Answer:
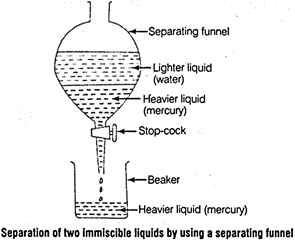
(a) Mercury and water are separated by decantation by using separating
funnel as the separation of two immiscible liquids by a separating funnel
depends on the difference in their densities. Mercury is heavier than water
therefore, forms the lower layer and separated from water.
(b) Potassium chloride and ammonium chloride are separated
by sublimation method
because ammonium chloride being a sublimate, sublimes
leaving behind the potassium chloride.
Sublimation is the process in which a solid changes
directly into vapours on heating and vapours change into solid on cooling
without going in liquid state.
 (b) Potassium chloride and ammonium chloride are separated
by sublimation method because ammonium chloride being a sublimate, sublimes
leaving behind the potassium chloride.
Sublimation is the process in which a solid changes
directly into vapours on heating and vapours change into solid on cooling
without going in liquid state.
(b) Potassium chloride and ammonium chloride are separated
by sublimation method because ammonium chloride being a sublimate, sublimes
leaving behind the potassium chloride.
Sublimation is the process in which a solid changes
directly into vapours on heating and vapours change into solid on cooling
without going in liquid state.
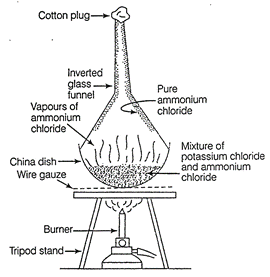 Separation of
potassium chloride and ammonium chloride by sublimation
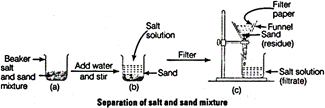
(c) Common salt, water and sand are separated by
(i) Decantation (or filtration) process is used to separate sand from
common salt solution in water because common salt is soluble in water whereas
sand is insoluble in water. Therefore, by filtration, sand will be separated as
insoluble substance as residue and filtrate will be common salt solution in
water.
Separation of
potassium chloride and ammonium chloride by sublimation
(c) Common salt, water and sand are separated by
(i) Decantation (or filtration) process is used to separate sand from
common salt solution in water because common salt is soluble in water whereas
sand is insoluble in water. Therefore, by filtration, sand will be separated as
insoluble substance as residue and filtrate will be common salt solution in
water.
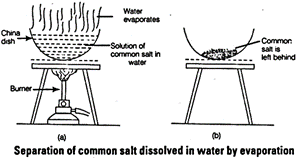
 (ii) Evaporation process is used to separate common salt from water.
Water evaporates and common salt remains as residue.
(ii) Evaporation process is used to separate common salt from water.
Water evaporates and common salt remains as residue.
 (d) (i) Decantation by using separating funnel is used to separate
kerosene nil from salt solution in water as they form separate layers (salt is
soluble in water).
(ii) Evaporation is used to separate salt from water.
(d) (i) Decantation by using separating funnel is used to separate
kerosene nil from salt solution in water as they form separate layers (salt is
soluble in water).
(ii) Evaporation is used to separate salt from water.
You need to login to perform this action.
You will be redirected in
3 sec
