-
question_answer1)
In the given figure, electrolyte, anode and cathode respectively are
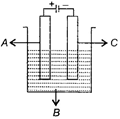
A)
A, B and C done
clear
B)
B, A and C done
clear
C)
B, C and A done
clear
D)
A, C and B done
clear
View Solution play_arrow
-
question_answer2)
In which of the following will the bulb glow?
A)
B)
C)
D)
View Solution play_arrow
-
question_answer3)
In which one of the following situations does a conventional electric current flow due north?
A)
Protons in a beam are moving due south. done
clear
B)
A water molecule is moving due north. done
clear
C)
Electrons in a beam are moving due south. done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer4)
The diagram shows a beaker containing a solution of zinc sulphate and two carbon electrodes. A battery is placed next to it. In order that the electrode P be plated with zinc,
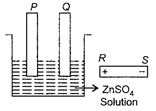
A)
P must be connected to S and Q to R done
clear
B)
P must be connected to Q and S to R done
clear
C)
P must be connected to R and Q to S done
clear
D)
P and Q must be connected to R. done
clear
View Solution play_arrow
-
question_answer5)
In the experiment, if A is plated with copper, the anode would be
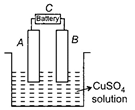
A)
A done
clear
B)
B done
clear
C)
C done
clear
D)
Data insufficient done
clear
View Solution play_arrow
-
question_answer6)
When electricity is passed through calcium nitrate solution in water, what is likely to be formed at the cathode and why?
A)
Hydrogen because it is a very reactive than calcium done
clear
B)
Calcium because it is a very reactive than hydrogen done
clear
C)
Hydrogen because it is less reactive than calcium done
clear
D)
Calcium because it is less reactive than hydrogen. done
clear
View Solution play_arrow
-
question_answer7)
An electric current is passed through a conducting solution. Following are the some observations:
| (i) Deposits of metal may be seen on electrodes |
| (ii) Solution may get heated |
| (iii) Bubbles of gas may be formed on the electrodes |
A)
Only (ii) is true. done
clear
B)
Only (i) and (ii) are true. done
clear
C)
Only (i) and (iii) are true. done
clear
D)
All (i), (ii) and (iii) are true. done
clear
View Solution play_arrow
-
question_answer8)
In the given figure, the filter paper contains starch soaked in potassium iodide solution is connected to the battery as shown. When we write on the wet paper, it has a blue black colour.
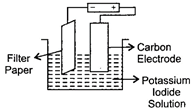
| (i) Iodine is formed at the anode. |
| (ii) Iodine turns blue black in the presence of starch. |
A)
Only (i) is correct. done
clear
B)
Only (ii) is correct. done
clear
C)
Both (i) and (ii) are correct. done
clear
D)
Neither (i) nor (ii) is correct. done
clear
View Solution play_arrow
-
question_answer9)
Which of the following statements is incorrect?
A)
Anode is an electrode connected to the positive terminal of the battery. done
clear
B)
Pure water is poor conductor of electricity. done
clear
C)
Electrolysis is used in the refining of impure metals. done
clear
D)
Ions can have only a positive charge. done
clear
View Solution play_arrow
-
question_answer10)
Which of the following is correct?
A)
Water can be used for extinguishing fires caused due to electrical faults. done
clear
B)
Carbon is a non-metal so it cannot be used in electrolytic cell. done
clear
C)
A liquid conducts electricity because of the presence of ions. done
clear
D)
Pure water forms ions to conduct electricity. done
clear
View Solution play_arrow
-
question_answer11)
Which of the following cells use(s) electric current to produce a chemical reaction?
A)
Dry cell done
clear
B)
Solar cell done
clear
C)
Electrolytic cell done
clear
D)
Both dry cell and electrolytic cell done
clear
View Solution play_arrow
-
question_answer12)
Electrolysis of a solution of sodium chloride produces
A)
Hydrogen gas at anode and chlorine at cathode done
clear
B)
Hydrogen gas at cathode, chlorine at anode and sodium hydroxide done
clear
C)
Hydrogen gas at anode and sodium hydroxide at cathode done
clear
D)
Chlorine gas at anode and sodium hydroxide at cathode. done
clear
View Solution play_arrow
-
question_answer13)
Adding common salt to distilled water makes it
A)
Good conductor done
clear
B)
Insulator done
clear
C)
Can't say done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer14)
Why is the switch for any electrical appliance in ac circuit always connected to the live wire?
A)
No current flows in the neutral wire. done
clear
B)
There will be a short circuit if the switch is in the Earth lead. done
clear
C)
The device can be switched off easily if the switch is in the neutral lead. done
clear
D)
The device can be isolated (mode safe) if the switch is in the live lead. done
clear
View Solution play_arrow
-
question_answer15)
To protect iron from corrosion and rust, it is coated with
A)
Tin done
clear
B)
Copper done
clear
C)
Mercury done
clear
D)
Zinc. done
clear
View Solution play_arrow
-
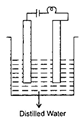
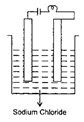
question_answer16)
There are two different solutions in set up P and Q as shown in figure. The bulb in the set up P glows more brightly as compared to that of the set up Q. What are the possible causes for this?
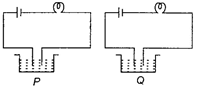
| (i) The connections of the circuit Q may be loose. |
| (ii) The liquid in Q may have small conductivity. |
| (iii) Liquid in P is equivalent to a battery while liquid in Q is equivalent to a cell of the battery of liquid P. |
A)
Only (i) and (ii) done
clear
B)
Only (ii) and (iii) done
clear
C)
Only (i) done
clear
D)
Only (ii) done
clear
View Solution play_arrow
-
question_answer17)
| Direction: Four substances were tested for their electrical conductivity. The results are shown in the table. Study the table and answer the following questions. |
| Substance | Bulb | Substance at Cathode | Substance at Anode |
| P | Lights up | Aluminium | Oxygen |
| Q | Lights up | Nothing | Nothing |
| R | Does not light up | Nothing | Nothing |
| S | Lights up | Hydrogen | Carbon dioxide |
In substance Q, bulb lights up but no substance is found either at cathode or at anode, why?
A)
Because Q is non-electrolyte. done
clear
B)
Because Q is an element which remains unchanged when it conducts electricity. done
clear
C)
Because Q is an electrolyte that conducts electricity when it is in the molten state. done
clear
D)
Because Q is a poor conductor of electricity. done
clear
View Solution play_arrow
-
question_answer18)
| Direction: Four substances were tested for their electrical conductivity. The results are shown in the table. Study the table and answer the following questions. |
| Substance | Bulb | Substance at Cathode | Substance at Anode |
| P | Lights up | Aluminium | Oxygen |
| Q | Lights up | Nothing | Nothing |
| R | Does not light up | Nothing | Nothing |
| S | Lights up | Hydrogen | Carbon dioxide |
Which solutions could be alumina and magnesium nitrate respectively?
A)
P and Q done
clear
B)
R and Q done
clear
C)
P and S done
clear
D)
P is the alumina but no one is magnesium nitrate. done
clear
View Solution play_arrow
-
question_answer19)
The bumpers and door handles of motor cars, taps etc. are coated with silvery layer of chromium because
| (i) It does not easily get corroded. |
| (ii) It forms a hard layer which does not easily get scratched. |
| (iii) It tarnishes quickly. |
| (iv) It is cheap. |
Which of the following options is correctly identifies true (T) or false (F)?
A)
B)
C)
D)
View Solution play_arrow
-
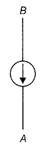
question_answer20)
The diagram shows a plotting compass placed above a copper wire when there is no current flowing in the wire. What happens to the compass needle when a large direct current flows from point A to point B?
A)
The compass needle points towards the right. done
clear
B)
The compass needle points towards the left. done
clear
C)
The compass needle points towards upward. done
clear
D)
The compass needle points toward downward. done
clear
View Solution play_arrow

 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear

