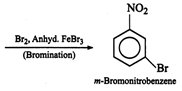
Answer:
(i) Electrophilic addition occurs
through the more stable carbocation intermediate
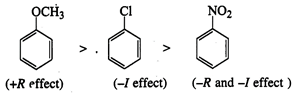
 (ii) Out of\[-O-\] and \[C=O\] groups, O
is electron-donating and o, p-directing. Therefore, it increases electron density
in the benzene ring directly attached to it. As a result, electrophilic
substitution occurs in this ring predominantly at the less hindered p-position
(ii) Out of\[-O-\] and \[C=O\] groups, O
is electron-donating and o, p-directing. Therefore, it increases electron density
in the benzene ring directly attached to it. As a result, electrophilic
substitution occurs in this ring predominantly at the less hindered p-position
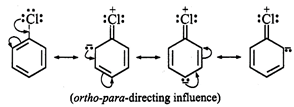 In contrast, the C = O group is
electron withdrawing and m-directiong. Since, it decreases the electron-density
in the ring to which it is attached, therefore, substitution does not occur in
this ring.
In contrast, the C = O group is
electron withdrawing and m-directiong. Since, it decreases the electron-density
in the ring to which it is attached, therefore, substitution does not occur in
this ring.
You need to login to perform this action.
You will be redirected in
3 sec
