Nomenclature Of Simple Aromatic Compounds
Category : JEE Main & Advanced
Aromatic compounds are those which contain one or more benzene rings in them. An aromatic compound has two main parts : (1) Nucleus, (2) Side chain
(1) Nucleus :The benzene ring represented by regular hexagon of six carbon atoms with three double bonds in the alternate positions is referred to as nucleus. The ring may be represented by any of the following ways,
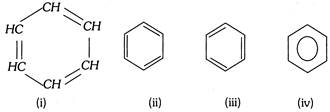
(2) Side chain : The alkyl or any other aliphatic group containing at least one carbon atom attached to the nucleus is called side chain. These are formed by replacing one or more hydrogen atoms in the ring by alkyl radicals i.e., R (R may be \[-C{{H}_{3}}\], \[-{{C}_{2}}{{H}_{5}}\], \[-{{C}_{3}}{{H}_{7}}\] etc.)

If one atom of hydrogen of benzene molecule is replaced by another atom or group of atoms, the derivative formed is called monovalent substituted derivative. It can exist only in one form because all the six hydrogens of benzene represent equivalent positions. For example, \[{{C}_{6}}{{H}_{5}}X\], where X is a monovalent group.
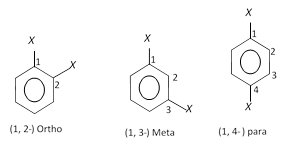
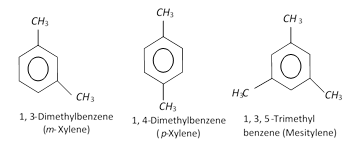
When two hydrogen atoms of benzene are replaced by two monovalent atoms or group of atoms, the resulting product disubstituted benzene derivative can have three different forms. These forms are distinguished by giving the numbers. The position occupied by the principle functional group is given as 1 and the other position is numbered in a clockwise direction which gvies lower locatant to the substituents.
(i) Ortho (or 1, 2-) : The compound is said to be ortho (or 1, 2-) if the two substituents are on the adjacent carbon atoms.
(ii) Meta (or 1, 3-) : The compound is said to be meta or (1, 3-) if the two substituents are on alternate carbon atoms.
(iii) Para (or 1, 4-) : The compound is said to be para or (1, 4-) if the two substituents are on diagonally situated carbon atoms.
Ortho, meta and para are generally represented as o-, m- and p- respectively as shown below,
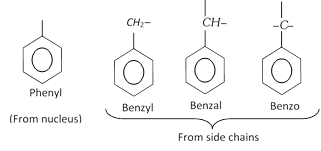
Aryl group : The radicals obtained by removal of one or more hydrogen atoms of the aromatic hydrocarbon molecules are known as aryl radicals or aryl groups. For example,
Nomenclature of different aromatic compounds : The names of few simple aromatic compounds are given below :
Hydrocarbons
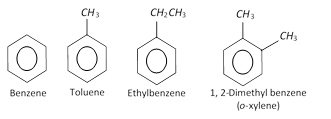
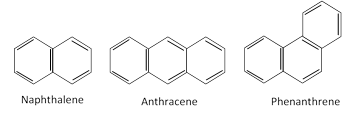
The aromatic hydrocarbons may also contain two or more benzene rings condensed together.
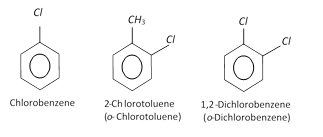
Halogen derivatives
Nuclear substituted
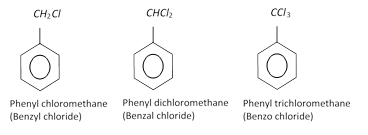
 Side chain substituted
Side chain substituted
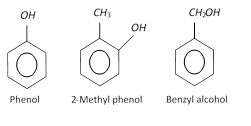
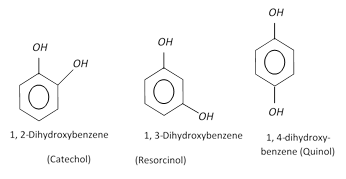
 Hydroxy derivatives
Hydroxy derivatives
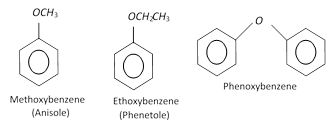
Ethers
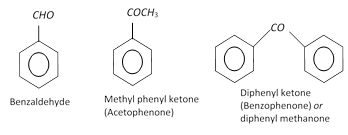
Aldehydes and ketones (Nuclear substituted)
Carboxylic acids (Nuclear substituted)
Acid derivatives
Amino derivatives (Nuclear substituted)
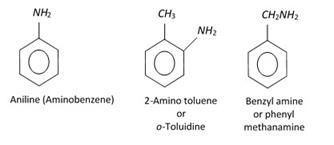
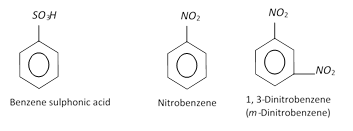
Sulphonic acids Nitro derivatives
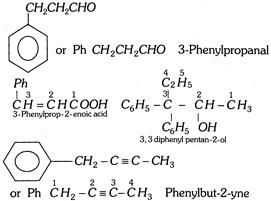
Some tips for nomenclature of aromatic compounds : For IUPAC nomenclature of substituted benzene compounds, the substituent is placed as prefix to the word benzene. It may be noted that common names of many substituted benzene compounds are still universally used. Some important tips for nomenclature of organic compounds are given below,
(i) When the benzene ring is named as substituent on the other molecule, it is named as phenyl group. It is treated in the nomenclature just like the name of an alkyl group. It is abbreviated as Ph. For example,
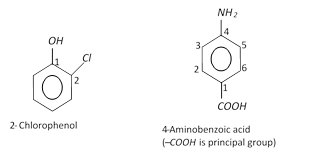
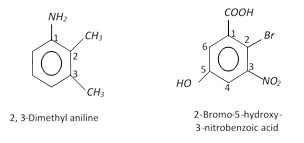
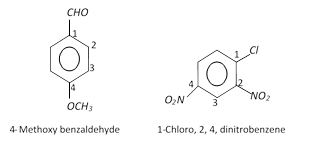
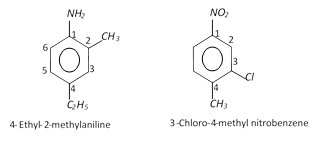
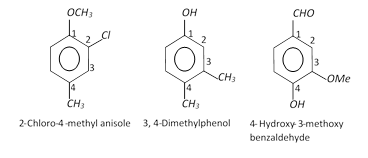
(ii) Disubstituted, trisubstituted or tetrasubstituted benzenes are named by using the numbers for the positions of the substituents.

(iii) If different groups are attached to the benzene ring, then the following rules are kept in mind,
(a) The principal group is fixed as number 1.
(b) The numbering of the chain is done in any direction (clockwise or anticlockwise) which gives lower number to the substituents.
(c) The substituents are written in alphabetical order. For example,
You need to login to perform this action.
You will be redirected in
3 sec
