Answer:
Certain compounds when dissolved in
suitable solvents either dissociate or associate. e.g., ethanoic acid dimerises
in benzene due to H-bonding, while in water, it dissociates and forms
ions.
As a result the number of chemical
species in solution increases or decreases as compared to the number of
chemical species of solute added to form the solution.
Since, the magnitude of
colligative property depends on the number of solute particles, it is
expected that the molar mass
determined on the basis of colligative properties will either higher or lower
than the expected value or the normal value and is called abnormal molar mass.
In order to account for the
extent of dissociation or association of molecules in solution, van't Hoff
introduced a factor, i, known as the van't Hoff factor.
![]()
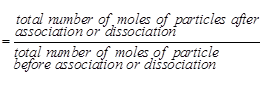
You need to login to perform this action.
You will be redirected in
3 sec
