-
In a reaction, 5.3 g of sodium carbonate reacted with 6 g of ethanoic acid. The products were 2.2 g of carbon dioxide, 0.9 g water and 8.2 g of sodium ethanoate. Show that the observations are in agreement with the law of conservation of mass.
Sodium carbonate + ethanoic acid → sodium ethanoate + carbon dioxide + water
View Answer play_arrow
-
Hydrogen and oxygen combine in the ratio of 1 : 8 by mass to form water. What mass of oxygen gas would be required to react completely with 3 g of hydrogen gas?
View Answer play_arrow
-
Which postulate of Dalton's atomic theory is the result of the law of conservation of mass?
View Answer play_arrow
-
Which postulate of Dalton's atomic theory can explain the law of definite proportions?
View Answer play_arrow
-
Define the atomic mass unit.
View Answer play_arrow
-
Why is it not possible to see an atom with naked eyes?
View Answer play_arrow
-
Write down the formulae of (i) sodium oxide, (ii) aluminium chloride, (iii) sodium sulphide, (iv) magnesium hydroxide.
View Answer play_arrow
-
Write down the names of compounds represented by the following formulae:
(i)Al2(SO4)3
(ii)CaCl2
(iii)K2SO4
(iv)KNO3
(v)CaCO3
View Answer play_arrow
-
What is meant by the term chemical formulae?
View Answer play_arrow
-
How many atoms are present in a
(i) H2S molecule and (ii) PO43- ion?
View Answer play_arrow
-
Calculate the molecular masses of H2, O2, Cl2, CO2, CH4, C2H6, C2H4, NH3, CH3OH.
View Answer play_arrow
-
Calculate the formula unit masses of ZnO, Na2O, K2CO3. Given atomic masses of Zn = 65 u, Na = 23 u, K= 39 u, C =12 u and O = 16 u.
View Answer play_arrow
-
If one mole of carbon atoms weighs 12 gram, what is the mass (in gram) of 1 atom of carbon?
View Answer play_arrow
-
Which has more number of atoms, 100 grams of sodium or 100 grams of iron (given atomic mass of Na = 23 u, Fe = 56 u) ?
View Answer play_arrow
-
A 0.24 g sample of compound of oxygen and boron was found by analysis to contain 0.096 g of boron and 0.144 g of oxygen. Calculate the percentage composition of the compound by weight.
View Answer play_arrow
-
When 3.0 g of carbon is burnt in 8.00 g oxygen, 11.00 g of carbon dioxide is produced. What mass of carbon dioxide will be formed when 3.00 g of carbon is burnt in 50.00 g of oxygen ?
Which law of chemical combination will govern your answer?
View Answer play_arrow
-
What are polyatomic ions? Give examples.
View Answer play_arrow
-
Write the chemical formulae of the following:
(a) Magnesium chloride
(b) Calcium oxide
(c) Copper nitrate
(d) Aluminium chloride
(e) Calcium carbonate
View Answer play_arrow
-
Give the names of the elements present in the following compounds.
(a) Quick lime (b) Hydrogen bromide
(c) Baking powder (d) Potassium sulphate
View Answer play_arrow
-
Calculate the molar mass of the following substances.
(a) Ethyne, C2H2
(b) Sulphur molecule, S8
(c) Phosphorus molecule, P4 (atomic mass of phosphorus = 31)
(d) Hydrochloric acid, HCl
(e) Nitric acid, HNO3.
View Answer play_arrow
-
What is the mass of
(a) 1 mole of nitrogen atoms?
(b) 4 moles of aluminium atoms (atomic mass of aluminium = 27)?
(c) 10 moles of sodium sulphite (Na2SO3) ?
View Answer play_arrow
-
Convert into mole.
(a) 12 g of oxygen gas
(b) 20 g of water
(c) 22 g of carbon dioxide
View Answer play_arrow
-
What is the mass of
(a) 0.2 mole of oxygen atoms?
(b) 0.5 mole of water molecules?
View Answer play_arrow
-
Calculate the number of molecules of sulphur (S8) present in 16 g of solid sulphur?
View Answer play_arrow
-
Calculate the number of aluminium ions present in 0.051 g of aluminium oxide.
(Hint: The mass of an ion is the same as that of an atom of the same element. Atomic mass of Al = 27 u)
View Answer play_arrow
-
question_answer26)
Which of the following correctly represents 360 g of water?
(i) 2 moles of 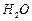 (ii)
20 moles of water
(iii) 6.022 x 1023 molecules of water (iv)
1.2044 x 1025 molecules of water
(a) only (i) (b) (i) and (iv) (c)
(ii) and (iii) (d) (ii) and (iv)
(ii)
20 moles of water
(iii) 6.022 x 1023 molecules of water (iv)
1.2044 x 1025 molecules of water
(a) only (i) (b) (i) and (iv) (c)
(ii) and (iii) (d) (ii) and (iv)
View Answer play_arrow
-
question_answer27)
Which of the following statements is not true about an
atom?
(a) Atoms are not able to exist independently
(b) Atoms are the basic units from which molecules and
ions are formed
(c) Atoms are always neutral in nature
(d) Atoms aggregate in large numbers to form the matter
that we can see, feel or touch
View Answer play_arrow
-
question_answer28)
The chemical symbol for nitrogen gas is
(a) Ni (b)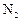 (c)
(c)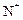 (d) N
(d) N
View Answer play_arrow
-
question_answer29)
The chemical symbol for sodium is
(a) So (b)
Sd (c) NA (d)
Na
View Answer play_arrow
-
question_answer30)
Which of the following would weigh the highest?
(a) 0.2 mole of sucrose 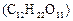 (b)
2 moles of
(b)
2 moles of  (c) 2 moles of
(c) 2 moles of  (d)
10 moles of
(d)
10 moles of 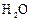
View Answer play_arrow
-
question_answer31)
Which of the following has maximum number of atoms?
(a) 18 g of 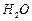 (b)
18 g of
(b)
18 g of 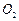 (c) 18 g of
(c) 18 g of 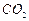 (d)
18 g of
(d)
18 g of 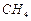
View Answer play_arrow
-
question_answer32)
Which of the following contains maximum number of molecules?
(a)  (b)
(b)
 (c)
(c) 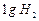 (d)
(d)
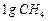
View Answer play_arrow
-
question_answer33)
Mass of one atom of oxygen is
(a) 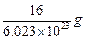 (b)
(b)
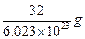 (c)
(c) 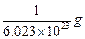 (d)
8 u
(d)
8 u
View Answer play_arrow
-
question_answer34)
3.42 g of sucrose are dissolved in 18 g of water in a
beaker. The number of oxygen atoms in the solution are
(a) 6.68 x1023 (b) 6.09
x1022 (c) 6.022 x 10 (d)
6.022 x1021
View Answer play_arrow
-
question_answer35)
A change in the physical state can be brought about
(a) only when energy is given to the system
(b) only when energy is taken out from the system
(c) when energy is either given to, or taken out from the
system
(d) without any energy change
View Answer play_arrow
-
question_answer36)
Which of the following represents a correct chemical
formula? Name it.
(a) CaCI (b) 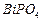 (c)
(c)
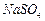 (d)
NaS
(d)
NaS
View Answer play_arrow
-
question_answer37)
Write the molecular formulae for the following compounds
(a) Copper (II) bromide (b)
Aluminium (III) nitrate
(c) Calcium (II) phosphate (d)
Iron (III) sulphide
(e) Mercury (II) chloride (f)
Magnesium (II) acetate
View Answer play_arrow
-
question_answer38)
Write the molecular formulae of all the
compounds that can be formed by the combination of following ions.
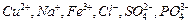
View Answer play_arrow
-
question_answer39)
Write the cat ions and anions present (if any) in the following compounds.
(a)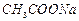 (b)
(b) (c)
(c)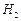 (d)
(d)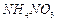
View Answer play_arrow
-
question_answer40)
Give the formulae of the compounds formed from the
following sets of elements.
(a) Calcium and fluorine (b)
Hydrogen and sulphur
(c) Nitrogen and hydrogen (d)
Carbon and chlorine
(e) Sodium and oxygen (f)
Carbon and oxygen
View Answer play_arrow
-
question_answer41)
Which of the following symbols of elements are incorrect?
Give their correct symbols.
(a) Cobalt CO (b) Carbon c (c)
Aluminium AL (d) Helium He
(e) Sodium So
View Answer play_arrow
-
question_answer42)
Give the chemical formulae for the following compounds and
compute the ratio by mass of the combining elements in each one of them.
(a) Ammonia (b) Carbon monoxide (c)
Hydrogen chloride (d) Aluminium fluoride
(e) Magnesium sulphide
View Answer play_arrow
-
question_answer43)
State the number of atoms present in each of the following
chemical species
(a)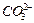 (b)
(b)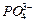 (c)
(c)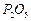 (d)
(d)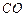
View Answer play_arrow
-
question_answer44)
What is the fraction of the mass of water due to neutrons?
View Answer play_arrow
-
question_answer45)
Does the solubility of a substance change with
temperature? Explain with the help of an example.
View Answer play_arrow
-
question_answer46)
Classify each of the following on the basis of their
atomicity.
(a)  (b)
(b)
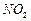 (c)
(c)
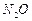 (d)
(d)
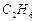 (e)
(e) (f)
(f) 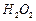 (g)
(g)
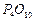 (h)
(h)
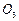 (i) HCI (j)
(i) HCI (j)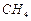 (k)
He (I) Ag
(k)
He (I) Ag
View Answer play_arrow
-
question_answer47)
You are provided with a fine white coloured powder which
is either sugar or salt. How would you identify it without testing?
View Answer play_arrow
-
question_answer48)
Calculate the number of moles of magnesium present in a
magnesium ribbon weighing 12g. Molar atomic mass of magnesium is 24 g 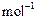 .
.
View Answer play_arrow
-
question_answer49)
Verify by calculating that
(a) 5 moles of 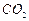 and
5 moles of
and
5 moles of  do not have
the same mass.
(b) 240 g of calcium and 240 g magnesium elements have a
mole ratio of 3 : 5.
do not have
the same mass.
(b) 240 g of calcium and 240 g magnesium elements have a
mole ratio of 3 : 5.
View Answer play_arrow
-
question_answer50)
Find the ratio by mass of the combining elements in the
following compounds.
(a) 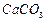 (b)
(b) (c)
(c) 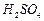 (d)
(d)
 (e)
(e) 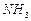 (f)
(f)
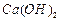
View Answer play_arrow
-
question_answer51)
Calcium chloride when dissolved in water dissociates into
its ions according to the following equation.
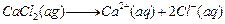 Calculate the number of ions obtained from
Calculate the number of ions obtained from 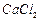 when 222 g
of it is dissolved in water.
when 222 g
of it is dissolved in water.
View Answer play_arrow
-
question_answer52)
The difference in the mass of 100 moles each of sodium
atoms and sodium ions is 5.48002 g. Compute the mass of an electron.
View Answer play_arrow
-
question_answer53)
Cinnabar (HgS) is a prominent ore of mercury. How many
grams of mercury are present in 225 g of pure HgS? Molar mass of Hg and S are 200.6
g 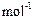 and 32g
and 32g 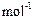 respectively.
respectively.
View Answer play_arrow
-
question_answer54)
The mass of one steel screw is 4.11 g. Find the mass of
one mole of these steel screws. Compare this value with the mass of the earth (5.98
x 1024 kg). Which one of the two is heavier and by how many times?
View Answer play_arrow
-
question_answer55)
A sample of vitamin C is known to contain 2.58 x 1024
oxygen atoms.
How many moles of oxygen atoms are present in the sample?
View Answer play_arrow
-
question_answer56)
Raunak took 5 moles of carbon atoms in a container and Krish also took 5
moles of sodium atoms in another container of same weight.
(a) Whose container is heavier?
(b) Whose container has more number of atoms?
View Answer play_arrow
-
question_answer57)
Fill in the missing data in the following table.
|
Species property
|
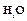
|
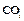
|
Na-atom
|

|
|
Number of particles
|
2
|
-
|
-
|
0.5
|
|
Number of particles
|
-
|
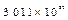
|
-
|
-
|
|
Mass
|
36 g
|
-
|
115 g
|
-
|
View Answer play_arrow
-
question_answer58)
The visible universe is estimated to contain 1022
stars. How many moles of stars are present in the visible universe?
View Answer play_arrow
-
question_answer59)
What is SI prefix for each of the following multiples and
submultiples of a unit?
(a) 103 (b)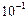 (c)
(c)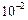 (d)
(d)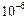 (e)
(e) 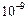 (f)
(f)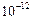
View Answer play_arrow
-
question_answer60)
Express each of the following in kilograms
(a) 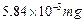 (b)
58.34 g (c) 0.584 g (d)
(b)
58.34 g (c) 0.584 g (d) 
View Answer play_arrow
-
question_answer61)
Compute the difference in masses of 103 moles
each of magnesium atoms and magnesium ions.
(Mass of an electron 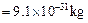 )
)
View Answer play_arrow
-
question_answer62)
Which has more number of atoms?
100 g of N3 or 100 g of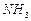
View Answer play_arrow
-
question_answer63)
Compute the number of ions present in 5.85 g of sodium
chloride.
View Answer play_arrow
-
question_answer64)
A gold sample contains 90% of gold and the rest copper.
How many atoms of gold are present in one gram of this sample of gold?
View Answer play_arrow
-
question_answer65)
What are ionic and molecular compounds? Give examples.
View Answer play_arrow
-
question_answer66)
Compute the difference in masses of one mole each of
aluminium atoms and one mole of its ions (Mass of an electron is 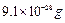 ). Which one
is heavier?
). Which one
is heavier?
View Answer play_arrow
-
question_answer67)
A silver ornament of mass 'm' gram is polished with gold
equivalent to 1% of the mass of silver. Compute the ratio of the number of
atoms of gold and silver in the ornament.
View Answer play_arrow
-
question_answer68)
A sample of ethane 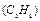 gas has the
same mass as 1.5 x 1020 molecules of methane
gas has the
same mass as 1.5 x 1020 molecules of methane 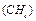 . How many
. How many 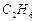 molecules
does the sample of gas contain?
molecules
does the sample of gas contain?
View Answer play_arrow
-
question_answer69)
Fill in the blanks.
(a) In a chemical reaction, the sum of the masses of the
reactants and products remains unchanged. This is called ......... ,
(b) A group of atoms carrying a fixed charge on them is
called ......... .
(c) The formula unit mass of 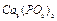 is??.
(d) Formula of sodium carbonate is ......... and that of
ammonium sulphate is ......... .
is??.
(d) Formula of sodium carbonate is ......... and that of
ammonium sulphate is ......... .
View Answer play_arrow
-
question_answer70)
Complete the following crossword puzzle (figure) by using the name of the
chemical elements. Use the data given in table following.
|
Across
|
Down
|
|
2. The
element used by Rutherford during his a- scattering
experiment.
|
1. A white
lustrous metal used for making ornaments and which tends to get tarnished
black in the presence of moist air.
|
|
3. An element which forms rust on exposure to moist
air.
|
4. Both brass and bronze are alloys of the element.
|
|
5. A very reactive non-metal stored under water.
|
6. The
metal which exists in the liquid state at room temperature.
|
|
7. Zinc
metal when treated with dilute hydrochloric acid produces a gas of this
element which when tested with burning splinter produces a pop sound.
|
8. An
element with symbol Pb.
|
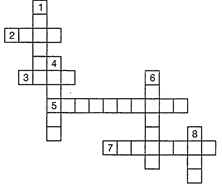 View Answer play_arrow
View Answer play_arrow
-
question_answer71)
(a) In this crossword puzzle (Figure), names of 11 elements are hidden.
Symbols of these are given below. Complete the puzzle.
1. Cl 2. H 3. Ar 4.
0
5. Xe 6. N 7. He
8. F
9. Kr 10. Rn 11. Ne
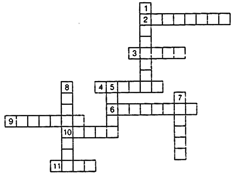 (b) Identify the total number of inert gases, their names
and symbols from this cross ward puzzle.
(b) Identify the total number of inert gases, their names
and symbols from this cross ward puzzle.
View Answer play_arrow
-
question_answer72)
Write the formulae for the following and calculate the
molecular mass for each one of them.
(a) Caustic potash (b) Baking powder (c)
Lime stone (d) Caustic soda
(e) Ethanol (f) Common salt
View Answer play_arrow
-
question_answer73)
In photosynthesis, 6 molecules of carbon dioxide combine
with an equal number of water molecules through a complex series of reactions
to give a molecule of glucose having a molecular formula 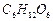 . How many grams
of water would be required to produce 18 g of glucose? Compute the volume of
water so consumed assuming the density of water to be
. How many grams
of water would be required to produce 18 g of glucose? Compute the volume of
water so consumed assuming the density of water to be 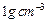 .
.
View Answer play_arrow
![]() (c)
(c)![]() (d) N
(d) N
![]() (b)
(b)
![]() (c)
(c) ![]() (d)
(d)
![]()
![]() (b)
(b)
![]() (c)
(c) ![]() (d)
8 u
(d)
8 u
![]() (b)
58.34 g (c) 0.584 g (d)
(b)
58.34 g (c) 0.584 g (d) ![]()
![]()
 View Answer play_arrow
View Answer play_arrow
