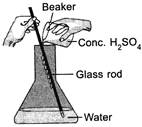
| 1. Large volume of acid should be added to a small volume of water slowly. |
| 2. The figure represents the process of concentrating dilute\[{{H}_{2}}S{{O}_{4}}\]. |
| 3. The process involves release of a large amount of heat. |
| 4. Water should not be stirred. |
A) 1 and 4 only
B) 2 and 3 only
C) 1, 2 and 4 only
D) 2 only
Correct Answer: C
Solution :
Dilution of \[{{H}_{2}}S{{O}_{4}}\]involves release of a large amount of heat. Therefore, instead of adding water to concentrated acid, acid should be added to a large volume of water gently along the sides of glass rod while stirring the water slowly.You need to login to perform this action.
You will be redirected in
3 sec
