A) \[{{120}^{o}}\] and \[{{109.5}^{o}}\]
B) \[{{90}^{o}}\] and \[{{109.5}^{o}}\]
C) \[{{109.5}^{o}}\] and \[{{90}^{o}}\]
D) \[{{109.5}^{o}}\] and \[{{120}^{o}}\]
Correct Answer: A
Solution :
\[120{}^\circ \] and \[109.5{}^\circ \] \[\underset{\text{Bond angle }=\text{ 12}{{\text{0}}^{\text{o}}}}{\mathop{\underset{s{{p}^{2}}\text{-hybridization}}{\mathop{Cl-\underset{Cl}{\mathop{\underset{|}{\mathop{C}}\,}}\,=\underset{Cl}{\mathop{\underset{|}{\mathop{C}}\,}}\,-Cl}}\,}}\,\]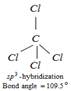
You need to login to perform this action.
You will be redirected in
3 sec
