A) 0
B) 1.5 gm
C) 2.5 gm
D) 2 gm
Correct Answer: B
Solution :
Remember mass of the metal deposited on cathode depends on the current through the voltameter and not on the current supplied by the battery. Hence by using \[m=Zit,\] we can say \[\frac{{{m}_{Parallel}}}{{{m}_{Series}}}=\frac{{{i}_{Parallel}}}{{{i}_{Series}}}\] Þ \[{{m}_{Parallel}}=\frac{5}{2}\times 1=2.5\,\,gm.\] Hence increase in mass = 2.5 ? 1 = 1.5 gm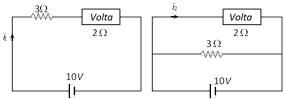
You need to login to perform this action.
You will be redirected in
3 sec
