-
question_answer1)
Study the given table carefully.
| |
Compounds |
Colour change on adding China rose indicator |
| (i) |
Hydroxide of group 2 element |
Magenta |
| (ii) |
Oxide of element (group 16 and period 3) |
Magenta |
| (iii) |
Oxide of element (electronic configuration 2, 4) |
Magenta |
| (IV) |
Hydroxide of element (at. no. 19) |
Green |
| (V) |
Lime water |
Magenta |
Which of the observations are correctly matched?
A)
(i), (ii) and (iv) only done
clear
B)
(ii), (iii) and (iv) only done
clear
C)
(ii), (iii) and (v) only done
clear
D)
(i), (iii) and (iv) only done
clear
View Solution play_arrow
-
question_answer2)
Which of the elements having following positions, have same number of valence electrons as that of element with atomic number 6?
| (i) Group 14 and period 5 |
| (ii) Group 14 and period 4 |
| (iii) Group 6 and period 4 |
| (iv) Group 14 and period 3 |
A)
(i) and (ii) only done
clear
B)
(i), (ii) and (iii) only done
clear
C)
(i), (ii) and (iv) only done
clear
D)
All of these done
clear
View Solution play_arrow
-
question_answer3)
|
Position of five elements P, Q, R, S and T is shown on the simplified form of the periodic table.
|
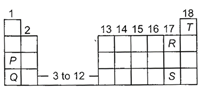
|
|
The element which has maximum tendency to lose electron, the element which has maximum tendency to gain electrons and the element which has no tendency to gain or lose electrons are respectively
|
A)
P, R and S done
clear
B)
S, R and T done
clear
C)
Q, S and P done
clear
D)
Q, R and T. done
clear
View Solution play_arrow
-
question_answer4)
Which of the following statements are incorrect?
| I. The atomic radii increase along a period from left to right. |
| II. In periodic table isotopes are given separate places. |
| III. The oxides of metals are basic in nature. |
| IV. Na, Mg and Al belong to the same period. |
A)
I and IV only done
clear
B)
II and IV only done
clear
C)
I and II only done
clear
D)
III and IV only done
clear
View Solution play_arrow
-
question_answer5)
Following elements are present in the second period of the periodic table from left to right. Li, Be, B, C, N, O, F, Ne Few statements are given about these elements:
| (i) O and F exist as a monoatomic gases |
| (ii) is the lightest element in the period |
| (iii) B is a metalloid while C is a metal. |
| (iv) Li and F react to form a compound LiF. |
The correct statements are
A)
(i) and (iii) only done
clear
B)
(ii) and (iv)only done
clear
C)
(ii) and (iii) only done
clear
D)
(i) and (iv) only done
clear
View Solution play_arrow
-
question_answer6)
Read the given statements and mark the correct option.
| Statement 1: According to Dobereiner, when the three similar elements are arranged in the order of their increasing atomic weights the weight of the middle element is equal to the arithmetic mean of the other two elements. |
| Statement 2: Atomic mass unit is equal to one twelfth of mass of a carbon atom. |
A)
Both statements 1 and 2 are true and statement 2 is the correct explanation of statement 1. done
clear
B)
Both statements 1 and 2 are true but statement 2 is not the correct explanation of statement 1. done
clear
C)
Statement 1 is true and statement 2 is false. done
clear
D)
Both statements 1 and 2 are false. done
clear
View Solution play_arrow
-
question_answer7)
The ionization energies of elements U, V, W and X are 2480, 550, 950 and 1690 kJ mol-1 respectively. Which of the statements about these elements are correct?
| (i) V is a reactive metal. |
| (ii) X is a reactive non-metal. |
| (iii) U is a noble gas. |
A)
(i) and (ii) only done
clear
B)
(ii) and (iii) only done
clear
C)
(i) and (iii) only done
clear
D)
All of these done
clear
View Solution play_arrow
-
question_answer8)
Three elements P, Q and R have consecutive increasing atomic numbers. If Q is a noble gas then which of the following is correct about P and R?
A)
P and R belong to the same period of the periodic table. done
clear
B)
P is a non-metal while R is a metal done
clear
C)
P and R belong to same group of the periodic table. done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer9)
Which of the following properties cannot be predicted from the position of the elements in the periodic table?
A)
Nature of oxide done
clear
B)
Formula of a particular compound done
clear
C)
Total number of isotopes done
clear
D)
Oxidizing or reducing nature done
clear
View Solution play_arrow
-
question_answer10)
To study the chemical properties, Mendeleev concentrated on the compounds formed by different elements with X and Y. Identify X and Y.
A)
B)
C)
D)
View Solution play_arrow
-
question_answer11)
Match column I with column II and mark the correct option from the codes given below.
| Column I |
Column II |
| (a) Highest electronegativity |
(i) Caesium |
| (b) Highest electron affinity |
(ii) Ruthennium |
| (c) Highest electropositivity |
(iii) Fluorine |
| (d) Highest oxidation number |
(iv) Chlorine |
A)
(a) - (i), (b) - (ii), (c) - (iii), (d) - (iv) done
clear
B)
(a) - (ii), (b) - (iii), (c) - (iv), (d) - (i) done
clear
C)
(a) - (iii), (b) - (iv), (c) - (i), (d) - (ii) done
clear
D)
(a) - (iv), (b) - (i), (c) - (ii), (d) - (iii) done
clear
View Solution play_arrow
-
question_answer12)
|
Element P belongs to first group and fourth period of the periodic table. Element Q succeeds P in the group and element R succeeds Q in the period.
|
|
The correct increasing order of their atomic radii is
|
A)
Q < R < P done
clear
B)
P < Q < R done
clear
C)
P < R < Q done
clear
D)
Cannot be predicted. done
clear
View Solution play_arrow
-
question_answer13)
Study the given flow chart carefully and fill in the blanks by choosing an appropriate option.
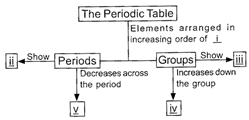
A)
B)
C)
D)
View Solution play_arrow
-
question_answer14)
The electron affinity of
A)
Carbon is greater than oxygen done
clear
B)
Oxygen is greater than fluorine done
clear
C)
Iodine is greater than bromine done
clear
D)
Bromine is less than chlorine. done
clear
View Solution play_arrow
-
question_answer15)
The given table shows a part of the periodic table
| Groups\[\to \] Periods\[\downarrow \] | 1 | 2 | 3 to 12 | 13 | 14 | 15 | 16 | 17 | 18 |
| 2 | | | | | | | Q | | R |
| 3 | | P | S | | | T | | | U |
Identify P, Q, R, T and U respectively.
A)
B)
C)
D)
View Solution play_arrow
-
question_answer16)
Electronic configurations of elements P, Q, R and S are given below :
| P : 2, 8, 1; |
Q : 2, 5 |
| R : 2, 8, 7; |
S : 2, 8, 5 |
Which of the following statements are correct? Elements O and S belong to the same
| I. period of the periodic table. |
| II. P is an alkali metal. |
| III. R is a metal. |
| IV. Valency of element S is 3. |
A)
I and II only done
clear
B)
II and IV only done
clear
C)
I and III only done
clear
D)
All of these done
clear
View Solution play_arrow
-
question_answer17)
A brief information about elements W, X, Y and Z is given below:
|
W : It has two electron shells both of which are completely filled.
|
|
X : It has three electron shells in total and there are four electrons in the valence shell.
|
|
Y : It has three electron shells in total and first and third shell have two electrons each.
|
|
Z : It has twice as many electrons in its second shell as in its first shell and second shell is the valence shell.
|
Identify W, X, Y and Z.
A)
B)
C)
D)
View Solution play_arrow
-
question_answer18)
The given part of the periodic table represents period 3 containing elements a to h.
| Group |
1 |
2 |
13 |
14 |
15 |
16 |
17 |
18 |
| Period 3 |
a |
b |
c |
d |
e |
f |
g |
h |
Which of the following statements is/are correct?
| I. a and b form basic oxides while c forms amphoteric oxide. |
| II. d is a typical non-metal. |
| III. h exists in monoatomic form. |
| IV. f is a non-metal. |
A)
I only done
clear
B)
II only done
clear
C)
I and III only done
clear
D)
I, III and IV only done
clear
View Solution play_arrow
-
question_answer19)
A brief information about the element X is given below :
| Element X |
\[I{{E}_{1}}\] |
\[I{{E}_{2}}\] |
\[I{{E}_{3}}\] |
\[I{{E}_{4}}\] |
| |
496 kJ/mol |
4564 kJ/mol |
6918 kJ/mol |
9542 kJ/mol |
The formulae of the sulphate and chloride of element X are respectively
A)
\[XS{{O}_{4}}\] and \[XC{{l}_{2}}\] done
clear
B)
\[X{{(S{{O}_{4}})}_{2}}\] and \[{{X}_{2}}Cl\] done
clear
C)
\[{{X}_{2}}S{{O}_{4}}\] and\[XCl\] done
clear
D)
\[XS{{O}_{4}}\] and \[{{X}_{2}}Cl\] done
clear
View Solution play_arrow
-
question_answer20)
The positions of four elements K, L, M and N in the periodic table are shown below:
| Group 13 |
Group 14 |
Group 15 |
| K |
- |
- |
| - |
L |
- |
| Eka-aluminium |
M |
N |
Which of the following statements about these elements are correct?
| I. K, L, M and N are all metalloids. |
| II. K is a metal while L, M and N are non-metals. |
| III. Among these four elements, K is smallest in size. |
| IV. K is a metal, L and M are metalloids, N is a non-metal. |
A)
Only II and III done
clear
B)
Only I and III done
clear
C)
Only III and IV done
clear
D)
None of these. done
clear
View Solution play_arrow
