-
question_answer1)
| Directions : (1 - 15) |
| For question numbers 1-15, a statement of assertion followed by a statement of reason is given. Choose the correct answer out of the following choices. |
Assertion: 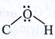 bond angle is less than the normal tetrahedral bond angle. bond angle is less than the normal tetrahedral bond angle. |
| Reason: Lone pair-lone pair repulsion decreases bond angle. |
A)
Assertion and reason both are correct statements and reason is correct explanation for assertion. done
clear
B)
Assertion and reason both are correct statements but reason is not correct explanation for assertion. done
clear
C)
Assertion is correct statement but reason is wrong statement. done
clear
D)
Assertion is wrong statement but reason is correct statement. done
clear
View Solution play_arrow
-
question_answer2)
| Assertion : o-Nitrophenol is more volatile than p-nitrophenol. |
| Reason: Intramolecular hydrogen bonding is present in o-nitrophenol while intermolecular H-bonding is injo-nitrophenol. |
A)
Assertion and reason both are correct statements and reason is correct explanation for assertion. done
clear
B)
Assertion and reason both are correct statements but reason is not correct explanation for assertion. done
clear
C)
Assertion is correct statement but reason is wrong statement. done
clear
D)
Assertion is wrong statement but reason is correct statement. done
clear
View Solution play_arrow
-
question_answer3)
| Assertion : \[C{{H}_{3}}OC{{H}_{3}}\] and \[{{C}_{2}}{{H}_{5}}OH\] has comparable molecular weight but boiling point of \[{{C}_{2}}{{H}_{5}}OH\] is more than dimethyl ether. |
| Reason: \[{{C}_{2}}{{H}_{5}}OH\] forms intermolecular H-bonding while \[C{{H}_{3}}OC{{H}_{3}}\] forms intramolecular H-bonding. |
A)
Assertion and reason both are correct statements and reason is correct explanation for assertion. done
clear
B)
Assertion and reason both are correct statements but reason is not correct explanation for assertion. done
clear
C)
Assertion is correct statement but reason is wrong statement. done
clear
D)
Assertion is wrong statement but reason is correct statement. done
clear
View Solution play_arrow
-
question_answer4)
| Assertion : With \[B{{r}_{2}}-{{H}_{2}}O\], phenol gives 2,4,6-tribromophenol but with \[B{{r}_{2}}-C{{S}_{2}}\], it gives 4-bromophenol as the major product. |
| Reason : In water, ionisation of phenol is enhanced but in \[C{{S}_{2}}\], it is greatly suppressed. |
A)
Assertion and reason both are correct statements and reason is correct explanation for assertion. done
clear
B)
Assertion and reason both are correct statements but reason is not correct explanation for assertion. done
clear
C)
Assertion is correct statement but reason is wrong statement. done
clear
D)
Assertion is wrong statement but reason is correct statement. done
clear
View Solution play_arrow
-
question_answer5)
| Assertion : Primary and secondary alcohols can be distinguished by Victor-Meyer's test. |
| Reason : Primary alcohols form nitrolic acid which dissolves in NaOH to form blood red colouration but secondary alcohols form pseudonitrols which give blue colouration with NaOH. |
A)
Assertion and reason both are correct statements and reason is correct explanation for assertion. done
clear
B)
Assertion and reason both are correct statements but reason is not correct explanation for assertion. done
clear
C)
Assertion is correct statement but reason is wrong statement. done
clear
D)
Assertion is wrong statement but reason is correct statement. done
clear
View Solution play_arrow
-
question_answer6)
| Assertion : Reimer-Tiemann reaction of phenol with \[CHC{{l}_{3}}\] in NaOH at 340 K gives salicylic acid as the major product. |
| Reason : The reaction occurs through intermediate formation of \[^{+}CHC{{l}_{2}}\] |
A)
Assertion and reason both are correct statements and reason is correct explanation for assertion. done
clear
B)
Assertion and reason both are correct statements but reason is not correct explanation for assertion. done
clear
C)
Assertion is correct statement but reason is wrong statement. done
clear
D)
Assertion is wrong statement but reason is correct statement. done
clear
View Solution play_arrow
-
question_answer7)
| Assertion : Lucas test can be used to distinguish between 1-propanol and 2-propanol. |
| Reason : Lucas test is based upon the difference in reactivity of primary, secondary and tertiary alcohols with cone. HCl and anhyd. \[ZnC{{l}_{2}}\]. |
A)
Assertion and reason both are correct statements and reason is correct explanation for assertion. done
clear
B)
Assertion and reason both are correct statements but reason is not correct explanation for assertion. done
clear
C)
Assertion is correct statement but reason is wrong statement. done
clear
D)
Assertion is wrong statement but reason is correct statement. done
clear
View Solution play_arrow
-
question_answer8)
| Assertion: Among n-butane, ethoxyethane, 1-propanol and 2-propanol, the increasing order of boiling points is, 1-butanol < 1-propanol < ethoxyzethane < n-butane. |
| Reason: Boiling point increases with increase in molecular mass. |
A)
Assertion and reason both are correct statements and reason is correct explanation for assertion. done
clear
B)
Assertion and reason both are correct statements but reason is not correct explanation for assertion. done
clear
C)
Assertion is correct statement but reason is wrong statement. done
clear
D)
Assertion is wrong statement but reason is correct statement. done
clear
View Solution play_arrow
-
question_answer9)
| Assertion : Phenol is less acidic than p-nitrophenol. |
| Reason : Phenolate ion is more stable than p-nitrophenolate ion. |
A)
Assertion and reason both are correct statements and reason is correct explanation for assertion. done
clear
B)
Assertion and reason both are correct statements but reason is not correct explanation for assertion. done
clear
C)
Assertion is correct statement but reason is wrong statement. done
clear
D)
Assertion is wrong statement but reason is correct statement. done
clear
View Solution play_arrow
-
question_answer10)
| Assertion : tert-Butyl alcohol undergoes acid catalysed dehydration readily than propanol. |
| Reason : \[3{}^\circ \]Alcohols do not give Victor-Meyer's test. |
A)
Assertion and reason both are correct statements and reason is correct explanation for assertion. done
clear
B)
Assertion and reason both are correct statements but reason is not correct explanation for assertion. done
clear
C)
Assertion is correct statement but reason is wrong statement. done
clear
D)
Assertion is wrong statement but reason is correct statement. done
clear
View Solution play_arrow
-
question_answer11)
| Assertion : Dimethyl ether and diethyl ether are soluble in water. |
| Reason : As the molecular mass increases, solubility of ethers in water decreases. |
A)
Assertion and reason both are correct statements and reason is correct explanation for assertion. done
clear
B)
Assertion and reason both are correct statements but reason is not correct explanation for assertion. done
clear
C)
Assertion is correct statement but reason is wrong statement. done
clear
D)
Assertion is wrong statement but reason is correct statement. done
clear
View Solution play_arrow
-
question_answer12)
| Assertion : Phenol decomposes \[NaHC{{O}_{3}}\] solution to evolve \[C{{O}_{2}}\] gas. |
| Reason : Picric acid is 2, 4, 6-trinitrophenol. |
A)
Assertion and reason both are correct statements and reason is correct explanation for assertion. done
clear
B)
Assertion and reason both are correct statements but reason is not correct explanation for assertion. done
clear
C)
Assertion is correct statement but reason is wrong statement. done
clear
D)
Assertion is wrong statement but reason is correct statement. done
clear
View Solution play_arrow
-
question_answer13)
| Assertion : Equimolar mixture of cone. HCl and anhydrous \[ZnC{{l}_{2}}\] is called Lucas reagent. |
| Reason: Lucas reagent can be used to distinguish between methanol and ethanol. |
A)
Assertion and reason both are correct statements and reason is correct explanation for assertion. done
clear
B)
Assertion and reason both are correct statements but reason is not correct explanation for assertion. done
clear
C)
Assertion is correct statement but reason is wrong statement. done
clear
D)
Assertion is wrong statement but reason is correct statement. done
clear
View Solution play_arrow
-
question_answer14)
| Assertion : Amongst the compounds, |
| \[{{H}_{2}}C=CHC{{H}_{2}}OH\](I), \[{{C}_{6}}{{H}_{5}}OH\](II) |
| \[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}OH\](III), \[{{\left( C{{H}_{3}} \right)}_{3}}COH\](IV) |
| only (IV) reacts with Lucas reagent at room temperature. |
| Reason : Tertiary alcohol gives turbidity immediately with Lucas reagent. |
A)
Assertion and reason both are correct statements and reason is correct explanation for assertion. done
clear
B)
Assertion and reason both are correct statements but reason is not correct explanation for assertion. done
clear
C)
Assertion is correct statement but reason is wrong statement. done
clear
D)
Assertion is wrong statement but reason is correct statement. done
clear
View Solution play_arrow
-
question_answer15)
| Assertion : Boiling points of alcohols are lower than hydrocarbons. |
| Reason : Among isomeric alcohols, boiling point decreases in the order :\[1{}^\circ \text{ }>\text{ }2{}^\circ \text{ }>\text{ }3{}^\circ \]. |
A)
Assertion and reason both are correct statements and reason is correct explanation for assertion. done
clear
B)
Assertion and reason both are correct statements but reason is not correct explanation for assertion. done
clear
C)
Assertion is correct statement but reason is wrong statement. done
clear
D)
Assertion is wrong statement but reason is correct statement. done
clear
View Solution play_arrow
