}{{s}^{-1}}\] will be:
A)
\[-1.25\times {{10}^{-4}}\] done
clear
B)
\[-2.50\times {{10}^{-4}}\] done
clear
C)
\[-3.75\times {{10}^{-4}}\] done
clear
D)
\[-5.00\times {{10}^{-4}}\] done
clear
View Solution play_arrow
-
question_answer33)
For the reaction, \[2{{N}_{2}}{{O}_{5}}\to 4N{{O}_{2}}+{{O}_{2}}\] the rate equation can be expressed in two ways\[-\frac{d[{{N}_{2}}{{O}_{5}}]}{dt}=k[{{N}_{2}}{{O}_{5}}]\] and \[+\frac{d[N{{O}_{2}}]}{dt}=k'[{{N}_{2}}{{O}_{5}}]\] k and k' are related as:
A)
\[k=k'\] done
clear
B)
\[2k=k'\] done
clear
C)
\[k=2k'\] done
clear
D)
\[k=4k'\] done
clear
View Solution play_arrow
-
question_answer34)
The reaction \[2{{N}_{2}}{{O}_{5}}\rightleftharpoons 2{{N}_{2}}{{O}_{4}}+{{O}_{2}}\]
A)
bimolecular and of second order done
clear
B)
unimolecular and of first order done
clear
C)
bimolecular and of first order done
clear
D)
bimolecular and of zero order done
clear
View Solution play_arrow
-
question_answer35)
For the reaction system: \[2NO\left( g \right)+{{O}_{2}}\left( g \right)\to 2N{{O}_{2}}\left( g \right)\] volume is suddenly reduced to half its value by increasing the pressure on it. If the reaction is of first order with respect to \[{{O}_{2}}\] and second order with respect to NO, the rate of reaction will
A)
diminish to one-eighth of its initial value done
clear
B)
increase to eight times of its initial value done
clear
C)
increase to four times of its initial value done
clear
D)
diminish to one-fourth of its initial value done
clear
View Solution play_arrow
-
question_answer36)
Units of rate constant of first and zero order reactions in terms of molarity M unit are respectively
A)
\[se{{c}^{-1}},\text{ }M.se{{c}^{-1}}\] done
clear
B)
\[se{{c}^{-1}},\text{ }M\] done
clear
C)
\[M.se{{c}^{-1}},\text{ }se{{c}^{-1}}\] done
clear
D)
\[M,\text{ }se{{c}^{-1}}\] done
clear
View Solution play_arrow
-
question_answer37)
The plot of concentration of the reactant Vs time for a reaction is a straight line with a negative slope. The reaction follows a rate equation of
A)
zero order done
clear
B)
first order done
clear
C)
second order done
clear
D)
third order done
clear
View Solution play_arrow
-
question_answer38)
The differential rate law for the reaction \[{{H}_{2}}(g)+{{I}_{2}}(g)\to 2HI(g)\] is
A)
\[-\frac{d[{{H}_{2}}]}{dt}=-\frac{d[{{I}_{2}}]}{dt}=-\frac{d[HI]}{dt}\] done
clear
B)
\[\frac{d[{{H}_{2}}]}{dt}=\frac{d[{{I}_{2}}]}{dt}=\frac{1}{2}\frac{d[HI]}{dt}\] done
clear
C)
\[\frac{1}{2}\frac{d[{{H}_{2}}]}{dt}=\frac{1}{2}\frac{d[{{I}_{2}}]}{dt}=-\frac{d[HI]}{dt}\] done
clear
D)
\[-2\frac{d[{{H}_{2}}]}{dt}=-2\frac{d[{{I}_{2}}]}{dt}=\frac{d[HI]}{dt}\] done
clear
View Solution play_arrow
-
question_answer39)
Which of the following will react at the highest rate?
A)
1 mole of A and 1 mole of B in a 1 -L vessel done
clear
B)
2 mole of A and 2 mole of B in a 2-L vessel done
clear
C)
3 mole of A and 3 mole of B in a 3-L vessel done
clear
D)
All would react at the same rate done
clear
View Solution play_arrow
-
question_answer40)
In the reaction,\[A+2B\xrightarrow{{}}6C+2D\], If the initial rate \[-\frac{d[A]}{dt}\] at t = 0 is \[2.6\times {{10}^{-2}}M\text{ }se{{c}^{-1}}\] what will be the value of \[\frac{d[B]}{dt}\] at\[t=0\]?
A)
\[8.5\times {{10}^{-2}}M\text{ }se{{c}^{-1}}\] done
clear
B)
\[2.5\times {{10}^{-2}}M\text{ }se{{c}^{-1}}\] done
clear
C)
\[5.2\times {{10}^{-2}}M\text{ }se{{c}^{-1}}\] done
clear
D)
\[7.5\times {{10}^{-2}}M\text{ }se{{c}^{-1}}\] done
clear
View Solution play_arrow
-
question_answer41)
The reaction \[L\xrightarrow{{}}M\] is started with 10.0 g of L. After 30 and 90 minutes 5.0 g and 1.25 g of L respectively are left. The order of the reaction is
A)
0 done
clear
B)
1 done
clear
C)
2 done
clear
D)
3 done
clear
View Solution play_arrow
-
question_answer42)
A first order reaction is half-completed in 45 minutes. How long does it need for 99.9% of the reaction to be completed?
A)
20 hours done
clear
B)
10 hours done
clear
C)
\[7\frac{1}{2}\] hours done
clear
D)
5 hours done
clear
View Solution play_arrow
-
question_answer43)
A reaction which is of first order w.r.t. reactant A, has a rate constant 6 \[{{\min }^{-1}}\]. If we start with \[\left[ A \right]=0.5\text{ }mol\text{ }{{L}^{-1}}\], when would [A] reach the value of \[0.05\text{ }mol\,{{L}^{-1}}\]
A)
0.384 min done
clear
B)
0.15 min done
clear
C)
3 min done
clear
D)
3.84 min done
clear
View Solution play_arrow
-
question_answer44)
The time taken for 90% of a first order reaction to complete is approximately
A)
1.1 times that of half-life done
clear
B)
2.2 times that of half-life done
clear
C)
3.3 times that of half-life done
clear
D)
4.4 times that of half-life done
clear
View Solution play_arrow
-
question_answer45)
The rate constant of a reaction is\[0.0693\text{ }mi{{n}^{-1}}\]. Starting with 10 mol, the rate of the reaction after 10 minis
A)
\[0.0693\text{ }mol\text{ }mi{{n}^{-1}}\] done
clear
B)
\[0.0693\times 2\text{ }mol\text{ }mi{{n}^{-1}}\] done
clear
C)
\[0.0693\times 5\text{ }mol\text{ }mi{{n}^{-1}}\] done
clear
D)
\[0.0693\times {{\left( 5 \right)}^{2}}mol\text{ }mi{{n}^{-1}}\] done
clear
View Solution play_arrow
-
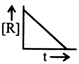
question_answer46)
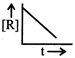
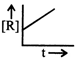
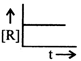
The plot that represents the zero order reaction is:
A)
B)
C)
D)
View Solution play_arrow
-
question_answer47)
Which of the following reactions is not of the first order?
A)
Inversion of sucrose in the presence of acid done
clear
B)
Acid-catalyzed hydrolysis of ethyl acetate done
clear
C)
Hydrolysis of tertiary butyl halide using alkali done
clear
D)
Oxidation of \[{{I}^{-}}\]ion by \[{{S}_{2}}O_{8}^{2-}\] ion done
clear
View Solution play_arrow
-
question_answer48)
A first order reaction is 50% completed in 20 minutes at \[27{}^\circ C\] and in 5 minutes at\[47{}^\circ C\]. The energy of activation of the reaction is:
A)
43.85 kJ/mol done
clear
B)
55.14 kJ/mol done
clear
C)
11.97 kJ/mol done
clear
D)
6.65 kJ/mol done
clear
View Solution play_arrow
-
question_answer49)
For the first order reaction \[A\xrightarrow{{}}B+C\] is carried out at\[27{}^\circ C\]. If \[3.8\times {{10}^{-16}}\] % of the reactant molecules exists in the activated state, the \[{{E}_{4}}\](activation energy) of the reaction is:
A)
12 kJ/mol done
clear
B)
831.4 kJ/mol done
clear
C)
100 kJ/mol done
clear
D)
88.57 kJ/mol done
clear
View Solution play_arrow
-
question_answer50)
A catalyst lowers the activation energy of a certain reaction from 83.314 to \[75\text{ }kJ\text{ }mo{{l}^{-1}}\] at 500 K. What will be the rate of reaction as compared to uncatalysed reaction? Assume other things being equal.
A)
Double done
clear
B)
28 times done
clear
C)
7.38 times done
clear
D)
\[7.38\times {{10}^{3}}\]times done
clear
View Solution play_arrow
-
question_answer51)
Rate of a reaction can be expressed by Arrhenius equation as: \[k=A{{e}^{-{{E}_{a}}}}^{/RT}\] In this equation, \[{{E}_{a}}\] represents
A)
the total energy of the reacting molecules at a temperature, T done
clear
B)
the fraction of molecules with energy greater than the activation energy of the reaction done
clear
C)
the energy below which all the colliding molecules will react done
clear
D)
the energy below which colliding molecules will not react done
clear
View Solution play_arrow
-
question_answer52)
The reason for almost doubling the rate of reaction on increasing the temperature of the reaction system by \[10{}^\circ C\] is
A)
The value of threshold energy increases done
clear
B)
Collision frequency increases done
clear
C)
The fraction of the molecule having energy equal to threshold energy or more increases done
clear
D)
Activation energy decreases done
clear
View Solution play_arrow
-
question_answer53)
In Arrhenius plot, intercept is equal to
A)
\[\frac{-{{E}_{a}}}{R}\] done
clear
B)
\[\ell nA\] done
clear
C)
\[\ell n\,K\] done
clear
D)
\[lo{{g}_{10}}A\] done
clear
View Solution play_arrow
-
question_answer54)
A reaction rate constant is given by \[k=1.2\times {{10}^{14}}{{e}^{-25000/RT}}se{{c}^{-1}}\]. It means
A)
log k versus log T will give a straight line with a slope as \[-25000\] done
clear
B)
log k versus T will give a straight line with slope as 25000 done
clear
C)
log k versus 1/T will give a straight line with slope as \[-25000/R\] done
clear
D)
log k versus 1/T will give a straight line done
clear
View Solution play_arrow
-
question_answer55)
The velocity of a reaction is doubled for every \[10{}^\circ C\] rise in temp. If the temp. is raised to \[50{}^\circ C\]from \[0{}^\circ C\] the reaction velocity increases by about
A)
12 times done
clear
B)
16 times done
clear
C)
32 times done
clear
D)
50 times done
clear
View Solution play_arrow
-
question_answer56)
Which of the following statements is incorrect?
A)
Activation energy for the forward reaction is equals to activation energy for the reverse reaction done
clear
B)
For a reversible reaction, an increase in temperature increases the reaction rate for both the forward and the backward reaction done
clear
C)
The larger the initial reactant concentration for a second order reaction, the shorter is its half-life. done
clear
D)
When \[\Delta t\] is infinitesimally small, the average rate equals the instantaneous rate done
clear
View Solution play_arrow
-
question_answer57)
Consider the following statements:
| I. Increase in concentration of reactant increases the rate of a zero order reaction. |
| II. Rate constant k is equal to collision frequency A if \[{{E}_{a}}=0.\] |
| III. Rate constant k is equal to collision frequency A if \[{{E}_{a}}=\infty .\] |
| IV. In k Vs T is a straight line. |
| V. In k Vs 1/T is a straight line. |
| Correct statements are |
A)
I and IV done
clear
B)
II and V done
clear
C)
III and IV done
clear
D)
II and III done
clear
View Solution play_arrow
-
question_answer58)
The time required for 10% completion of a first order reaction at 298 K is equal to that required for its 25% completion at 308 K, If the pre-exponential factor for the reaction is \[3.56\times {{10}^{9}}{{s}^{-1}}\] the rate constant at 318 K is:
A)
\[18.39\text{ }kcal\text{ }mo{{l}^{-1}}\] done
clear
B)
\[20\,kcal\,mo{{l}^{-1}}\] done
clear
C)
\[16\,kcal\,mo{{l}^{-1}}\] done
clear
D)
\[21.5\,kcal\,mo{{l}^{-1}}\] done
clear
View Solution play_arrow
-
question_answer59)
For the reaction, \[3A+2B\to C+D\], the differential rate law can be written as:
A)
\[\frac{1}{3}\frac{d[A]}{dt}=\frac{d[C]}{dt}=k{{[A]}^{n}}{{[B]}^{m}}\] done
clear
B)
\[-\frac{d[A]}{dt}=\frac{d[C]}{dt}=k{{[A]}^{n}}{{[B]}^{m}}\] done
clear
C)
\[+\frac{1}{3}\frac{d[A]}{dt}=-\frac{d[C]}{dt}=k{{[A]}^{n}}{{[B]}^{m}}\] done
clear
D)
\[-\frac{1}{3}\frac{d[A]}{dt}=\frac{d[C]}{dt}=k{{[A]}^{n}}{{[B]}^{m}}\] done
clear
View Solution play_arrow
-
question_answer60)
The instantaneous rate of disappearance of \[MnO_{4}^{-}\] ion in the following reaction is
| \[4.56\times {{10}^{-3}}M{{s}^{-1}}2MnO_{4}^{-}+10{{I}^{-}}+16{{H}^{+}}\to \] \[2M{{n}^{2+}}+5{{I}_{2}}+8{{H}_{2}}O\] |
| The rate of appearance \[{{I}_{2}}\] is: |
A)
\[4.56\times {{10}^{-4}}M{{s}^{-1}}\] done
clear
B)
\[1.14\times {{10}^{-2}}M{{s}^{-1}}\] done
clear
C)
\[1.14\times {{10}^{-3}}M{{s}^{-1}}\] done
clear
D)
\[5.7\times {{10}^{-3}}M{{s}^{-1}}\] done
clear
View Solution play_arrow
-
question_answer61)
The rate constant of a zero order reaction is\[2.0\times {{10}^{-2}}mol\,{{L}^{-1}}{{s}^{-1}}\]. If the concentration of the reactant after 25 seconds is 0.5 M. What is the initial concentration?
A)
0.5 M done
clear
B)
1.25 M done
clear
C)
12.5 M done
clear
D)
\[1.0\,M\] done
clear
View Solution play_arrow
-
question_answer62)
The reaction \[A\to B\] follows first order kinetics. The time taken for 0.8 mole of A to produce 0.6 mole of B is 1 hour. What is the time taken for conversion of 0.9 mole of A to produce 0.675 mole of B?
A)
2 hours done
clear
B)
1 hour done
clear
C)
0.5 hour done
clear
D)
0.25 hour done
clear
View Solution play_arrow
-
question_answer63)
The rate of a first order reaction is \[1.5\times {{10}^{-2}}mol\text{ }{{L}^{-1}}{{\min }^{-1}}\] at 0.5 M concentration of the reactant. The half-life of the reaction is
A)
0.383 min done
clear
B)
23.1 min done
clear
C)
8.73 min done
clear
D)
7.53 min done
clear
View Solution play_arrow
-
question_answer64)
For a first order reaction \[A\to B\] the reaction rate at reactant concentration of 0.01 M is found to be\[2.0\times {{10}^{-5}}mol\text{ }{{L}^{-1}}\text{ }{{s}^{-1}}\]. The half-life period of the reaction is
A)
30 s done
clear
B)
220 s done
clear
C)
300 s done
clear
D)
347 s done
clear
View Solution play_arrow
-
question_answer65)
A reaction proceeds by first order, 75% of this reaction was completed in 32 min. The time required for 50% completion is
A)
8 min done
clear
B)
16 min done
clear
C)
20 min done
clear
D)
24 min done
clear
View Solution play_arrow
-
question_answer66)
Consider the reaction: \[{{N}_{2}}(g)+3{{H}_{2}}(g)\to 2N{{H}_{3}}(g)\] The equality relationship between \[\frac{d[N{{H}_{3}}]}{dt}\] and \[-\frac{d[{{H}_{2}}]}{dt}\] is
A)
\[+\frac{d[N{{H}_{3}}]}{dt}=-\frac{2}{3}\frac{d[{{H}_{2}}]}{dt}\] done
clear
B)
\[+\frac{d[N{{H}_{3}}]}{dt}=-\frac{3}{2}\frac{d[{{H}_{2}}]}{dt}\] done
clear
C)
\[\frac{d[N{{H}_{3}}]}{dt}=-\frac{d[{{H}_{2}}]}{dt}\] done
clear
D)
\[\frac{d[N{{H}_{3}}]}{dt}=-\frac{1}{3}\frac{d[{{H}_{2}}]}{dt}\] done
clear
View Solution play_arrow
-
question_answer67)
The rate of a reaction increases four-fold when the concentration of reactant is increased 16 times. If the rate of reaction is \[4\times {{10}^{-6}}mol\text{ }{{L}^{-1}}\text{ }{{s}^{-1}}\]when the concentration of the reactant is\[4\times {{10}^{-4}}mol\text{ }{{L}^{-1}}\]. The rate constant of the reaction will be
A)
\[2\times {{10}^{-4}}mo{{l}^{1/2}}{{L}^{-1/2}}{{s}^{-1}}\] done
clear
B)
\[1\times {{10}^{-2}}{{s}^{-1}}\] done
clear
C)
\[2\times {{10}^{-4}}mo{{l}^{-1/2}}{{L}^{1/2}}{{s}^{-1}}\] done
clear
D)
\[25\text{ }mo{{l}^{-1}}L\text{ }mi{{n}^{-1}}\] done
clear
View Solution play_arrow
-
question_answer68)
In the reaction\[A\to B+C\], rate constant is\[0.001\text{ }M{{s}^{-1}}\]. If we start with 1 M of A then cone. of A and B after 10 minuter are respectively.
A)
0.5 M, 0.5 M done
clear
B)
0.6 M, 0.4 M done
clear
C)
0.4 M, 0.6 M done
clear
D)
0.6 M 0.5 M done
clear
View Solution play_arrow
-
question_answer69)
The half-life for the virus inactivation if in the beginning 1.5% of the virus is inactivated per minute is (Given: The reaction is of first order)
A)
76 min done
clear
B)
66 min done
clear
C)
56 min done
clear
D)
46 min done
clear
View Solution play_arrow
-
question_answer70)
The reaction of \[{{A}_{2}}\] and \[{{B}_{2}}\] follows the equation \[{{A}_{2}}(g)+{{B}_{2}}(g)\to 2AB(g)\] The following data were observed
| \[{{[{{A}_{2}}]}_{0}}\] |
\[{{[{{B}_{2}}]}_{0}}\] |
Initial rate of appearance of \[AB(g)\,(in\,M{{s}^{-1}})\] |
| 0.10 |
0.10 |
\[2.5\times {{10}^{-4}}\] |
| 0.20 |
0.10 |
\[5\times {{10}^{-4}}\] |
| 0.20 |
0.20 |
\[10\times {{10}^{-4}}\] |
The value of rate constant for the above reaction is:
A)
\[2.5\times {{10}^{-4}}\] done
clear
B)
\[2.5\times {{10}^{-2}}\] done
clear
C)
\[1.25\times {{10}^{-2}}\] done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer71)
In the presence of an acid, the initial concentration of cane sugar was reduced from 0.20 to 0.10 M in 5 hours and from 0.2 to 0.05 M in 10 hours. The reaction is of;
A)
Zero order done
clear
B)
First order done
clear
C)
Second order done
clear
D)
Third order done
clear
View Solution play_arrow
-
question_answer72)
The activation energies of the forward and backward reactions in the case of a chemical reaction are 30.5 and 45.4 kJ/mol respectively. The reaction is:
A)
exothermic done
clear
B)
endothermic done
clear
C)
neither exothermic nor endothermic done
clear
D)
independent of temperature done
clear
View Solution play_arrow
-
question_answer73)
A homogeneous catalytic reaction takes place through the three alternative plots A, B and C shown in the given figure. Which one of the following indicates the relative ease with which the reaction can take place?
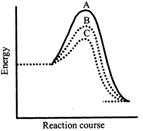
A)
A>B>C done
clear
B)
C>B>A done
clear
C)
A>C>B done
clear
D)
A=B=C done
clear
View Solution play_arrow
-
question_answer74)
A catalyst is a substance which:
A)
is always in the same phase as in the reaction done
clear
B)
alters the equilibrium in a reaction done
clear
C)
does not participate in the reaction but alters the rate of reaction done
clear
D)
participates in the reaction and provides an easier pathway for the same done
clear
View Solution play_arrow
-
question_answer75)
For an exothermic reaction, the energy of activation of the reactants is
A)
equal to the energy of activation of products done
clear
B)
less than the energy of activation of products done
clear
C)
greater than the energy of activation of products done
clear
D)
sometimes greater and sometimes less than that of the products done
clear
View Solution play_arrow
-
question_answer76)
When a biochemical reaction is carried out in laboratory in the absence of enzyme then rate of reaction obtained is \[{{10}^{-6}}\] times, then activation energy of reaction in the presence of enzyme is
A)
\[\frac{6}{RT}\] done
clear
B)
different from \[{{E}_{a}}\] obtained in laboratory, done
clear
C)
P is required done
clear
D)
can't say anything done
clear
View Solution play_arrow
-
question_answer77)
A chemical reaction was carried out at 300 K and 280 K. The rate constants were found to be \[{{k}_{1}}\] and \[{{k}_{2}}\] respectively, then
A)
\[{{k}_{2}}=4{{k}_{1}}\] done
clear
B)
\[{{k}_{2}}=2{{k}_{1}}\] done
clear
C)
\[{{k}_{2}}=0.25\,{{k}_{1}}\] done
clear
D)
\[{{k}_{2}}=0.5{{k}_{1}}\] done
clear
View Solution play_arrow
-
question_answer78)
In a reaction at \[27{}^\circ C,{{10}^{-3}}\]% reactant molecules manage to cross over the barrier of transition state. The energy of these molecules in excess of the average value will be \[(R=2cal\text{ }{{K}^{-1}}\text{ }mo{{l}^{-1}})\]:
A)
\[6.91\text{ kc}al\text{ }mo{{l}^{-1}}\] done
clear
B)
\[3.00\text{ kc}al\text{ }mo{{l}^{-1}}\] done
clear
C)
\[4.15\,kcal\,mo{{l}^{-1}}\] done
clear
D)
\[5.10kcal\,mo{{l}^{-1}}\] done
clear
View Solution play_arrow
-
question_answer79)
The activation energy for a simple chemical reaction \[A\to B\] is \[{{E}_{a}}\] in forward direction. The activation energy for reverse reaction
A)
is always double of \[{{E}_{a}}\] done
clear
B)
is negative of \[{{E}_{a}}\] done
clear
C)
is always less than \[{{E}_{a}}\] done
clear
D)
can be less than or more than \[{{E}_{a}}\] done
clear
View Solution play_arrow
-
question_answer80)
The slope in Arrhenius plot, is equal to:
A)
\[-\frac{{{E}_{a}}}{2.303R}\] done
clear
B)
\[\frac{{{E}_{a}}}{R}\] done
clear
C)
\[-\frac{R}{2.303{{E}_{a}}}\] done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer81)
A radioactive isotope having a half - life period of 3 days was received after 12 days. If 3g of the isotope is left in the container, what would be the initial mass of the isotope?
A)
12g done
clear
B)
36g done
clear
C)
48g done
clear
D)
24g done
clear
View Solution play_arrow
-
question_answer82)
In a chemical reaction A is converted into B. The rates of reaction, starting with initial concentrations of A as \[2\times {{10}^{-3}}M\] and \[1\times {{10}^{-3}}M,\] are equal to \[2.40\times {{10}^{-4}}M{{s}^{-1}}\] and \[0.60\times {{10}^{-4}}M{{s}^{-1}}\] respectively. The order of reaction with respect to reactant A will be
A)
0 done
clear
B)
1.5 done
clear
C)
1 done
clear
D)
2 done
clear
View Solution play_arrow
-
question_answer83)
For a reaction \[A\to \] Products, a plot of log \[{{t}_{1/2}}\]versus \[\log {{a}_{0}}\] is shown in the figure. If the initial concentration of A is represented by \[{{a}_{0}}\], the order of the reaction is
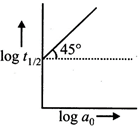
A)
one done
clear
B)
zero done
clear
C)
two done
clear
D)
three done
clear
View Solution play_arrow
-
question_answer84)
The rate constant of a reaction with a virus is \[3.3\times {{10}^{-4}}{{s}^{-1}}\]. Time required for the virus to become 75% inactivated is
A)
35 min done
clear
B)
70 min done
clear
C)
105 min done
clear
D)
17.5 min done
clear
View Solution play_arrow
-
question_answer85)
A Geigger Muller counter is used to study the radioactive process. In the absence of radioactive substance A, it counts 3 disintegration per second (dps). At the start in the presence of A, it records 23 dps; and after 10 min 13 dps,
| (i) What does it count after 20 min |
| (ii) What is the half life of A? |
A)
8 dps, 10 min done
clear
B)
5 dps, 10 min done
clear
C)
5 dps, 20 min done
clear
D)
5 dps, 5 min done
clear
View Solution play_arrow
-
question_answer86)
The half life period for catalytic decomposition of \[A{{B}_{3}}\] at 50 mm Hg is 4 hrs and at 100 mm Hg it is 2 hrs. The order of reaction is
A)
1 done
clear
B)
3 done
clear
C)
2 done
clear
D)
0 done
clear
View Solution play_arrow
-
question_answer87)
The rate equation for a reaction, \[{{N}_{2}}O\xrightarrow{{}}{{N}_{2}}+1/2{{O}_{2}}\] is Rate \[=k{{[{{N}_{2}}O]}^{0}}=k.\] If the initial concentration of the reactant is a mol\[Li{{t}^{-1}}\], the half-life period of the reaction is
A)
\[{{t}_{\frac{1}{2}}}=\frac{a}{2k}\] done
clear
B)
\[-{{t}_{\frac{1}{2}}}=ka\] done
clear
C)
\[{{t}_{\frac{1}{2}}}=\frac{a}{k}\] done
clear
D)
\[{{t}_{\frac{1}{2}}}=\frac{k}{a}\] done
clear
View Solution play_arrow
-
question_answer88)
The rate constant for a first order reaction whose half life is 480 sec, is:
A)
\[1.44\times {{10}^{-3}}se{{c}^{-1}}\] done
clear
B)
\[1.44\times se{{c}^{-1}}\] done
clear
C)
\[0.72\times {{10}^{-3}}se{{c}^{-1}}\] done
clear
D)
\[2.88\times {{10}^{-3}}se{{c}^{-1}}\] done
clear
View Solution play_arrow
-
question_answer89)
The hypothetical reaction \[{{A}_{2}}+{{B}_{2}}\xrightarrow{{}}2AB\]; follows the following mechanism \[{{A}_{2}}\xrightarrow{Fast}A+A,\] \[A+{{B}_{2}}\xrightarrow{slow}AB+B,A+B\xrightarrow{Fast}AB\] The order of the overall reaction is
A)
0 done
clear
B)
1 done
clear
C)
2 done
clear
D)
3/2 done
clear
View Solution play_arrow
-
question_answer90)
If half-life of a substance is 5 yrs, then the total amount of substance left after 15 years, when initial amount is 64 grams is
A)
16g done
clear
B)
2g done
clear
C)
32g done
clear
D)
8g done
clear
View Solution play_arrow
-
question_answer91)
In a reversible reaction the energy of activation of the forward reaction is 50 kcal. The energy of activation for the reverse reaction will be
A)
< 50 kcal done
clear
B)
either greater than or less than 50 kcal done
clear
C)
50 kcal done
clear
D)
> 50 kcal done
clear
View Solution play_arrow
-
question_answer92)
Activation energy of a chemical reaction can be determined by
A)
evaluating rate constant at standard temperature done
clear
B)
evaluating velocities of reaction at two different temperatures done
clear
C)
evaluating rate constants at two different temperatures done
clear
D)
changing concentration of reactants done
clear
View Solution play_arrow
-
question_answer93)
For the exothermic reaction \[A+B\to C+D,\text{ }\Delta H\]is the heat of reaction and \[{{E}_{a}}\]is the energy of activation. The energy of activation for the formation of A + B will be
A)
\[{{E}_{a}}\] done
clear
B)
\[\Delta H\] done
clear
C)
\[{{E}_{a}}+\Delta H\] done
clear
D)
\[\Delta H-{{E}_{a}}\] done
clear
View Solution play_arrow
-
question_answer94)
A reaction having equal energies of activation for forward and reverse reaction has:
A)
\[\Delta G=0\] done
clear
B)
\[\Delta H=0\] done
clear
C)
\[\Delta H=\Delta G=\Delta S=0\] done
clear
D)
\[\Delta S=0\] done
clear
View Solution play_arrow
-
question_answer95)
The rate coefficient (K) for a particular reactions is \[1.3\times {{10}^{-4}}{{M}^{-1}}{{s}^{-1}}\] at\[100{}^\circ C\], and \[1.3\times {{10}^{-3}}{{M}^{-1}}{{s}^{-1}}\] at\[150{}^\circ C\]. What is the energy of activation \[\left( {{E}_{a}} \right)\] (in kJ) for this reaction? (R = molar gas constant\[=8.314\text{ }J{{K}^{-1}}mo{{l}^{-1}}\])
A)
16 done
clear
B)
60 done
clear
C)
99 done
clear
D)
132 done
clear
View Solution play_arrow
-
question_answer96)
The reaction \[X\to Y\] is an exothermic reaction. Activation energy of the reaction for X into Y is\[150\,kJ\,mo{{l}^{-1}}\]. Enthalpy of reaction is\[135\text{ }k\,J\,mo{{l}^{-1}}\]. The activation energy for the reverse reaction, \[Y\to X\] will be:
A)
\[280\,kJ\,mo{{l}^{-1}}\] done
clear
B)
\[285\text{ }kJmo{{l}^{-1}}\] done
clear
C)
\[270\text{ }kJ\,mo{{l}^{-1}}\] done
clear
D)
\[15\,kJ\,mo{{l}^{-1}}\] done
clear
View Solution play_arrow
-
question_answer97)
The activation energy for a reaction which doubles the rate when the temperature is raised from 298 K to 308 K is
A)
\[59.2\text{ }kJ\,mo{{l}^{-1}}\] done
clear
B)
\[39.2\text{ kJ }mo{{l}^{-1}}\] done
clear
C)
\[52.9\,kJ\,mo{{l}^{-1}}\] done
clear
D)
\[29.5\,kJ\,mo{{l}^{-1}}\] done
clear
View Solution play_arrow
-
question_answer98)
\[{{t}_{1/4}}\] can be taken as the time taken for the concentration of a reactant to drop to \[\frac{3}{4}\] of its initial value. If the rate constant for a first order reaction is k, the \[{{t}_{1/4}}\] can be written as
A)
\[0.75/k\] done
clear
B)
\[0.69/k\] done
clear
C)
\[0.29/k\] done
clear
D)
\[0.10/k\] done
clear
View Solution play_arrow
-
question_answer99)
\[A\to B,\Delta H=10kJ\,mo{{l}^{-1}},{{E}_{a(f)}}=50kJ\,mo{{l}^{-1}}\], then \[{{E}_{a}}\] of \[B\to A\] will be
A)
\[40kJ\,mo{{l}^{-1}}\] done
clear
B)
\[50kJ\,mo{{l}^{-1}}\] done
clear
C)
\[-50kJ\,mo{{l}^{-1}}\] done
clear
D)
\[60kJ\,mo{{l}^{-1}}\] done
clear
View Solution play_arrow
-
question_answer100)
For a certain reaction, rate\[=k\times {{\left[ {{H}^{+}} \right]}^{n}}\]. If pH of reaction changes from two to one, the rate becomes 100 times of its value at pH = 2, the order of reaction is-
A)
1 done
clear
B)
2 done
clear
C)
0 done
clear
D)
3 done
clear
View Solution play_arrow
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
