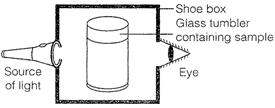
 (a) Explain why the milk sample was illuminated? Name the
phenomenon involved.
(b) Same results were not observed with a salt solution.
Explain.
(c) Can you suggest two more solutions which would show
the same effect as shown by the milk solution?
(a) Explain why the milk sample was illuminated? Name the
phenomenon involved.
(b) Same results were not observed with a salt solution.
Explain.
(c) Can you suggest two more solutions which would show
the same effect as shown by the milk solution?
Answer:
(a) The milk sample was illuminated because milk is a
colloidal solution and its particles are big enough to scatter the light,
hence, they scatter the light passing through it. The phenomenon observed is
called "Tyndall effect".
(b) As salt solution is a true solution i.e., solute
particle size is too small to scatter the light, hence, it does not show
"Tyndall effect".
(c) Examples of colloid are gold sol, arsenius sulphide ![]() sol.,
Blood etc,
sol.,
Blood etc,
You need to login to perform this action.
You will be redirected in
3 sec
