-
question_answer1)
What kind of solution is milk?
A)
Suspension done
clear
B)
Colloid done
clear
C)
True solution done
clear
D)
Alloy done
clear
View Solution play_arrow
-
question_answer2)
A liquid and a solid are found together in a single phase. What is this known as?
A)
Solution done
clear
B)
Solute done
clear
C)
Solvent done
clear
D)
Emulsion done
clear
View Solution play_arrow
-
question_answer3)
Which of the following is a homogeneous solution?
A)
Muddy water done
clear
B)
Bread done
clear
C)
Concrete done
clear
D)
A solution of sugar in water done
clear
View Solution play_arrow
-
question_answer4)
What kind of a colloidal solution is an emulsion?
A)
Solid dispersed in solid. done
clear
B)
Liquid dispersed in liquid. done
clear
C)
Gas dispersed in liquid. done
clear
D)
Liquid dispersed in solid. done
clear
View Solution play_arrow
-
question_answer5)
In which of the following does scattering of light takes place?
A)
Electrolytic solutions done
clear
B)
Colloidal solutions done
clear
C)
Electro dialysis done
clear
D)
Alcoholic solutions done
clear
View Solution play_arrow
-
question_answer6)
Which of the following forms a colloidal solution in water?
A)
Salt done
clear
B)
Glucose done
clear
C)
Starch done
clear
D)
Barium nitrate done
clear
View Solution play_arrow
-
question_answer7)
Which of the following is NOT a physical change?
A)
Tearing a paper done
clear
B)
Bending an iron rod done
clear
C)
Freezing water into ice done
clear
D)
Curdling of milk done
clear
View Solution play_arrow
-
question_answer8)
In which of the following is Tyndall effect observed?
A)
Solution done
clear
B)
Precipitate done
clear
C)
Sol done
clear
D)
Vapour done
clear
View Solution play_arrow
-
question_answer9)
How is Brownian motion caused?
A)
Through temperature fluctuations within the liquid phase done
clear
B)
Through attraction and repulsion between the charges on the colloidal particles done
clear
C)
Through collision of molecules between the colloidal particles done
clear
D)
Through pressure variations within the liquid phase done
clear
View Solution play_arrow
-
question_answer10)
To which of the following category does ice-cream belong?
A)
Solution done
clear
B)
Emulsion done
clear
C)
Element done
clear
D)
Suspension done
clear
View Solution play_arrow
-
question_answer11)
What can be observed when sunlight is passed through a colloidal solution?
A)
Adsorption done
clear
B)
Coagulation done
clear
C)
Tyndall effect done
clear
D)
Brownian motion done
clear
View Solution play_arrow
-
question_answer12)
What is a sol?
A)
A solid dispersed in a liquid done
clear
B)
A liquid dispersed in a gas done
clear
C)
A gas dispersed in a liquid done
clear
D)
A gas dispersed in a solid done
clear
View Solution play_arrow
-
question_answer13)
In both dialysis and osmosis which particles do not pass through the semipermeable membrane?
A)
Water done
clear
B)
Small molecules done
clear
C)
Colloids done
clear
D)
All of the above done
clear
View Solution play_arrow
-
question_answer14)
What is the name of the process for the separation of colloidal panicles from those of crystalloids called?
A)
Dialysis done
clear
B)
Pyrolysis done
clear
C)
Peptization done
clear
D)
Photolysis done
clear
View Solution play_arrow
-
question_answer15)
Shaving cream produces foam. What kind of colloid is shaving cream?
A)
Liquid dispersed in a gas done
clear
B)
Gas dispersed in a liquid done
clear
C)
Solid dispersed in a liquid done
clear
D)
Solid dispersed in a gas done
clear
View Solution play_arrow
-
question_answer16)
What kind of solution is drinking soda?
A)
Gas in liquid done
clear
B)
Liquid in gas done
clear
C)
Gas in gas done
clear
D)
Solid in liquid done
clear
View Solution play_arrow
-
question_answer17)
What kind of solution is amalgam?
A)
Solid in solid done
clear
B)
Solid in mercury done
clear
C)
Liquid in solid done
clear
D)
Liquid in liquid done
clear
View Solution play_arrow
-
question_answer18)
Which of the following is a true solution?
A)
\[NaCl\] in sulphur dioxide. done
clear
B)
Copper in silver. done
clear
C)
Salt in petrol. done
clear
D)
Mud in water. done
clear
View Solution play_arrow
-
question_answer19)
Which of the following is a chemical change?
A)
Boiling of eggs. done
clear
B)
Evaporation of water. done
clear
C)
Precipitation of snow. done
clear
D)
Melting of wax. done
clear
View Solution play_arrow
-
question_answer20)
What are the materials which contain at least two pure substances and show the properties of their constituents called?
A)
A compound done
clear
B)
An element done
clear
C)
A mixture done
clear
D)
A solution done
clear
View Solution play_arrow
-
question_answer21)
Which of the following is a solid-in-solid colloid?
A)
Shaving cream done
clear
B)
Milk of magnesia done
clear
C)
Milky glass done
clear
D)
Cheese done
clear
View Solution play_arrow
-
question_answer22)
What is a solution of iodine in carbon tetrachloride called?
A)
Aqueous solution done
clear
B)
Alcoholic solution done
clear
C)
Non-aqueous solution done
clear
D)
Tincture of iodine done
clear
View Solution play_arrow
-
question_answer23)
Which of the following is a characteristic of both mixtures and compounds?
A)
They contain components in fixed proportions. done
clear
B)
Their properties are the same as those of their components. done
clear
C)
Their weight is equal to the sum of the weights of their components. done
clear
D)
Energy is given out when they are being prepared. done
clear
View Solution play_arrow
-
question_answer24)
What happens when salt dissolved in water is heated?
A)
There is an increase in the boiling point. done
clear
B)
There is no change in the boiling point. done
clear
C)
There is a decrease in the boiling point. done
clear
D)
All of the above done
clear
View Solution play_arrow
-
question_answer25)
Which of the following is NOT a compound?
A)
Sugar done
clear
B)
Common salt done
clear
C)
Diamond done
clear
D)
Plaster of Paris done
clear
View Solution play_arrow
-
question_answer26)
Which of the following is an example of a mixture?
A)
Sugar done
clear
B)
Brass done
clear
C)
\[C{{O}_{2}}\] done
clear
D)
\[N{{O}_{2}}\] done
clear
View Solution play_arrow
-
question_answer27)
Which of the following obey the law of constant proportions in their formation?
A)
Mixtures done
clear
B)
Compounds done
clear
C)
Elements done
clear
D)
Colloids done
clear
View Solution play_arrow
-
question_answer28)
By which process the drugs from the blood are separated?
A)
Distillation done
clear
B)
Chromatography done
clear
C)
Sublimation done
clear
D)
Separating funnel done
clear
View Solution play_arrow
-
question_answer29)
How can sugar from sugar solution be separated?
A)
By distillation done
clear
B)
By evaporation done
clear
C)
By sublimation done
clear
D)
By filtration done
clear
View Solution play_arrow
-
question_answer30)
Which of the following processes of separating substances involves both evaporation and condensation?
A)
Filtration done
clear
B)
Crystallisation done
clear
C)
Distillation done
clear
D)
Sedimentation done
clear
View Solution play_arrow
-
question_answer31)
Which of the following processes is useful to collect pure water from a solution of sugar and water or salt and water?
A)
Filtration done
clear
B)
Evaporation done
clear
C)
Distillation done
clear
D)
Crystallisation done
clear
View Solution play_arrow
-
question_answer32)
Which of the following processes can be useful in separating the salt from a mixture of sand and salt?
A)
Dissolution done
clear
B)
Filtration done
clear
C)
Evaporation done
clear
D)
All of the above done
clear
View Solution play_arrow
-
question_answer33)
What is the process involved in making candy?
A)
Filtration done
clear
B)
Crystallisation done
clear
C)
Sedimentation done
clear
D)
Centrifugation done
clear
View Solution play_arrow
-
question_answer34)
By which method can ammonium chloride be separated from sand?
A)
By hand picking done
clear
B)
By filtration done
clear
C)
By sublimation done
clear
D)
By diffusion done
clear
View Solution play_arrow
-
question_answer35)
By which process can we separate camphor from ammonium chloride?
A)
Filtration done
clear
B)
Sublimation done
clear
C)
Dissolution and evaporation done
clear
D)
Distillation done
clear
View Solution play_arrow
-
question_answer36)
Which of the following apparatus is used to separate a mixture of immiscible liquids?
A)
Centrifuge done
clear
B)
Condenser done
clear
C)
Separating funnel done
clear
D)
Distillation flask done
clear
View Solution play_arrow
-
question_answer37)
What is the technique used to separate dirt particles from clothes in a washing machine?
A)
Magnetic separation done
clear
B)
Filtration done
clear
C)
Evaporation done
clear
D)
Centrifugation done
clear
View Solution play_arrow
-
question_answer38)
What kind of change is undergone when an electric bulb glows
A)
A physical change done
clear
B)
A chemical change done
clear
C)
Both a physical and a chemical change done
clear
D)
A permanent change done
clear
View Solution play_arrow
-
question_answer39)
Both cooking oil and water are liquids at room temperature. By which of the given characteristics are they different chemically?
A)
Same colour and flammability done
clear
B)
Odour and in flammability done
clear
C)
Solubility and similar taste done
clear
D)
Odour and flammability done
clear
View Solution play_arrow
-
question_answer40)
What type of mixture is steel?
A)
Solid-liquid heterogeneous mixture done
clear
B)
Solid-solid heterogeneous mixture done
clear
C)
Solid-solid homogeneous mixture done
clear
D)
Pure substance done
clear
View Solution play_arrow
-
question_answer41)
Two substances \[P\] and \[Q\] when brought together, form substance \[R\] with the evolution of heat. The properties of \[R\] are different from both \[P\] and\[Q\]. What is substance\[R\]?
A)
A compound done
clear
B)
An element done
clear
C)
A metal done
clear
D)
A mixture done
clear
View Solution play_arrow
-
question_answer42)
Which of the following pairs of colloidal solutions have dispersed phase as liquid and dispersing medium as gas?
A)
Fog, mist done
clear
B)
Butter, milk done
clear
C)
Fog, smoke done
clear
D)
Smoke, foam done
clear
View Solution play_arrow
-
question_answer43)
Which of the following is a true solution?
A)
Copper in gold done
clear
B)
Sulphur in water done
clear
C)
Milk done
clear
D)
\[KCl\] in sulphur dioxide done
clear
View Solution play_arrow
-
question_answer44)
Identify a pure substance from the following?
A)
Steel done
clear
B)
Magnalium done
clear
C)
Ammonia done
clear
D)
Gun powder done
clear
View Solution play_arrow
-
question_answer45)
What is the smallest constituent of matter that retains its chemical identity?
A)
Atom done
clear
B)
Molecule done
clear
C)
Ion done
clear
D)
Radical done
clear
View Solution play_arrow
-
question_answer46)
Which of the following solutions shows Tyndall effect?
A)
A solution of common salt done
clear
B)
A solution of sodium carbonate done
clear
C)
Starch solution done
clear
D)
Sugar solution done
clear
View Solution play_arrow
-
question_answer47)
The size of a colloidal particle is
A)
\[{{10}^{-1}}\]to\[{{10}^{-3}}cm\] done
clear
B)
\[{{10}^{-5}}\]to\[{{10}^{-7}}cm\] done
clear
C)
\[{{10}^{-8}}\]to\[{{10}^{-5}}cm\] done
clear
D)
\[{{10}^{-6}}\]to\[{{10}^{-8}}cm\] done
clear
View Solution play_arrow
-
question_answer48)
Which of these statements is/are true?
A)
The components of a suspension can be separated by filtration. done
clear
B)
The particles of a colloid can pass through a filter paper. done
clear
C)
The constituents of a compound can be separated easily. done
clear
D)
Both [a] and [b] done
clear
View Solution play_arrow
-
question_answer49)
A student added \[10\,\,ml-12\,\,ml\] of Conc. \[{{H}_{2}}S{{O}_{4}}\]in a test tube containing \[5\,\,g-6\,\,g\] of sugar crystals and warmed it gently. He noticed that white crystals slowly turned brown and became a black mass. What happened in this change?
A)
\[{{H}_{2}}S{{O}_{4}}\] turned black but no change took place. done
clear
B)
Sugar got charred by the action of hot. conc. \[{{H}_{2}}S{{O}_{4}}\]and a chemical change took place. done
clear
C)
It is only a physical change. done
clear
D)
It is an irreversible physical change. done
clear
View Solution play_arrow
-
question_answer50)
A dispersed phase particle in a zig-zag movement is shown below.
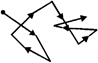
What kind of motion is exhibited by the particle?
A)
Transitional motion done
clear
B)
Circular motion done
clear
C)
Linear motion done
clear
D)
Brownian motion done
clear
View Solution play_arrow
