-
question_answer1)
Radius of atom is of the order
A)
\[{{10}^{-10}}-{{10}^{-8}}cm\] done
clear
B)
\[{{10}^{-12}}-{{10}^{-14}}cm\] done
clear
C)
\[{{10}^{-14}}-{{10}^{-16}}cm\] done
clear
D)
\[{{10}^{-16}}-{{10}^{-20}}cm\] done
clear
View Solution play_arrow
-
question_answer2)
Mass of electron is
A)
\[\text{9}.\text{1}0\text{83 }\times \text{ 1}{{0}^{-\text{31}}}\text{ kg}\] done
clear
B)
\[\text{9}.\text{1}0\text{83 }\times \text{ 1}{{0}^{-\text{24}}}\,\text{kg}\] done
clear
C)
\[\text{9}.\text{1}0\text{83 }\times \text{ 1}{{0}^{-\text{28}}}\text{ kg}\] done
clear
D)
\[\text{1}.\text{67 }\times \text{ 1}{{0}^{-\text{24}}}\text{ gm}\] done
clear
View Solution play_arrow
-
question_answer3)
Rutherford's scattering experiment is related to
A)
neutrons done
clear
B)
atom done
clear
C)
election done
clear
D)
nucleus done
clear
View Solution play_arrow
-
question_answer4)
Size of the nucleus is of the order
A)
\[{{10}^{-12}}m\] done
clear
B)
\[\text{1}{{0}^{-\text{8}}}\text{m}\] done
clear
C)
\[\text{1}{{0}^{-\text{15}}}\text{ m}\] done
clear
D)
\[\text{1}{{0}^{\text{-10}}}\text{ m}\] done
clear
View Solution play_arrow
-
question_answer5)
The electron is
A)
\[\alpha -\text{ray}\] particle done
clear
B)
\[\beta -\text{ray}\] particle done
clear
C)
positron done
clear
D)
\[\gamma -\text{ray}\] done
clear
View Solution play_arrow
-
question_answer6)
The \[\frac{e}{m}\] ratio of an electron for all types of substances is
A)
constant done
clear
B)
less than one done
clear
C)
greater than one done
clear
D)
zero done
clear
View Solution play_arrow
-
question_answer7)
A proton is identical to
A)
the nucleus of helium done
clear
B)
the nucleus of a hydrogen atom done
clear
C)
a molecule of a hydrogen done
clear
D)
an atom of hydrogen done
clear
View Solution play_arrow
-
question_answer8)
Isotopes have the same
A)
Atomic number done
clear
B)
Electronic configuration done
clear
C)
Mass number-neutron done
clear
D)
All of the above done
clear
View Solution play_arrow
-
question_answer9)
Designation of an orbital with n = 6 and\[l=4\], is
A)
6s done
clear
B)
\[6d\] done
clear
C)
\[6f\] done
clear
D)
\[6p\] done
clear
View Solution play_arrow
-
question_answer10)
Principal quantum number determines,
A)
Atomic size done
clear
B)
Orbital angular momentum done
clear
C)
Shape of the electronic cloud done
clear
D)
Orientation of orbitals done
clear
View Solution play_arrow
-
question_answer11)
Electronic configuration \[\text{1}{{\text{s}}^{\text{2}}},\text{ 2}{{\text{s}}^{\text{2}}},\text{ 2}{{\text{p}}^{\text{3}}}\] represents the element
A)
\[O\] done
clear
B)
\[P\] done
clear
C)
\[N\] done
clear
D)
\[Na\] done
clear
View Solution play_arrow
-
question_answer12)
Electron promotion rule is related to
A)
\[2s\to 2p\] done
clear
B)
\[2s\to 3s\] done
clear
C)
\[2s\to 3p\] done
clear
D)
\[1s\to 2s\] done
clear
View Solution play_arrow
-
question_answer13)
The number of valency electrons for carbon are
A)
2 done
clear
B)
4 done
clear
C)
6 done
clear
D)
8 done
clear
View Solution play_arrow
-
question_answer14)
Atom and ion differ in
A)
the relative size done
clear
B)
the configuration done
clear
C)
the presence of charge done
clear
D)
all of these done
clear
View Solution play_arrow
-
question_answer15)
Value of \[\frac{e}{m}\] of an electron was measured by
A)
Millikan done
clear
B)
J.J. Thomson done
clear
C)
Dalton done
clear
D)
Rutherford done
clear
View Solution play_arrow
-
question_answer16)
The atomic number of an element is 11 and its mass number is 23. The perspective number of electrons, protons and neutrons in this atom will be
A)
11, 11, 12 done
clear
B)
11, 12, 11 done
clear
C)
12, 11, 11 done
clear
D)
23, 11, 23 done
clear
View Solution play_arrow
-
question_answer17)
Number of electrons in the outer shell of the most stable or inert atoms is
A)
1 done
clear
B)
4 done
clear
C)
6 done
clear
D)
8 done
clear
View Solution play_arrow
-
question_answer18)
Which of the following pairs are isotopes?
A)
Oxygen and ozone done
clear
B)
Ice and steam done
clear
C)
Nitric oxide and nitrogen dioxide done
clear
D)
Hydrogen and deuterium done
clear
View Solution play_arrow
-
question_answer19)
An atom which has a mass number of 14 and has 8 neutrons is an
A)
isotope of oxygen done
clear
B)
isobar of oxygen done
clear
C)
isotope of carbon done
clear
D)
isobar of carbon done
clear
View Solution play_arrow
-
question_answer20)
The space between proton and electron in hydrogen atom is
A)
absolutely empty done
clear
B)
full of electromagnetic radiation done
clear
C)
full of air done
clear
D)
full of ether done
clear
View Solution play_arrow
-
question_answer21)
The number of orbitals present in Y shell are
A)
4 done
clear
B)
2 done
clear
C)
1 done
clear
D)
3 done
clear
View Solution play_arrow
-
question_answer22)
Ne, \[\text{N}{{\text{a}}^{+}}\]and \[{{F}^{-}}\] have the same
A)
Mass number done
clear
B)
Number of nucleous done
clear
C)
Number of electrons done
clear
D)
Number of protons done
clear
View Solution play_arrow
-
question_answer23)
Name of scientist associated with the discovery of neutron is
A)
J.J. Thomson done
clear
B)
James Chadwick done
clear
C)
Rutherford done
clear
D)
Yukawa done
clear
View Solution play_arrow
-
question_answer24)
Which of the following has an equal number of neutrons and protons?
A)
Hydrogen done
clear
B)
Deuterium done
clear
C)
Fluorine done
clear
D)
Chlorine done
clear
View Solution play_arrow
-
question_answer25)
Members of which of the following have similar chemical properties?
A)
Isotope done
clear
B)
Isobars done
clear
C)
Allotropes done
clear
D)
Both isotopes and allotropes done
clear
View Solution play_arrow
-
question_answer26)
An atom of an element has 26 electrons and has a mass number 56. The nucleus of this atom contains .......... neutrons.
A)
26 done
clear
B)
36 done
clear
C)
30 done
clear
D)
56 done
clear
View Solution play_arrow
-
question_answer27)
For an element with atomic number 19, the 19th electron will occupy
A)
L-shell done
clear
B)
M-shell done
clear
C)
N-shell done
clear
D)
K-shell done
clear
View Solution play_arrow
-
question_answer28)
Density of neutron is of the order of
A)
\[\text{1}{{0}^{\text{3}}}\text{ kg}/\text{cc}\] done
clear
B)
\[\text{1}{{0}^{\text{6}}}\text{ kg}/\text{cc}\] done
clear
C)
\[\text{1}{{0}^{\text{12}}}\text{ kg}/\text{cc}\] done
clear
D)
\[\text{1}{{0}^{\text{2}}}\text{ kg}/\text{cc}\] done
clear
View Solution play_arrow
-
question_answer29)
A completely filled and half filled orbit is spherically symmetrical. Point out which has spherical symmetry.
A)
\[\text{Na}\] done
clear
B)
\[C\] done
clear
C)
\[C{{l}^{-}}\] done
clear
D)
\[S\] done
clear
View Solution play_arrow
-
question_answer30)
There is no difference between a 2p and 3p orbitals regarding
A)
shape done
clear
B)
size done
clear
C)
energy done
clear
D)
value of n done
clear
View Solution play_arrow
-
question_answer31)
Following electronic configuration violates
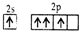
A)
only Paulis rule done
clear
B)
only Hund's rule done
clear
C)
only (n + 1) rule done
clear
D)
all of these done
clear
View Solution play_arrow
-
question_answer32)
The presence of nucleus in an atom was established by
A)
John Dalton done
clear
B)
Rutherford done
clear
C)
J.J. Thomson done
clear
D)
J. Moore done
clear
View Solution play_arrow
-
question_answer33)
The number of electrons possessed by a cation are equal to
A)
The atomic number of the element done
clear
B)
The atomic number + charge on the cation done
clear
C)
The atomic number \[-\] charge on the cation done
clear
D)
Mass number \[-\] atomic number done
clear
View Solution play_arrow
-
question_answer34)
The atomic weight of an atom is due to
A)
Electrons and protons done
clear
B)
Electrons and neutrons done
clear
C)
Neutrons and protons done
clear
D)
Electrons, neutrons and protons done
clear
View Solution play_arrow
-
question_answer35)
The relative atomic masses of many elements are not whole numbers because
A)
they cannot be determined very accurately done
clear
B)
the atoms ionize during the determinations done
clear
C)
of the existence of isotopes done
clear
D)
of the presence of impurities done
clear
View Solution play_arrow
-
question_answer36)
Explanation of the presence of three unpaired electrons in nitrogen atom is given by
A)
Fault's principle done
clear
B)
Hund's rule done
clear
C)
Aufbau principle done
clear
D)
Uncertainty principle done
clear
View Solution play_arrow
-
question_answer37)
Though three fundamental particles are present in almost all elements. One element does not have
A)
neutron done
clear
B)
proton done
clear
C)
electron done
clear
D)
nucleons done
clear
View Solution play_arrow
-
question_answer38)
In a given atom no two electrons can have the same values for all the four quantum numbers This is called
A)
Hund's rule done
clear
B)
Aufbau principle done
clear
C)
Uncertainty principle done
clear
D)
Fault's exclusion principle done
clear
View Solution play_arrow
-
question_answer39)
Energy of an electron is specified by
A)
n done
clear
B)
1 done
clear
C)
n and 1 done
clear
D)
s done
clear
View Solution play_arrow
-
question_answer40)
Which of the following atoms has a valency equal to zero?
A)
Hydrogen done
clear
B)
Lithium done
clear
C)
Neon done
clear
D)
Oxygen done
clear
View Solution play_arrow
-
question_answer41)
Which of the following is the configuration of silver atom in ground energy state?
A)
\[\left[ \text{Kr} \right]\text{ 3}{{\text{d}}^{\text{1}0}}\text{ 4}{{\text{s}}^{\text{1}}}\] done
clear
B)
\[[Xe]4{{f}^{14}}5{{d}^{10}}6{{s}^{1}}\] done
clear
C)
\[\left[ \text{Kr} \right]\text{ 4}{{\text{d}}^{\text{1}0}}\text{ 5}{{\text{s}}^{\text{1}}}\] done
clear
D)
\[\left[ \text{Kr} \right]\text{ 4}{{\text{d}}^{\text{9}}}\text{ 5}{{\text{s}}^{\text{2}}}\] done
clear
View Solution play_arrow
-
question_answer42)
A correct set of quantum numbers for the unpaired electron of chlorine atom is
A)
\[n-2\,\,\,\,\,\,\,\,\,\,\,l-1\,\,\,\,\,\,\,\,m-0\] done
clear
B)
\[n-2\,\,\,\,\,\,\,\,\,\,\,l-1\,\,\,\,\,\,\,\,m-1\] done
clear
C)
\[n-3\,\,\,\,\,\,\,\,\,\,\,l-1\,\,\,\,\,\,\,\,m-1\] done
clear
D)
\[n-3\,\,\,\,\,\,\,\,\,\,\,l-0\,\,\,\,\,\,\,\,m-0\] done
clear
View Solution play_arrow
-
question_answer43)
Which of the following is the correct configuration of chromium atom in ground energy state?
A)
\[~\left[ \text{Ar} \right]\text{ 3}{{\text{d}}^{\text{5}}}\text{ 4}{{\text{s}}^{\text{1}}}\] done
clear
B)
\[~[Ar]\text{3}{{\text{d}}^{4}}\text{ 4}{{\text{s}}^{2}}\] done
clear
C)
\[\left[ \text{Ar} \right]\text{ 3}{{\text{d}}^{\text{6}}}\text{ 4}{{\text{s}}^{\text{0}}}\] done
clear
D)
\[\left[ \text{Kr} \right]\text{ 4}{{\text{d}}^{\text{5}}}\text{ 5}{{\text{s}}^{\text{1}}}\] done
clear
View Solution play_arrow
-
question_answer44)
According to uncertainty principle
A)
\[E=h\upsilon \] done
clear
B)
\[\lambda =\frac{h}{p}\] done
clear
C)
\[\Delta x\times \Delta p=\frac{h}{4\pi }\] done
clear
D)
\[\Delta x\times \Delta p=\frac{h}{2\pi }\] done
clear
View Solution play_arrow
-
question_answer45)
2p orbitals have :
A)
n = 1,\[l\] = 2 done
clear
B)
n = 1,\[l\] = 0 done
clear
C)
n =2,\[l\] = 1 done
clear
D)
n = 2,\[l\] = 0 done
clear
View Solution play_arrow
-
question_answer46)
Size of nucleus is measured in
A)
amu done
clear
B)
Angstrom done
clear
C)
Fermi done
clear
D)
cm done
clear
View Solution play_arrow
-
question_answer47)
Bohr's model explains
A)
the spectrum of hydrogen atom only done
clear
B)
the spectrum of any atom or ion having on electron only done
clear
C)
the spectrum of hydrogen molecule done
clear
D)
the solar spectrum done
clear
View Solution play_arrow
-
question_answer48)
Positron has nearly the same mass as that of
A)
proton done
clear
B)
neutron done
clear
C)
electron done
clear
D)
a-particle done
clear
View Solution play_arrow
-
question_answer49)
Magnetic quantum number is related to
A)
size of orbitals done
clear
B)
shape of orbitals done
clear
C)
orientation of orbitals in the space done
clear
D)
All of these done
clear
View Solution play_arrow
-
question_answer50)
De-broglie relation describes the relationship of wavelength of an electron with
A)
mass done
clear
B)
energy done
clear
C)
momentum done
clear
D)
charge done
clear
View Solution play_arrow
-
question_answer51)
Total number of unpaired electrons in d-orbitals of an atom of element of atomic number 29 is
A)
10 done
clear
B)
1 done
clear
C)
Zero done
clear
D)
5 done
clear
View Solution play_arrow
-
question_answer52)
Which of the following quantities is always in whole number?
A)
Atomic radius done
clear
B)
Atomic number done
clear
C)
Mass number done
clear
D)
Equivalent weight done
clear
View Solution play_arrow
-
question_answer53)
he cathode rays tube experiments carried out by J.J. Thomson demonstrated that
A)
at ratio of charge to-mass for the particles of the cathode rays varies as different gases are placed in the tube done
clear
B)
the mass of an atom is essentially all contained in its very small nucleus done
clear
C)
cathode rays are streams of negatively charged ions done
clear
D)
the charge-to-mass ratio for a proton done
clear
View Solution play_arrow
-
question_answer54)
For n = 3 energy level, the number of possible orbitals is
A)
1 done
clear
B)
3 done
clear
C)
4 done
clear
D)
9 done
clear
View Solution play_arrow
-
question_answer55)
Penetration power of proton is
A)
more than electron done
clear
B)
less than electron done
clear
C)
more than neutron done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer56)
\[_{18}A{{r}^{40}}\] and \[_{19}{{K}^{40}}\] is called
A)
Isomers done
clear
B)
Isobars done
clear
C)
Isotopes done
clear
D)
Isotones done
clear
View Solution play_arrow
-
question_answer57)
Number of neutrons in heavy hydrogen atom is
A)
0 done
clear
B)
1 done
clear
C)
2 done
clear
D)
3 done
clear
View Solution play_arrow
-
question_answer58)
For an atom of a given element, the number of electrons are equal to
A)
The number of protons + number of neutrons done
clear
B)
The atomic number of the element done
clear
C)
The number of proton-number of neutrons done
clear
D)
The-mass number of the element done
clear
View Solution play_arrow
-
question_answer59)
"The exact path of electron in 2p-orbital can not be determined", This statement is based upon
A)
Hund's rule done
clear
B)
Bohr's rule done
clear
C)
Uncertainly principal done
clear
D)
Aufbau principal done
clear
View Solution play_arrow
-
question_answer60)
Maximum number of \[4f\] electrons that can have \[s=-\frac{1}{2}\] values, is
A)
14 done
clear
B)
7 done
clear
C)
5 done
clear
D)
10 done
clear
View Solution play_arrow
-
question_answer61)
The radius of the atom is of the order of
A)
\[\text{1}{{0}^{-\text{1}0}}\text{ cm}\] done
clear
B)
\[\text{1}{{0}^{-\text{13}}}\text{ cm}\] done
clear
C)
\[\text{1}{{0}^{-\text{15}}}\text{ cm}\] done
clear
D)
\[\text{1}{{0}^{-\text{8}}}\text{ cm}\] done
clear
View Solution play_arrow
-
question_answer62)
Total energy of an electron in an atom is
A)
less than zero done
clear
B)
more than zero done
clear
C)
may be less or more than zero done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer63)
Remaining part of atom except outer orbit is called
A)
kernel done
clear
B)
core done
clear
C)
empty done
clear
D)
none of these done
clear
View Solution play_arrow
-
question_answer64)
Maximum number of electrons in a subshell is given by
A)
\[(2l+1)\] done
clear
B)
\[2(2l+1)\] done
clear
C)
\[{{(2l+1)}^{2}}\] done
clear
D)
\[2{{(2l+1)}^{2}}\] done
clear
View Solution play_arrow
-
question_answer65)
Two electrons occupying an orbital are distinguished by
A)
principal quantum number done
clear
B)
angular momentum quantum number done
clear
C)
magnetic quantum number done
clear
D)
spin quantum number done
clear
View Solution play_arrow
-
question_answer66)
The number of unpaired electrons in a chromic conclusion \[C{{r}^{+3}}\] (atomic number 24) is
A)
6 done
clear
B)
4 done
clear
C)
3 done
clear
D)
1 done
clear
View Solution play_arrow
-
question_answer67)
Rutherford's scattering experiment led to the conclusion that?
A)
nucleus is very small in size done
clear
B)
nucleus is very heavy and carries positive charge done
clear
C)
most of the space in the atom is empty done
clear
D)
all of these done
clear
View Solution play_arrow
-
question_answer68)
The example of a pair of isobar is
A)
\[_{1}{{\text{H}}^{1}}\,\text{and}{{\,}_{1}}{{H}^{2}}\] done
clear
B)
\[_{1}{{\text{H}}^{2}}\,\text{and}{{\,}_{1}}{{H}^{3}}\] done
clear
C)
\[_{1}{{\text{H}}^{3}}\,\text{and}{{\,}_{2}}H{{e}^{3}}\] done
clear
D)
\[_{1}\text{H}{{\text{e}}^{3}}\,\text{and}{{\,}_{1}}H{{e}^{4}}\] done
clear
View Solution play_arrow
-
question_answer69)
The number of electrons in 3d shell for element with Atomic number 26 is
A)
4 done
clear
B)
6 done
clear
C)
8 done
clear
D)
10 done
clear
View Solution play_arrow
-
question_answer70)
Nuclei of isotopes differ in
A)
number of protons done
clear
B)
number of neutrons done
clear
C)
number of neutrons and protons both done
clear
D)
none of these done
clear
View Solution play_arrow
![]()
